Yiyu Shi
When Small Variations Become Big Failures: Reliability Challenges in Compute-in-Memory Neural Accelerators
Mar 03, 2026Abstract:Compute-in-memory (CiM) architectures promise significant improvements in energy efficiency and throughput for deep neural network acceleration by alleviating the von Neumann bottleneck. However, their reliance on emerging non-volatile memory devices introduces device-level non-idealities-such as write variability, conductance drift, and stochastic noise-that fundamentally challenge reliability, predictability, and safety, especially in safety-critical applications. This talk examines the reliability limits of CiM-based neural accelerators and presents a series of techniques that bridge device physics, architecture, and learning algorithms to address these challenges. We first demonstrate that even small device variations can lead to disproportionately large accuracy degradation and catastrophic failures in safety-critical inference workloads, revealing a critical gap between average-case evaluations and worst-case behavior. Building on this insight, we introduce SWIM, a selective write-verify mechanism that strategically applies verification only where it is most impactful, significantly improving reliability while maintaining CiM's efficiency advantages. Finally, we explore a learning-centric solution that improves realistic worst-case performance by training neural networks with right-censored Gaussian noise, aligning training assumptions with hardware-induced variability and enabling robust deployment without excessive hardware overhead. Together, these works highlight the necessity of cross-layer co-design for CiM accelerators and provide a principled path toward dependable, efficient neural inference on emerging memory technologies-paving the way for their adoption in safety- and reliability-critical systems.
ADRD-Bench: A Preliminary LLM Benchmark for Alzheimer's Disease and Related Dementias
Feb 12, 2026Abstract:Large language models (LLMs) have shown great potential for healthcare applications. However, existing evaluation benchmarks provide minimal coverage of Alzheimer's Disease and Related Dementias (ADRD). To address this gap, we introduce ADRD-Bench, the first ADRD-specific benchmark dataset designed for rigorous evaluation of LLMs. ADRD-Bench has two components: 1) ADRD Unified QA, a synthesis of 1,352 questions consolidated from seven established medical benchmarks, providing a unified assessment of clinical knowledge; and 2) ADRD Caregiving QA, a novel set of 149 questions derived from the Aging Brain Care (ABC) program, a widely used, evidence-based brain health management program. Guided by a program with national expertise in comprehensive ADRD care, this new set was designed to mitigate the lack of practical caregiving context in existing benchmarks. We evaluated 33 state-of-the-art LLMs on the proposed ADRD-Bench. Results showed that the accuracy of open-weight general models ranged from 0.63 to 0.93 (mean: 0.78; std: 0.09). The accuracy of open-weight medical models ranged from 0.48 to 0.93 (mean: 0.82; std: 0.13). The accuracy of closed-source general models ranged from 0.83 to 0.91 (mean: 0.89; std: 0.03). While top-tier models achieved high accuracies (>0.9), case studies revealed that inconsistent reasoning quality and stability limit their reliability, highlighting a critical need for domain-specific improvement to enhance LLMs' knowledge and reasoning grounded in daily caregiving data. The entire dataset is available at https://github.com/IIRL-ND/ADRD-Bench.
Patient-Conditioned Adaptive Offsets for Reliable Diagnosis across Subgroups
Jan 19, 2026Abstract:AI models for medical diagnosis often exhibit uneven performance across patient populations due to heterogeneity in disease prevalence, imaging appearance, and clinical risk profiles. Existing algorithmic fairness approaches typically seek to reduce such disparities by suppressing sensitive attributes. However, in medical settings these attributes often carry essential diagnostic information, and removing them can degrade accuracy and reliability, particularly in high-stakes applications. In contrast, clinical decision making explicitly incorporates patient context when interpreting diagnostic evidence, suggesting a different design direction for subgroup-aware models. In this paper, we introduce HyperAdapt, a patient-conditioned adaptation framework that improves subgroup reliability while maintaining a shared diagnostic model. Clinically relevant attributes such as age and sex are encoded into a compact embedding and used to condition a hypernetwork-style module, which generates small residual modulation parameters for selected layers of a shared backbone. This design preserves the general medical knowledge learned by the backbone while enabling targeted adjustments that reflect patient-specific variability. To ensure efficiency and robustness, adaptations are constrained through low-rank and bottlenecked parameterizations, limiting both model complexity and computational overhead. Experiments across multiple public medical imaging benchmarks demonstrate that the proposed approach consistently improves subgroup-level performance without sacrificing overall accuracy. On the PAD-UFES-20 dataset, our method outperforms the strongest competing baseline by 4.1% in recall and 4.4% in F1 score, with larger gains observed for underrepresented patient populations.
H-CNN-ViT: A Hierarchical Gated Attention Multi-Branch Model for Bladder Cancer Recurrence Prediction
Nov 19, 2025Abstract:Bladder cancer is one of the most prevalent malignancies worldwide, with a recurrence rate of up to 78%, necessitating accurate post-operative monitoring for effective patient management. Multi-sequence contrast-enhanced MRI is commonly used for recurrence detection; however, interpreting these scans remains challenging, even for experienced radiologists, due to post-surgical alterations such as scarring, swelling, and tissue remodeling. AI-assisted diagnostic tools have shown promise in improving bladder cancer recurrence prediction, yet progress in this field is hindered by the lack of dedicated multi-sequence MRI datasets for recurrence assessment study. In this work, we first introduce a curated multi-sequence, multi-modal MRI dataset specifically designed for bladder cancer recurrence prediction, establishing a valuable benchmark for future research. We then propose H-CNN-ViT, a new Hierarchical Gated Attention Multi-Branch model that enables selective weighting of features from the global (ViT) and local (CNN) paths based on contextual demands, achieving a balanced and targeted feature fusion. Our multi-branch architecture processes each modality independently, ensuring that the unique properties of each imaging channel are optimally captured and integrated. Evaluated on our dataset, H-CNN-ViT achieves an AUC of 78.6%, surpassing state-of-the-art models. Our model is publicly available at https://github.com/XLIAaron/H-CNN-ViT.
AT-CXR: Uncertainty-Aware Agentic Triage for Chest X-rays
Aug 26, 2025Abstract:Agentic AI is advancing rapidly, yet truly autonomous medical-imaging triage, where a system decides when to stop, escalate, or defer under real constraints, remains relatively underexplored. To address this gap, we introduce AT-CXR, an uncertainty-aware agent for chest X-rays. The system estimates per-case confidence and distributional fit, then follows a stepwise policy to issue an automated decision or abstain with a suggested label for human intervention. We evaluate two router designs that share the same inputs and actions: a deterministic rule-based router and an LLM-decided router. Across five-fold evaluation on a balanced subset of NIH ChestX-ray14 dataset, both variants outperform strong zero-shot vision-language models and state-of-the-art supervised classifiers, achieving higher full-coverage accuracy and superior selective-prediction performance, evidenced by a lower area under the risk-coverage curve (AURC) and a lower error rate at high coverage, while operating with lower latency that meets practical clinical constraints. The two routers provide complementary operating points, enabling deployments to prioritize maximal throughput or maximal accuracy. Our code is available at https://github.com/XLIAaron/uncertainty-aware-cxr-agent.
HybridQ: Hybrid Classical-Quantum Generative Adversarial Network for Skin Disease Image Generation
Jun 26, 2025Abstract:Machine learning-assisted diagnosis is gaining traction in skin disease detection, but training effective models requires large amounts of high-quality data. Skin disease datasets often suffer from class imbalance, privacy concerns, and object bias, making data augmentation essential. While classical generative models are widely used, they demand extensive computational resources and lengthy training time. Quantum computing offers a promising alternative, but existing quantum-based image generation methods can only yield grayscale low-quality images. Through a novel classical-quantum latent space fusion technique, our work overcomes this limitation and introduces the first classical-quantum generative adversarial network (GAN) capable of generating color medical images. Our model outperforms classical deep convolutional GANs and existing hybrid classical-quantum GANs in both image generation quality and classification performance boost when used as data augmentation. Moreover, the performance boost is comparable with that achieved using state-of-the-art classical generative models, yet with over 25 times fewer parameters and 10 times fewer training epochs. Such results suggest a promising future for quantum image generation as quantum hardware advances. Finally, we demonstrate the robust performance of our model on real IBM quantum machine with hardware noise.
Unsupervised Out-of-Distribution Detection in Medical Imaging Using Multi-Exit Class Activation Maps and Feature Masking
May 13, 2025Abstract:Out-of-distribution (OOD) detection is essential for ensuring the reliability of deep learning models in medical imaging applications. This work is motivated by the observation that class activation maps (CAMs) for in-distribution (ID) data typically emphasize regions that are highly relevant to the model's predictions, whereas OOD data often lacks such focused activations. By masking input images with inverted CAMs, the feature representations of ID data undergo more substantial changes compared to those of OOD data, offering a robust criterion for differentiation. In this paper, we introduce a novel unsupervised OOD detection framework, Multi-Exit Class Activation Map (MECAM), which leverages multi-exit CAMs and feature masking. By utilizing mult-exit networks that combine CAMs from varying resolutions and depths, our method captures both global and local feature representations, thereby enhancing the robustness of OOD detection. We evaluate MECAM on multiple ID datasets, including ISIC19 and PathMNIST, and test its performance against three medical OOD datasets, RSNA Pneumonia, COVID-19, and HeadCT, and one natural image OOD dataset, iSUN. Comprehensive comparisons with state-of-the-art OOD detection methods validate the effectiveness of our approach. Our findings emphasize the potential of multi-exit networks and feature masking for advancing unsupervised OOD detection in medical imaging, paving the way for more reliable and interpretable models in clinical practice.
The Cost of Local and Global Fairness in Federated Learning
Mar 27, 2025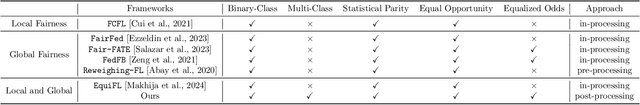

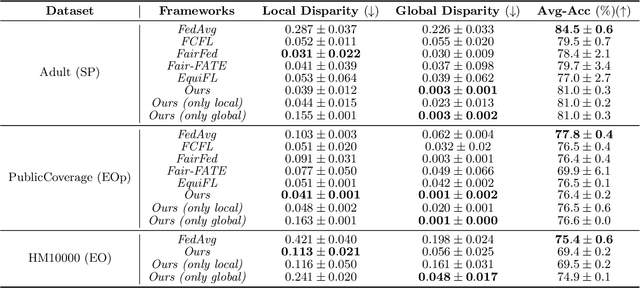
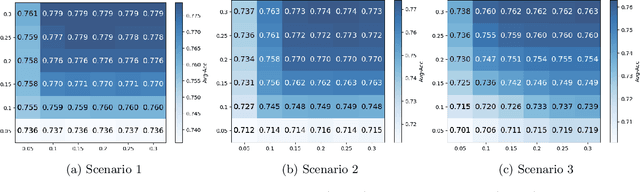
Abstract:With the emerging application of Federated Learning (FL) in finance, hiring and healthcare, FL models are regulated to be fair, preventing disparities with respect to legally protected attributes such as race or gender. Two concepts of fairness are important in FL: global and local fairness. Global fairness addresses the disparity across the entire population and local fairness is concerned with the disparity within each client. Prior fair FL frameworks have improved either global or local fairness without considering both. Furthermore, while the majority of studies on fair FL focuses on binary settings, many real-world applications are multi-class problems. This paper proposes a framework that investigates the minimum accuracy lost for enforcing a specified level of global and local fairness in multi-class FL settings. Our framework leads to a simple post-processing algorithm that derives fair outcome predictors from the Bayesian optimal score functions. Experimental results show that our algorithm outperforms the current state of the art (SOTA) with regard to the accuracy-fairness tradoffs, computational and communication costs. Codes are available at: https://github.com/papersubmission678/The-cost-of-local-and-global-fairness-in-FL .
Combating Partial Perception Deficit in Autonomous Driving with Multimodal LLM Commonsense
Mar 10, 2025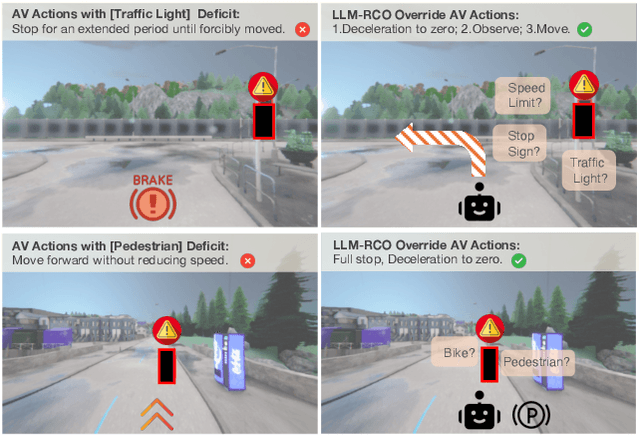
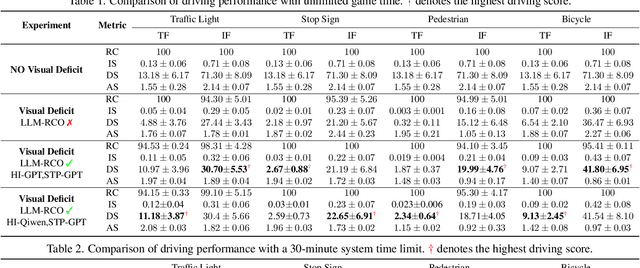
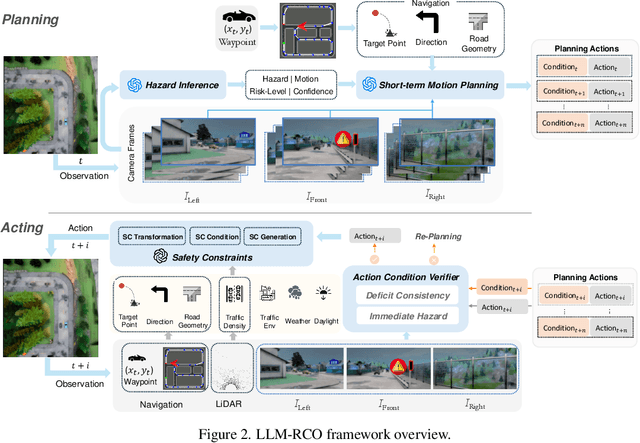

Abstract:Partial perception deficits can compromise autonomous vehicle safety by disrupting environmental understanding. Current protocols typically respond with immediate stops or minimal-risk maneuvers, worsening traffic flow and lacking flexibility for rare driving scenarios. In this paper, we propose LLM-RCO, a framework leveraging large language models to integrate human-like driving commonsense into autonomous systems facing perception deficits. LLM-RCO features four key modules: hazard inference, short-term motion planner, action condition verifier, and safety constraint generator. These modules interact with the dynamic driving environment, enabling proactive and context-aware control actions to override the original control policy of autonomous agents. To improve safety in such challenging conditions, we construct DriveLM-Deficit, a dataset of 53,895 video clips featuring deficits of safety-critical objects, complete with annotations for LLM-based hazard inference and motion planning fine-tuning. Extensive experiments in adverse driving conditions with the CARLA simulator demonstrate that systems equipped with LLM-RCO significantly improve driving performance, highlighting its potential for enhancing autonomous driving resilience against adverse perception deficits. Our results also show that LLMs fine-tuned with DriveLM-Deficit can enable more proactive movements instead of conservative stops in the context of perception deficits.
MPBD-LSTM: A Predictive Model for Colorectal Liver Metastases Using Time Series Multi-phase Contrast-Enhanced CT Scans
Dec 02, 2024Abstract:Colorectal cancer is a prevalent form of cancer, and many patients develop colorectal cancer liver metastasis (CRLM) as a result. Early detection of CRLM is critical for improving survival rates. Radiologists usually rely on a series of multi-phase contrast-enhanced computed tomography (CECT) scans done during follow-up visits to perform early detection of the potential CRLM. These scans form unique five-dimensional data (time, phase, and axial, sagittal, and coronal planes in 3D CT). Most of the existing deep learning models can readily handle four-dimensional data (e.g., time-series 3D CT images) and it is not clear how well they can be extended to handle the additional dimension of phase. In this paper, we build a dataset of time-series CECT scans to aid in the early diagnosis of CRLM, and build upon state-of-the-art deep learning techniques to evaluate how to best predict CRLM. Our experimental results show that a multi-plane architecture based on 3D bi-directional LSTM, which we call MPBD-LSTM, works best, achieving an area under curve (AUC) of 0.79. On the other hand, analysis of the results shows that there is still great room for further improvement.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge