Marinka Zitnik
Department of Biomedical Informatics, Harvard Medical School, Boston, MA, USA, Kempner Institute for the Study of Natural and Artificial Intelligence, Allston, MA, USA, Broad Institute of MIT and Harvard, Cambridge, MA, USA, Harvard Data Science Initiative, Cambridge, MA, USA
Evaluating Relational Reasoning in LLMs with REL
Apr 14, 2026Abstract:Relational reasoning is the ability to infer relations that jointly bind multiple entities, attributes, or variables. This ability is central to scientific reasoning, but existing evaluations of relational reasoning in large language models often focus on structured inputs such as tables, graphs, or synthetic tasks, and do not isolate the difficulty introduced by higher-arity relational binding. We study this problem through the lens of Relational Complexity (RC), which we define as the minimum number of independent entities or operands that must be simultaneously bound to apply a relation. RC provides a principled way to vary reasoning difficulty while controlling for confounders such as input size, vocabulary, and representational choices. Building on RC, we introduce REL, a generative benchmark framework spanning algebra, chemistry, and biology that varies RC within each domain. Across frontier LLMs, performance degrades consistently and monotonically as RC increases, even when the total number of entities is held fixed. This failure mode persists with increased test-time compute and in-context learning, suggesting a limitation tied to the arity of the required relational binding rather than to insufficient inference steps or lack of exposure to examples. Our results identify a regime of higher-arity reasoning in which current models struggle, and motivate re-examining benchmarks through the lens of relational complexity.
Distribution-Conditioned Transport
Mar 05, 2026Abstract:Learning a transport model that maps a source distribution to a target distribution is a canonical problem in machine learning, but scientific applications increasingly require models that can generalize to source and target distributions unseen during training. We introduce distribution-conditioned transport (DCT), a framework that conditions transport maps on learned embeddings of source and target distributions, enabling generalization to unseen distribution pairs. DCT also allows semi-supervised learning for distributional forecasting problems: because it learns from arbitrary distribution pairs, it can leverage distributions observed at only one condition to improve transport prediction. DCT is agnostic to the underlying transport mechanism, supporting models ranging from flow matching to distributional divergence-based models (e.g. Wasserstein, MMD). We demonstrate the practical performance benefits of DCT on synthetic benchmarks and four applications in biology: batch effect transfer in single-cell genomics, perturbation prediction from mass cytometry data, learning clonal transcriptional dynamics in hematopoiesis, and modeling T-cell receptor sequence evolution.
When Sensors Fail: Temporal Sequence Models for Robust PPO under Sensor Drift
Mar 04, 2026Abstract:Real-world reinforcement learning systems must operate under distributional drift in their observation streams, yet most policy architectures implicitly assume fully observed and noise-free states. We study robustness of Proximal Policy Optimization (PPO) under temporally persistent sensor failures that induce partial observability and representation shift. To respond to this drift, we augment PPO with temporal sequence models, including Transformers and State Space Models (SSMs), to enable policies to infer missing information from history and maintain performance. Under a stochastic sensor failure process, we prove a high-probability bound on infinite-horizon reward degradation that quantifies how robustness depends on policy smoothness and failure persistence. Empirically, on MuJoCo continuous-control benchmarks with severe sensor dropout, we show Transformer-based sequence policies substantially outperform MLP, RNN, and SSM baselines in robustness, maintaining high returns even when large fractions of sensors are unavailable. These results demonstrate that temporal sequence reasoning provides a principled and practical mechanism for reliable operation under observation drift caused by sensor unreliability.
STRAND: Sequence-Conditioned Transport for Single-Cell Perturbations
Feb 10, 2026Abstract:Predicting how genetic perturbations change cellular state is a core problem for building controllable models of gene regulation. Perturbations targeting the same gene can produce different transcriptional responses depending on their genomic locus, including different transcription start sites and regulatory elements. Gene-level perturbation models collapse these distinct interventions into the same representation. We introduce STRAND, a generative model that predicts single-cell transcriptional responses by conditioning on regulatory DNA sequence. STRAND represents a perturbation by encoding the sequence at its genomic locus and uses this representation to parameterize a conditional transport process from control to perturbed cell states. Representing perturbations by sequence, rather than by a fixed set of gene identifiers, supports zero-shot inference at loci not seen during training and expands inference-time genomic coverage from ~1.5% for gene-level single-cell foundation models to ~95% of the genome. We evaluate STRAND on CRISPR perturbation datasets in K562, Jurkat, and RPE1 cells. STRAND improves discrimination scores by up to 33% in low-sample regimes, achieves the best average rank on unseen gene perturbation benchmarks, and improves transfer to novel cell lines by up to 0.14 in Pearson correlation. Ablations isolate the gains to sequence conditioning and transport, and case studies show that STRAND resolves functionally alternative transcription start sites missed by gene-level models.
Autonomous Knowledge Graph Exploration with Adaptive Breadth-Depth Retrieval
Jan 20, 2026Abstract:Retrieving evidence for language model queries from knowledge graphs requires balancing broad search across the graph with multi-hop traversal to follow relational links. Similarity-based retrievers provide coverage but remain shallow, whereas traversal-based methods rely on selecting seed nodes to start exploration, which can fail when queries span multiple entities and relations. We introduce ARK: Adaptive Retriever of Knowledge, an agentic KG retriever that gives a language model control over this breadth-depth tradeoff using a two-operation toolset: global lexical search over node descriptors and one-hop neighborhood exploration that composes into multi-hop traversal. ARK alternates between breadth-oriented discovery and depth-oriented expansion without depending on a fragile seed selection, a pre-set hop depth, or requiring retrieval training. ARK adapts tool use to queries, using global search for language-heavy queries and neighborhood exploration for relation-heavy queries. On STaRK, ARK reaches 59.1% average Hit@1 and 67.4 average MRR, improving average Hit@1 by up to 31.4% and average MRR by up to 28.0% over retrieval-based and agentic training-free methods. Finally, we distill ARK's tool-use trajectories from a large teacher into an 8B model via label-free imitation, improving Hit@1 by +7.0, +26.6, and +13.5 absolute points over the base 8B model on AMAZON, MAG, and PRIME datasets, respectively, while retaining up to 98.5% of the teacher's Hit@1 rate.
Beyond Affinity: A Benchmark of 1D, 2D, and 3D Methods Reveals Critical Trade-offs in Structure-Based Drug Design
Jan 13, 2026Abstract:Currently, the field of structure-based drug design is dominated by three main types of algorithms: search-based algorithms, deep generative models, and reinforcement learning. While existing works have typically focused on comparing models within a single algorithmic category, cross-algorithm comparisons remain scarce. In this paper, to fill the gap, we establish a benchmark to evaluate the performance of fifteen models across these different algorithmic foundations by assessing the pharmaceutical properties of the generated molecules and their docking affinities and poses with specified target proteins. We highlight the unique advantages of each algorithmic approach and offer recommendations for the design of future SBDD models. We emphasize that 1D/2D ligand-centric drug design methods can be used in SBDD by treating the docking function as a black-box oracle, which is typically neglected. Our evaluation reveals distinct patterns across model categories. 3D structure-based models excel in binding affinities but show inconsistencies in chemical validity and pose quality. 1D models demonstrate reliable performance in standard molecular metrics but rarely achieve optimal binding affinities. 2D models offer balanced performance, maintaining high chemical validity while achieving moderate binding scores. Through detailed analysis across multiple protein targets, we identify key improvement areas for each model category, providing insights for researchers to combine strengths of different approaches while addressing their limitations. All the code that are used for benchmarking is available in https://github.com/zkysfls/2025-sbdd-benchmark
Graph AI generates neurological hypotheses validated in molecular, organoid, and clinical systems
Dec 13, 2025Abstract:Neurological diseases are the leading global cause of disability, yet most lack disease-modifying treatments. We present PROTON, a heterogeneous graph transformer that generates testable hypotheses across molecular, organoid, and clinical systems. To evaluate PROTON, we apply it to Parkinson's disease (PD), bipolar disorder (BD), and Alzheimer's disease (AD). In PD, PROTON linked genetic risk loci to genes essential for dopaminergic neuron survival and predicted pesticides toxic to patient-derived neurons, including the insecticide endosulfan, which ranked within the top 1.29% of predictions. In silico screens performed by PROTON reproduced six genome-wide $α$-synuclein experiments, including a split-ubiquitin yeast two-hybrid system (normalized enrichment score [NES] = 2.30, FDR-adjusted $p < 1 \times 10^{-4}$), an ascorbate peroxidase proximity labeling assay (NES = 2.16, FDR $< 1 \times 10^{-4}$), and a high-depth targeted exome sequencing study in 496 synucleinopathy patients (NES = 2.13, FDR $< 1 \times 10^{-4}$). In BD, PROTON predicted calcitriol as a candidate drug that reversed proteomic alterations observed in cortical organoids derived from BD patients. In AD, we evaluated PROTON predictions in health records from $n = 610,524$ patients at Mass General Brigham, confirming that five PROTON-predicted drugs were associated with reduced seven-year dementia risk (minimum hazard ratio = 0.63, 95% CI: 0.53-0.75, $p < 1 \times 10^{-7}$). PROTON generated neurological hypotheses that were evaluated across molecular, organoid, and clinical systems, defining a path for AI-driven discovery in neurological disease.
Protein Structure Tokenization via Geometric Byte Pair Encoding
Nov 13, 2025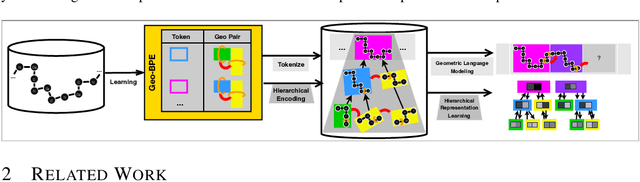
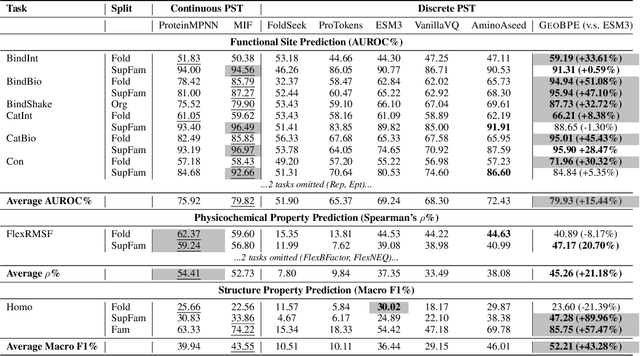
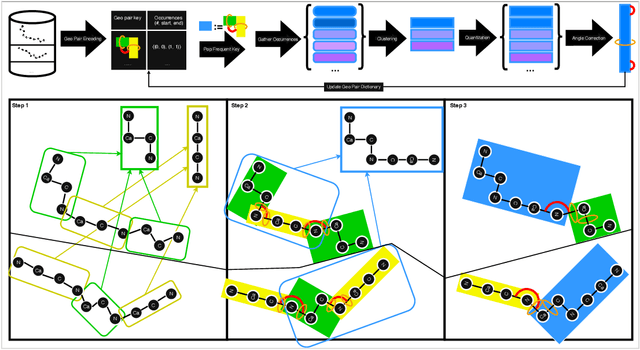
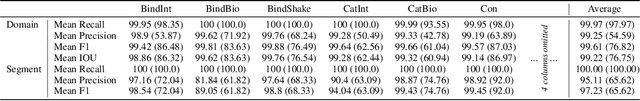
Abstract:Protein structure is central to biological function, and enabling multimodal protein models requires joint reasoning over sequence, structure, and function. A key barrier is the lack of principled protein structure tokenizers (PSTs): existing approaches fix token size or rely on continuous vector codebooks, limiting interpretability, multi-scale control, and transfer across architectures. We introduce GeoBPE, a geometry-grounded PST that transforms continuous, noisy, multi-scale backbone conformations into discrete ``sentences'' of geometry while enforcing global constraints. Analogous to byte-pair encoding, GeoBPE generates a hierarchical vocabulary of geometric primitives by iteratively (i) clustering Geo-Pair occurrences with k-medoids to yield a resolution-controllable vocabulary; (ii) quantizing each Geo-Pair to its closest medoid prototype; and (iii) reducing drift through differentiable inverse kinematics that optimizes boundary glue angles under an $\mathrm{SE}(3)$ end-frame loss. GeoBPE offers compression ($>$10x reduction in bits-per-residue at similar distortion rate), data efficiency ($>$10x less training data), and generalization (maintains test/train distortion ratio of $1.0-1.1$). It is architecture-agnostic: (a) its hierarchical vocabulary provides a strong inductive bias for coarsening residue-level embeddings from large PLMs into motif- and protein-level representations, consistently outperforming leading PSTs across $12$ tasks and $24$ test splits; (b) paired with a transformer, GeoBPE supports unconditional backbone generation via language modeling; and (c) tokens align with CATH functional families and support expert-interpretable case studies, offering functional meaning absent in prior PSTs. Code is available at https://github.com/shiningsunnyday/PT-BPE/.
One Patient, Many Contexts: Scaling Medical AI Through Contextual Intelligence
Jun 11, 2025

Abstract:Medical foundation models, including language models trained on clinical notes, vision-language models on medical images, and multimodal models on electronic health records, can summarize clinical notes, answer medical questions, and assist in decision-making. Adapting these models to new populations, specialties, or settings typically requires fine-tuning, careful prompting, or retrieval from knowledge bases. This can be impractical, and limits their ability to interpret unfamiliar inputs and adjust to clinical situations not represented during training. As a result, models are prone to contextual errors, where predictions appear reasonable but fail to account for critical patient-specific or contextual information. These errors stem from a fundamental limitation that current models struggle with: dynamically adjusting their behavior across evolving contexts of medical care. In this Perspective, we outline a vision for context-switching in medical AI: models that dynamically adapt their reasoning without retraining to new specialties, populations, workflows, and clinical roles. We envision context-switching AI to diagnose, manage, and treat a wide range of diseases across specialties and regions, and expand access to medical care.
Prompting Decision Transformers for Zero-Shot Reach-Avoid Policies
May 25, 2025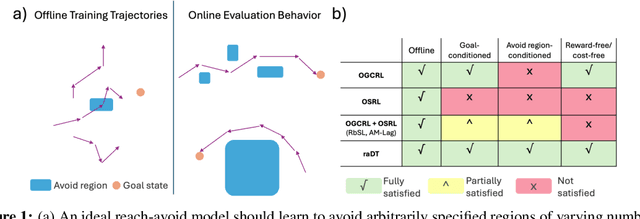
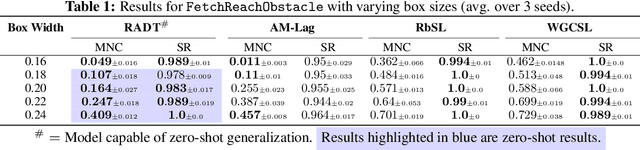
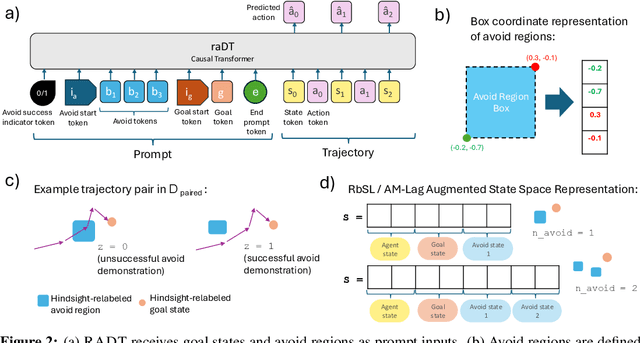
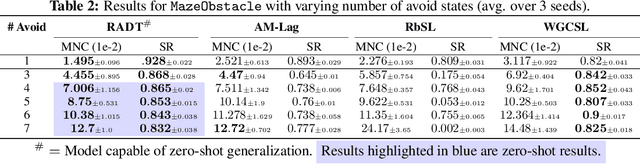
Abstract:Offline goal-conditioned reinforcement learning methods have shown promise for reach-avoid tasks, where an agent must reach a target state while avoiding undesirable regions of the state space. Existing approaches typically encode avoid-region information into an augmented state space and cost function, which prevents flexible, dynamic specification of novel avoid-region information at evaluation time. They also rely heavily on well-designed reward and cost functions, limiting scalability to complex or poorly structured environments. We introduce RADT, a decision transformer model for offline, reward-free, goal-conditioned, avoid region-conditioned RL. RADT encodes goals and avoid regions directly as prompt tokens, allowing any number of avoid regions of arbitrary size to be specified at evaluation time. Using only suboptimal offline trajectories from a random policy, RADT learns reach-avoid behavior through a novel combination of goal and avoid-region hindsight relabeling. We benchmark RADT against 3 existing offline goal-conditioned RL models across 11 tasks, environments, and experimental settings. RADT generalizes in a zero-shot manner to out-of-distribution avoid region sizes and counts, outperforming baselines that require retraining. In one such zero-shot setting, RADT achieves 35.7% improvement in normalized cost over the best retrained baseline while maintaining high goal-reaching success. We apply RADT to cell reprogramming in biology, where it reduces visits to undesirable intermediate gene expression states during trajectories to desired target states, despite stochastic transitions and discrete, structured state dynamics.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge