Gongning Luo
Faculty of Computing, Harbin Institute of Technology, Harbin, China
PCA-Enhanced Probabilistic U-Net for Effective Ambiguous Medical Image Segmentation
Mar 12, 2026Abstract:Ambiguous Medical Image Segmentation (AMIS) is significant to address the challenges of inherent uncertainties from image ambiguities, noise, and subjective annotations. Existing conditional variational autoencoder (cVAE)-based methods effectively capture uncertainty but face limitations including redundancy in high-dimensional latent spaces and limited expressiveness of single posterior networks. To overcome these issues, we introduce a novel PCA-Enhanced Probabilistic U-Net (\textbf{PEP U-Net}). Our method effectively incorporates Principal Component Analysis (PCA) for dimensionality reduction in the posterior network to mitigate redundancy and improve computational efficiency. Additionally, we further employ an inverse PCA operation to reconstruct critical information, enhancing the latent space's representational capacity. Compared to conventional generative models, our method preserves the ability to generate diverse segmentation hypotheses while achieving a superior balance between segmentation accuracy and predictive variability, thereby advancing the performance of generative modeling in medical image segmentation.
MedTri: A Platform for Structured Medical Report Normalization to Enhance Vision-Language Pretraining
Feb 25, 2026Abstract:Medical vision-language pretraining increasingly relies on medical reports as large-scale supervisory signals; however, raw reports often exhibit substantial stylistic heterogeneity, variable length, and a considerable amount of image-irrelevant content. Although text normalization is frequently adopted as a preprocessing step in prior work, its design principles and empirical impact on vision-language pretraining remain insufficiently and systematically examined. In this study, we present MedTri, a deployable normalization framework for medical vision-language pretraining that converts free-text reports into a unified [Anatomical Entity: Radiologic Description + Diagnosis Category] triplet. This structured, anatomy-grounded normalization preserves essential morphological and spatial information while removing stylistic noise and image-irrelevant content, providing consistent and image-grounded textual supervision at scale. Across multiple datasets spanning both X-ray and computed tomography (CT) modalities, we demonstrate that structured, anatomy-grounded text normalization is an important factor in medical vision-language pretraining quality, yielding consistent improvements over raw reports and existing normalization baselines. In addition, we illustrate how this normalization can easily support modular text-level augmentation strategies, including knowledge enrichment and anatomy-grounded counterfactual supervision, which provide complementary gains in robustness and generalization without altering the core normalization process. Together, our results position structured text normalization as a critical and generalizable preprocessing component for medical vision-language learning, while MedTri provides this normalization platform. Code and data will be released at https://github.com/Arturia-Pendragon-Iris/MedTri.
Beyond Benchmarks of IUGC: Rethinking Requirements of Deep Learning Methods for Intrapartum Ultrasound Biometry from Fetal Ultrasound Videos
Feb 13, 2026Abstract:A substantial proportion (45\%) of maternal deaths, neonatal deaths, and stillbirths occur during the intrapartum phase, with a particularly high burden in low- and middle-income countries. Intrapartum biometry plays a critical role in monitoring labor progression; however, the routine use of ultrasound in resource-limited settings is hindered by a shortage of trained sonographers. To address this challenge, the Intrapartum Ultrasound Grand Challenge (IUGC), co-hosted with MICCAI 2024, was launched. The IUGC introduces a clinically oriented multi-task automatic measurement framework that integrates standard plane classification, fetal head-pubic symphysis segmentation, and biometry, enabling algorithms to exploit complementary task information for more accurate estimation. Furthermore, the challenge releases the largest multi-center intrapartum ultrasound video dataset to date, comprising 774 videos (68,106 frames) collected from three hospitals, providing a robust foundation for model training and evaluation. In this study, we present a comprehensive overview of the challenge design, review the submissions from eight participating teams, and analyze their methods from five perspectives: preprocessing, data augmentation, learning strategy, model architecture, and post-processing. In addition, we perform a systematic analysis of the benchmark results to identify key bottlenecks, explore potential solutions, and highlight open challenges for future research. Although encouraging performance has been achieved, our findings indicate that the field remains at an early stage, and further in-depth investigation is required before large-scale clinical deployment. All benchmark solutions and the complete dataset have been publicly released to facilitate reproducible research and promote continued advances in automatic intrapartum ultrasound biometry.
Fully Kolmogorov-Arnold Deep Model in Medical Image Segmentation
Feb 03, 2026Abstract:Deeply stacked KANs are practically impossible due to high training difficulties and substantial memory requirements. Consequently, existing studies can only incorporate few KAN layers, hindering the comprehensive exploration of KANs. This study overcomes these limitations and introduces the first fully KA-based deep model, demonstrating that KA-based layers can entirely replace traditional architectures in deep learning and achieve superior learning capacity. Specifically, (1) the proposed Share-activation KAN (SaKAN) reformulates Sprecher's variant of Kolmogorov-Arnold representation theorem, which achieves better optimization due to its simplified parameterization and denser training samples, to ease training difficulty, (2) this paper indicates that spline gradients contribute negligibly to training while consuming huge GPU memory, thus proposes the Grad-Free Spline to significantly reduce memory usage and computational overhead. (3) Building on these two innovations, our ALL U-KAN is the first representative implementation of fully KA-based deep model, where the proposed KA and KAonv layers completely replace FC and Conv layers. Extensive evaluations on three medical image segmentation tasks confirm the superiority of the full KA-based architecture compared to partial KA-based and traditional architectures, achieving all higher segmentation accuracy. Compared to directly deeply stacked KAN, ALL U-KAN achieves 10 times reduction in parameter count and reduces memory consumption by more than 20 times, unlocking the new explorations into deep KAN architectures.
FUGC: Benchmarking Semi-Supervised Learning Methods for Cervical Segmentation
Jan 22, 2026Abstract:Accurate segmentation of cervical structures in transvaginal ultrasound (TVS) is critical for assessing the risk of spontaneous preterm birth (PTB), yet the scarcity of labeled data limits the performance of supervised learning approaches. This paper introduces the Fetal Ultrasound Grand Challenge (FUGC), the first benchmark for semi-supervised learning in cervical segmentation, hosted at ISBI 2025. FUGC provides a dataset of 890 TVS images, including 500 training images, 90 validation images, and 300 test images. Methods were evaluated using the Dice Similarity Coefficient (DSC), Hausdorff Distance (HD), and runtime (RT), with a weighted combination of 0.4/0.4/0.2. The challenge attracted 10 teams with 82 participants submitting innovative solutions. The best-performing methods for each individual metric achieved 90.26\% mDSC, 38.88 mHD, and 32.85 ms RT, respectively. FUGC establishes a standardized benchmark for cervical segmentation, demonstrates the efficacy of semi-supervised methods with limited labeled data, and provides a foundation for AI-assisted clinical PTB risk assessment.
Revisiting Data Scaling Law for Medical Segmentation
Nov 17, 2025Abstract:The population loss of trained deep neural networks often exhibits power law scaling with the size of the training dataset, guiding significant performance advancements in deep learning applications. In this study, we focus on the scaling relationship with data size in the context of medical anatomical segmentation, a domain that remains underexplored. We analyze scaling laws for anatomical segmentation across 15 semantic tasks and 4 imaging modalities, demonstrating that larger datasets significantly improve segmentation performance, following similar scaling trends. Motivated by the topological isomorphism in images sharing anatomical structures, we evaluate the impact of deformation-guided augmentation strategies on data scaling laws, specifically random elastic deformation and registration-guided deformation. We also propose a novel, scalable image augmentation approach that generates diffeomorphic mappings from geodesic subspace based on image registration to introduce realistic deformation. Our experimental results demonstrate that both registered and generated deformation-based augmentation considerably enhance data utilization efficiency. The proposed generated deformation method notably achieves superior performance and accelerated convergence, surpassing standard power law scaling trends without requiring additional data. Overall, this work provides insights into the understanding of segmentation scalability and topological variation impact in medical imaging, thereby leading to more efficient model development with reduced annotation and computational costs.
Ambiguity-aware Truncated Flow Matching for Ambiguous Medical Image Segmentation
Nov 10, 2025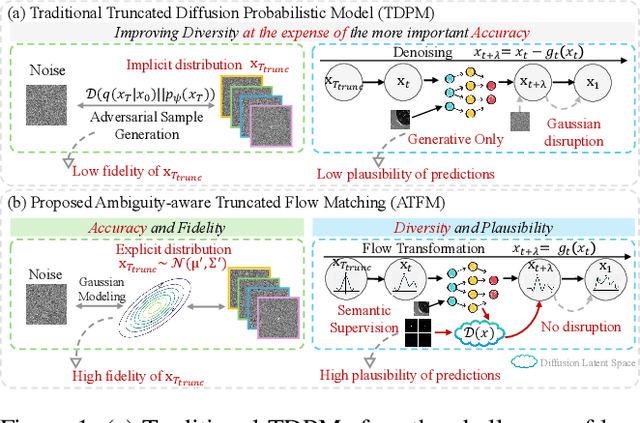
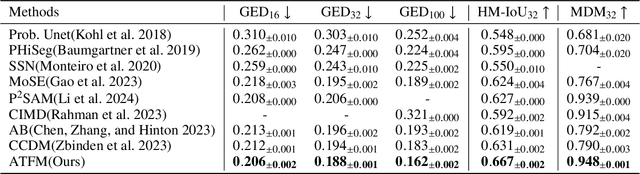
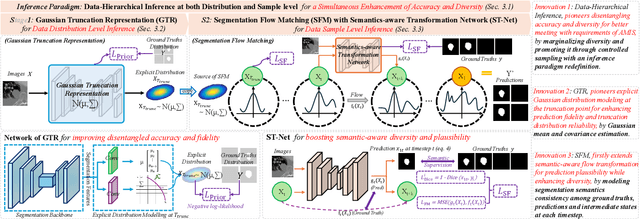
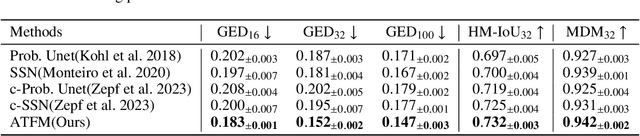
Abstract:A simultaneous enhancement of accuracy and diversity of predictions remains a challenge in ambiguous medical image segmentation (AMIS) due to the inherent trade-offs. While truncated diffusion probabilistic models (TDPMs) hold strong potential with a paradigm optimization, existing TDPMs suffer from entangled accuracy and diversity of predictions with insufficient fidelity and plausibility. To address the aforementioned challenges, we propose Ambiguity-aware Truncated Flow Matching (ATFM), which introduces a novel inference paradigm and dedicated model components. Firstly, we propose Data-Hierarchical Inference, a redefinition of AMIS-specific inference paradigm, which enhances accuracy and diversity at data-distribution and data-sample level, respectively, for an effective disentanglement. Secondly, Gaussian Truncation Representation (GTR) is introduced to enhance both fidelity of predictions and reliability of truncation distribution, by explicitly modeling it as a Gaussian distribution at $T_{\text{trunc}}$ instead of using sampling-based approximations.Thirdly, Segmentation Flow Matching (SFM) is proposed to enhance the plausibility of diverse predictions by extending semantic-aware flow transformation in Flow Matching (FM). Comprehensive evaluations on LIDC and ISIC3 datasets demonstrate that ATFM outperforms SOTA methods and simultaneously achieves a more efficient inference. ATFM improves GED and HM-IoU by up to $12\%$ and $7.3\%$ compared to advanced methods.
Structure and Smoothness Constrained Dual Networks for MR Bias Field Correction
Jul 02, 2025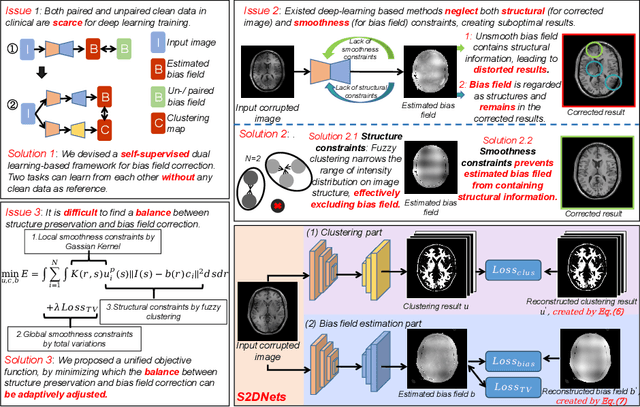
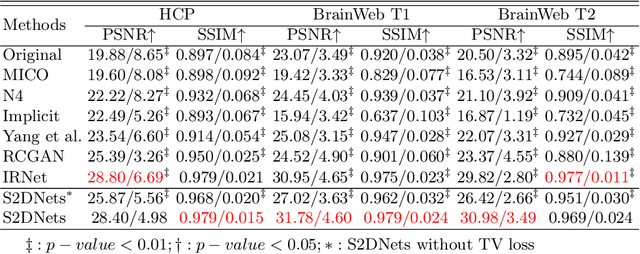
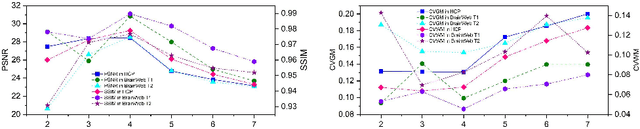
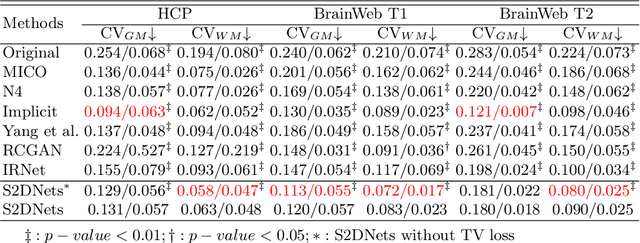
Abstract:MR imaging techniques are of great benefit to disease diagnosis. However, due to the limitation of MR devices, significant intensity inhomogeneity often exists in imaging results, which impedes both qualitative and quantitative medical analysis. Recently, several unsupervised deep learning-based models have been proposed for MR image improvement. However, these models merely concentrate on global appearance learning, and neglect constraints from image structures and smoothness of bias field, leading to distorted corrected results. In this paper, novel structure and smoothness constrained dual networks, named S2DNets, are proposed aiming to self-supervised bias field correction. S2DNets introduce piece-wise structural constraints and smoothness of bias field for network training to effectively remove non-uniform intensity and retain much more structural details. Extensive experiments executed on both clinical and simulated MR datasets show that the proposed model outperforms other conventional and deep learning-based models. In addition to comparison on visual metrics, downstream MR image segmentation tasks are also used to evaluate the impact of the proposed model. The source code is available at: https://github.com/LeongDong/S2DNets}{https://github.com/LeongDong/S2DNets.
* 11 pages, 3 figures, accepted by MICCAI
Finding Local Diffusion Schrödinger Bridge using Kolmogorov-Arnold Network
Feb 27, 2025Abstract:In image generation, Schr\"odinger Bridge (SB)-based methods theoretically enhance the efficiency and quality compared to the diffusion models by finding the least costly path between two distributions. However, they are computationally expensive and time-consuming when applied to complex image data. The reason is that they focus on fitting globally optimal paths in high-dimensional spaces, directly generating images as next step on the path using complex networks through self-supervised training, which typically results in a gap with the global optimum. Meanwhile, most diffusion models are in the same path subspace generated by weights $f_A(t)$ and $f_B(t)$, as they follow the paradigm ($x_t = f_A(t)x_{Img} + f_B(t)\epsilon$). To address the limitations of SB-based methods, this paper proposes for the first time to find local Diffusion Schr\"odinger Bridges (LDSB) in the diffusion path subspace, which strengthens the connection between the SB problem and diffusion models. Specifically, our method optimizes the diffusion paths using Kolmogorov-Arnold Network (KAN), which has the advantage of resistance to forgetting and continuous output. The experiment shows that our LDSB significantly improves the quality and efficiency of image generation using the same pre-trained denoising network and the KAN for optimising is only less than 0.1MB. The FID metric is reduced by \textbf{more than 15\%}, especially with a reduction of 48.50\% when NFE of DDIM is $5$ for the CelebA dataset. Code is available at https://github.com/Qiu-XY/LDSB.
Tumor Detection, Segmentation and Classification Challenge on Automated 3D Breast Ultrasound: The TDSC-ABUS Challenge
Jan 26, 2025



Abstract:Breast cancer is one of the most common causes of death among women worldwide. Early detection helps in reducing the number of deaths. Automated 3D Breast Ultrasound (ABUS) is a newer approach for breast screening, which has many advantages over handheld mammography such as safety, speed, and higher detection rate of breast cancer. Tumor detection, segmentation, and classification are key components in the analysis of medical images, especially challenging in the context of 3D ABUS due to the significant variability in tumor size and shape, unclear tumor boundaries, and a low signal-to-noise ratio. The lack of publicly accessible, well-labeled ABUS datasets further hinders the advancement of systems for breast tumor analysis. Addressing this gap, we have organized the inaugural Tumor Detection, Segmentation, and Classification Challenge on Automated 3D Breast Ultrasound 2023 (TDSC-ABUS2023). This initiative aims to spearhead research in this field and create a definitive benchmark for tasks associated with 3D ABUS image analysis. In this paper, we summarize the top-performing algorithms from the challenge and provide critical analysis for ABUS image examination. We offer the TDSC-ABUS challenge as an open-access platform at https://tdsc-abus2023.grand-challenge.org/ to benchmark and inspire future developments in algorithmic research.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge