Vishwesh Nath
LUMEN: Longitudinal Multi-Modal Radiology Model for Prognosis and Diagnosis
Feb 24, 2026Abstract:Large vision-language models (VLMs) have evolved from general-purpose applications to specialized use cases such as in the clinical domain, demonstrating potential for decision support in radiology. One promising application is assisting radiologists in decision-making by the analysis of radiology imaging data such as chest X-rays (CXR) via a visual and natural language question-answering (VQA) interface. When longitudinal imaging is available, radiologists analyze temporal changes, which are essential for accurate diagnosis and prognosis. The manual longitudinal analysis is a time-consuming process, motivating the development of a training framework that can provide prognostic capabilities. We introduce a novel training framework LUMEN, that is optimized for longitudinal CXR interpretation, leveraging multi-image and multi-task instruction fine-tuning to enhance prognostic and diagnostic performance. We conduct experiments on the publicly available MIMIC-CXR and its associated Medical-Diff-VQA datasets. We further formulate and construct a novel instruction-following dataset incorporating longitudinal studies, enabling the development of a prognostic VQA task. Our method demonstrates significant improvements over baseline models in diagnostic VQA tasks, and more importantly, shows promising potential for prognostic capabilities. These results underscore the value of well-designed, instruction-tuned VLMs in enabling more accurate and clinically meaningful radiological interpretation of longitudinal radiological imaging data.
Text2CT: Towards 3D CT Volume Generation from Free-text Descriptions Using Diffusion Model
May 07, 2025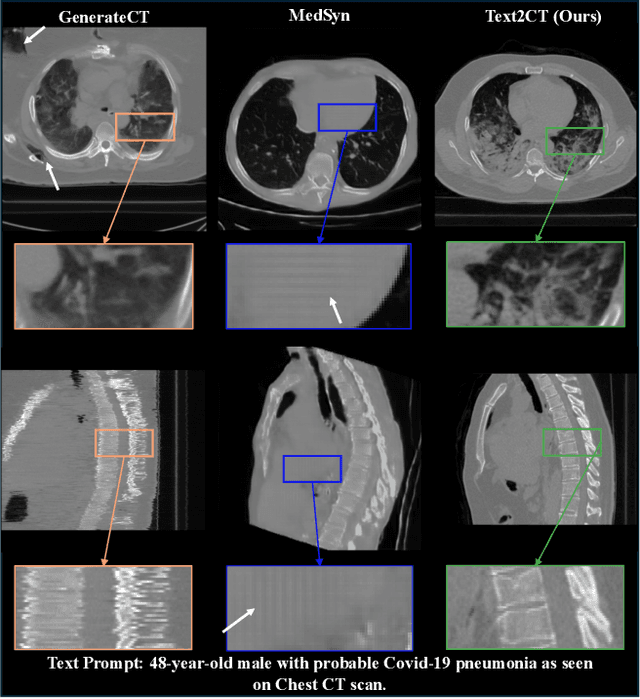
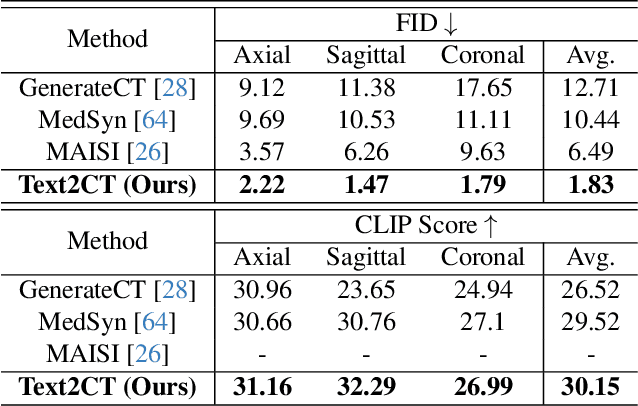
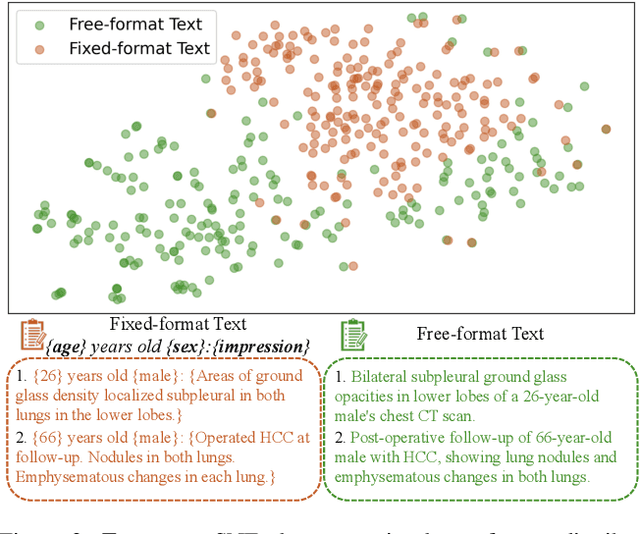
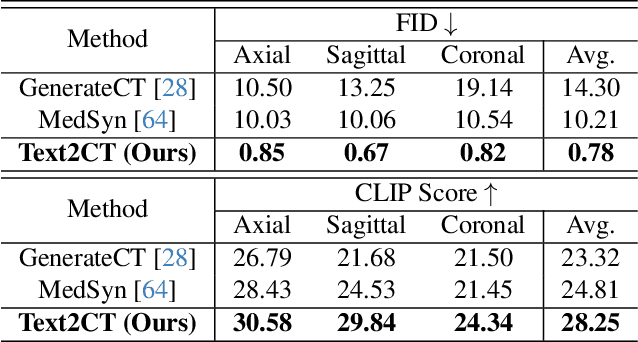
Abstract:Generating 3D CT volumes from descriptive free-text inputs presents a transformative opportunity in diagnostics and research. In this paper, we introduce Text2CT, a novel approach for synthesizing 3D CT volumes from textual descriptions using the diffusion model. Unlike previous methods that rely on fixed-format text input, Text2CT employs a novel prompt formulation that enables generation from diverse, free-text descriptions. The proposed framework encodes medical text into latent representations and decodes them into high-resolution 3D CT scans, effectively bridging the gap between semantic text inputs and detailed volumetric representations in a unified 3D framework. Our method demonstrates superior performance in preserving anatomical fidelity and capturing intricate structures as described in the input text. Extensive evaluations show that our approach achieves state-of-the-art results, offering promising potential applications in diagnostics, and data augmentation.
NVILA: Efficient Frontier Visual Language Models
Dec 05, 2024
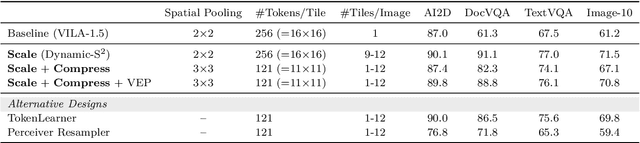

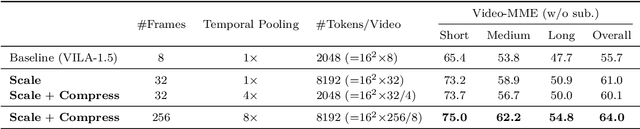
Abstract:Visual language models (VLMs) have made significant advances in accuracy in recent years. However, their efficiency has received much less attention. This paper introduces NVILA, a family of open VLMs designed to optimize both efficiency and accuracy. Building on top of VILA, we improve its model architecture by first scaling up the spatial and temporal resolutions, and then compressing visual tokens. This "scale-then-compress" approach enables NVILA to efficiently process high-resolution images and long videos. We also conduct a systematic investigation to enhance the efficiency of NVILA throughout its entire lifecycle, from training and fine-tuning to deployment. NVILA matches or surpasses the accuracy of many leading open and proprietary VLMs across a wide range of image and video benchmarks. At the same time, it reduces training costs by 4.5X, fine-tuning memory usage by 3.4X, pre-filling latency by 1.6-2.2X, and decoding latency by 1.2-2.8X. We will soon make our code and models available to facilitate reproducibility.
VILA-M3: Enhancing Vision-Language Models with Medical Expert Knowledge
Nov 19, 2024



Abstract:Generalist vision language models (VLMs) have made significant strides in computer vision, but they fall short in specialized fields like healthcare, where expert knowledge is essential. In traditional computer vision tasks, creative or approximate answers may be acceptable, but in healthcare, precision is paramount.Current large multimodal models like Gemini and GPT-4o are insufficient for medical tasks due to their reliance on memorized internet knowledge rather than the nuanced expertise required in healthcare. VLMs are usually trained in three stages: vision pre-training, vision-language pre-training, and instruction fine-tuning (IFT). IFT has been typically applied using a mixture of generic and healthcare data. In contrast, we propose that for medical VLMs, a fourth stage of specialized IFT is necessary, which focuses on medical data and includes information from domain expert models. Domain expert models developed for medical use are crucial because they are specifically trained for certain clinical tasks, e.g. to detect tumors and classify abnormalities through segmentation and classification, which learn fine-grained features of medical data$-$features that are often too intricate for a VLM to capture effectively especially in radiology. This paper introduces a new framework, VILA-M3, for medical VLMs that utilizes domain knowledge via expert models. Through our experiments, we show an improved state-of-the-art (SOTA) performance with an average improvement of ~9% over the prior SOTA model Med-Gemini and ~6% over models trained on the specific tasks. Our approach emphasizes the importance of domain expertise in creating precise, reliable VLMs for medical applications.
MICCAI-CDMRI 2023 QuantConn Challenge Findings on Achieving Robust Quantitative Connectivity through Harmonized Preprocessing of Diffusion MRI
Nov 14, 2024



Abstract:White matter alterations are increasingly implicated in neurological diseases and their progression. International-scale studies use diffusion-weighted magnetic resonance imaging (DW-MRI) to qualitatively identify changes in white matter microstructure and connectivity. Yet, quantitative analysis of DW-MRI data is hindered by inconsistencies stemming from varying acquisition protocols. There is a pressing need to harmonize the preprocessing of DW-MRI datasets to ensure the derivation of robust quantitative diffusion metrics across acquisitions. In the MICCAI-CDMRI 2023 QuantConn challenge, participants were provided raw data from the same individuals collected on the same scanner but with two different acquisitions and tasked with preprocessing the DW-MRI to minimize acquisition differences while retaining biological variation. Submissions are evaluated on the reproducibility and comparability of cross-acquisition bundle-wise microstructure measures, bundle shape features, and connectomics. The key innovations of the QuantConn challenge are that (1) we assess bundles and tractography in the context of harmonization for the first time, (2) we assess connectomics in the context of harmonization for the first time, and (3) we have 10x additional subjects over prior harmonization challenge, MUSHAC and 100x over SuperMUDI. We find that bundle surface area, fractional anisotropy, connectome assortativity, betweenness centrality, edge count, modularity, nodal strength, and participation coefficient measures are most biased by acquisition and that machine learning voxel-wise correction, RISH mapping, and NeSH methods effectively reduce these biases. In addition, microstructure measures AD, MD, RD, bundle length, connectome density, efficiency, and path length are least biased by these acquisition differences.
* Accepted for publication at the Journal of Machine Learning for Biomedical Imaging (MELBA) https://melba-journal.org/2024/019
MAISI: Medical AI for Synthetic Imaging
Sep 13, 2024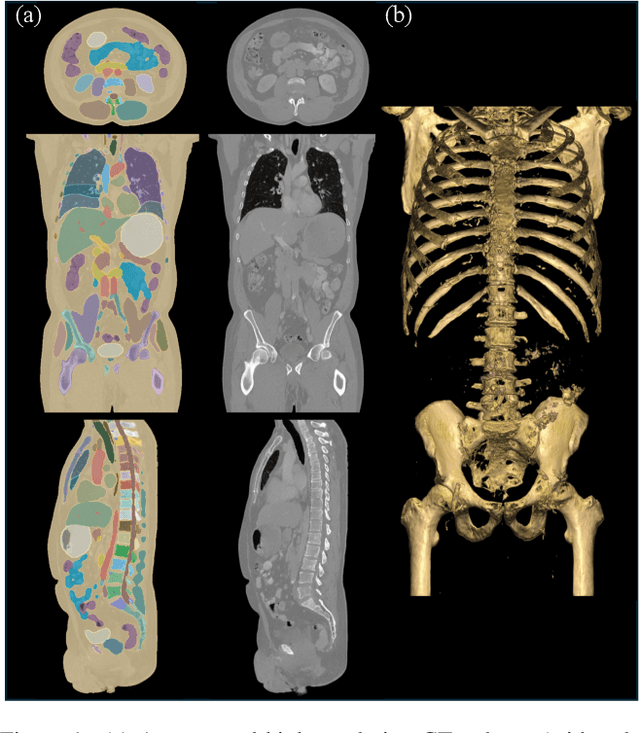
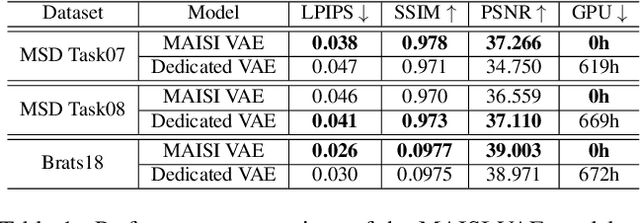
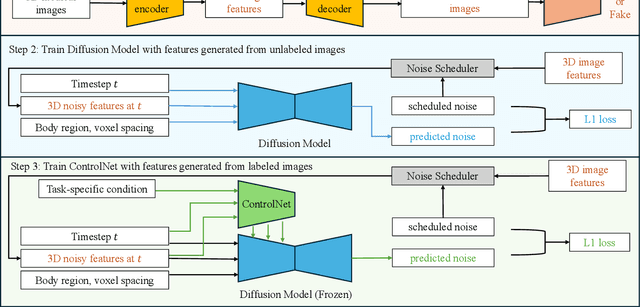
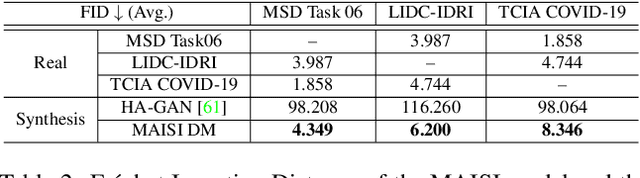
Abstract:Medical imaging analysis faces challenges such as data scarcity, high annotation costs, and privacy concerns. This paper introduces the Medical AI for Synthetic Imaging (MAISI), an innovative approach using the diffusion model to generate synthetic 3D computed tomography (CT) images to address those challenges. MAISI leverages the foundation volume compression network and the latent diffusion model to produce high-resolution CT images (up to a landmark volume dimension of 512 x 512 x 768 ) with flexible volume dimensions and voxel spacing. By incorporating ControlNet, MAISI can process organ segmentation, including 127 anatomical structures, as additional conditions and enables the generation of accurately annotated synthetic images that can be used for various downstream tasks. Our experiment results show that MAISI's capabilities in generating realistic, anatomically accurate images for diverse regions and conditions reveal its promising potential to mitigate challenges using synthetic data.
A Short Review and Evaluation of SAM2's Performance in 3D CT Image Segmentation
Aug 20, 2024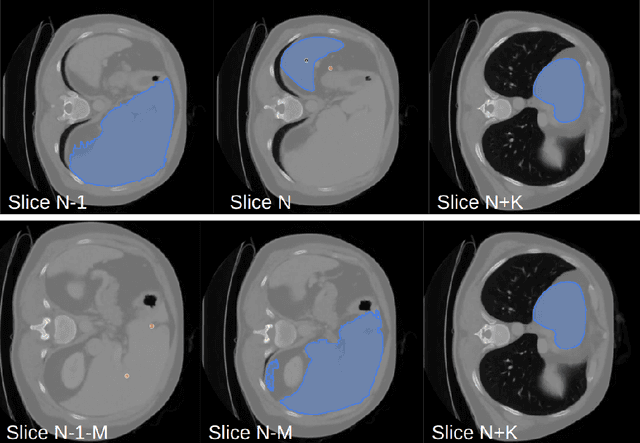
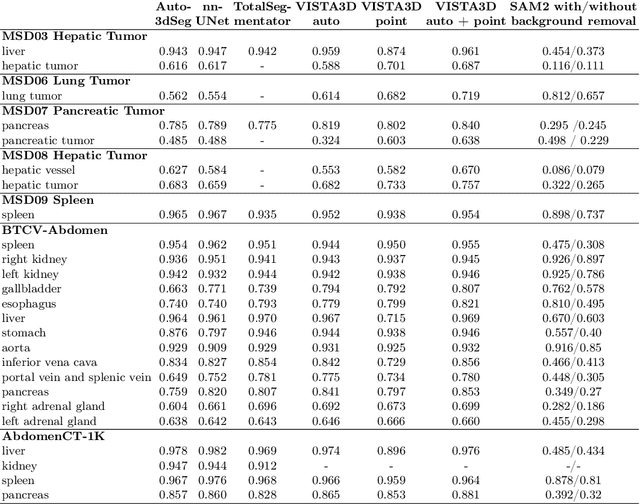
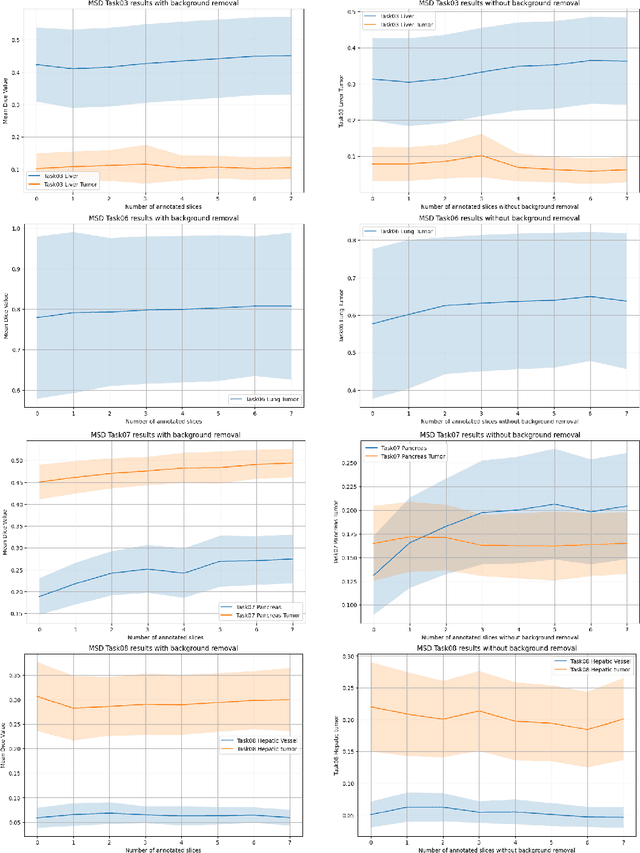
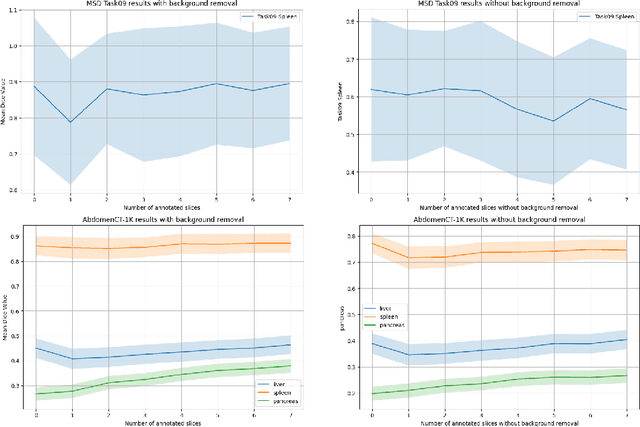
Abstract:Since the release of Segment Anything 2 (SAM2), the medical imaging community has been actively evaluating its performance for 3D medical image segmentation. However, different studies have employed varying evaluation pipelines, resulting in conflicting outcomes that obscure a clear understanding of SAM2's capabilities and potential applications. We shortly review existing benchmarks and point out that the SAM2 paper clearly outlines a zero-shot evaluation pipeline, which simulates user clicks iteratively for up to eight iterations. We reproduced this interactive annotation simulation on 3D CT datasets and provided the results and code~\url{https://github.com/Project-MONAI/VISTA}. Our findings reveal that directly applying SAM2 on 3D medical imaging in a zero-shot manner is far from satisfactory. It is prone to generating false positives when foreground objects disappear, and annotating more slices cannot fully offset this tendency. For smaller single-connected objects like kidney and aorta, SAM2 performs reasonably well but for most organs it is still far behind state-of-the-art 3D annotation methods. More research and innovation are needed for 3D medical imaging community to use SAM2 correctly.
HoloHisto: End-to-end Gigapixel WSI Segmentation with 4K Resolution Sequential Tokenization
Jul 03, 2024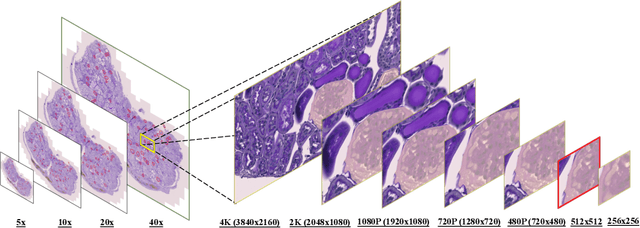
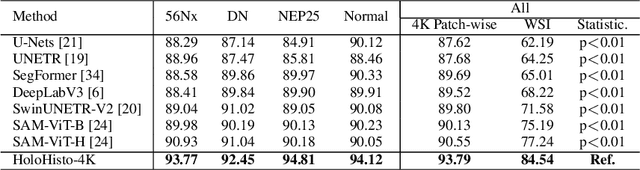
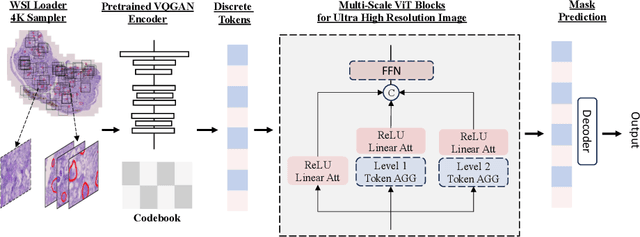
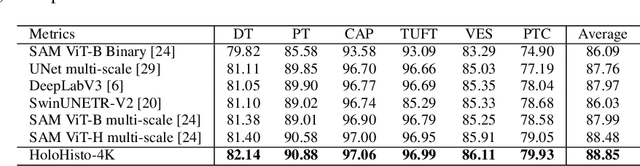
Abstract:In digital pathology, the traditional method for deep learning-based image segmentation typically involves a two-stage process: initially segmenting high-resolution whole slide images (WSI) into smaller patches (e.g., 256x256, 512x512, 1024x1024) and subsequently reconstructing them to their original scale. This method often struggles to capture the complex details and vast scope of WSIs. In this paper, we propose the holistic histopathology (HoloHisto) segmentation method to achieve end-to-end segmentation on gigapixel WSIs, whose maximum resolution is above 80,000$\times$70,000 pixels. HoloHisto fundamentally shifts the paradigm of WSI segmentation to an end-to-end learning fashion with 1) a large (4K) resolution base patch for elevated visual information inclusion and efficient processing, and 2) a novel sequential tokenization mechanism to properly model the contextual relationships and efficiently model the rich information from the 4K input. To our best knowledge, HoloHisto presents the first holistic approach for gigapixel resolution WSI segmentation, supporting direct I/O of complete WSI and their corresponding gigapixel masks. Under the HoloHisto platform, we unveil a random 4K sampler that transcends ultra-high resolution, delivering 31 and 10 times more pixels than standard 2D and 3D patches, respectively, for advancing computational capabilities. To facilitate efficient 4K resolution dense prediction, we leverage sequential tokenization, utilizing a pre-trained image tokenizer to group image features into a discrete token grid. To assess the performance, our team curated a new kidney pathology image segmentation (KPIs) dataset with WSI-level glomeruli segmentation from whole mouse kidneys. From the results, HoloHisto-4K delivers remarkable performance gains over previous state-of-the-art models.
D-Rax: Domain-specific Radiologic assistant leveraging multi-modal data and eXpert model predictions
Jul 02, 2024



Abstract:Large vision language models (VLMs) have progressed incredibly from research to applicability for general-purpose use cases. LLaVA-Med, a pioneering large language and vision assistant for biomedicine, can perform multi-modal biomedical image and data analysis to provide a natural language interface for radiologists. While it is highly generalizable and works with multi-modal data, it is currently limited by well-known challenges that exist in the large language model space. Hallucinations and imprecision in responses can lead to misdiagnosis which currently hinder the clinical adaptability of VLMs. To create precise, user-friendly models in healthcare, we propose D-Rax -- a domain-specific, conversational, radiologic assistance tool that can be used to gain insights about a particular radiologic image. In this study, we enhance the conversational analysis of chest X-ray (CXR) images to support radiological reporting, offering comprehensive insights from medical imaging and aiding in the formulation of accurate diagnosis. D-Rax is achieved by fine-tuning the LLaVA-Med architecture on our curated enhanced instruction-following data, comprising of images, instructions, as well as disease diagnosis and demographic predictions derived from MIMIC-CXR imaging data, CXR-related visual question answer (VQA) pairs, and predictive outcomes from multiple expert AI models. We observe statistically significant improvement in responses when evaluated for both open and close-ended conversations. Leveraging the power of state-of-the-art diagnostic models combined with VLMs, D-Rax empowers clinicians to interact with medical images using natural language, which could potentially streamline their decision-making process, enhance diagnostic accuracy, and conserve their time.
VISTA3D: Versatile Imaging SegmenTation and Annotation model for 3D Computed Tomography
Jun 07, 2024Abstract:Segmentation foundation models have attracted great interest, however, none of them are adequate enough for the use cases in 3D computed tomography scans (CT) images. Existing works finetune on medical images with 2D foundation models trained on natural images, but interactive segmentation, especially in 2D, is too time-consuming for 3D scans and less useful for large cohort analysis. Models that can perform out-of-the-box automatic segmentation are more desirable. However, the model trained in this way lacks the ability to perform segmentation on unseen objects like novel tumors. Thus for 3D medical image analysis, an ideal segmentation solution might expect two features: accurate out-of-the-box performance covering major organ classes, and effective adaptation or zero-shot ability to novel structures. In this paper, we discuss what features a 3D CT segmentation foundation model should have, and introduce VISTA3D, Versatile Imaging SegmenTation and Annotation model. The model is trained systematically on 11454 volumes encompassing 127 types of human anatomical structures and various lesions and provides accurate out-of-the-box segmentation. The model's design also achieves state-of-the-art zero-shot interactive segmentation in 3D. The novel model design and training recipe represent a promising step toward developing a versatile medical image foundation model. Code and model weights will be released shortly. The early version of online demo can be tried on https://build.nvidia.com/nvidia/vista-3d.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge