Jiazhen Pan
Evo-MedAgent: Beyond One-Shot Diagnosis with Agents That Remember, Reflect, and Improve
Apr 15, 2026Abstract:Tool-augmented large language model (LLM) agents can orchestrate specialist classifiers, segmentation models, and visual question-answering modules to interpret chest X-rays. However, these agents still solve each case in isolation: they fail to accumulate experience across cases, correct recurrent reasoning mistakes, or adapt their tool-use behavior without expensive reinforcement learning. While a radiologist naturally improves with every case, current agents remain static. In this work, we propose Evo-MedAgent, a self-evolving memory module that equips a medical agent with the capacity for inter-case learning at test time. Our memory comprises three complementary stores: (1)~\emph{Retrospective Clinical Episodes} that retrieve problem-solving experiences from similar past cases, (2)~an \emph{Adaptive Procedural Heuristics} bank curating priority-tagged diagnostic rules that evolves via reflection, much like a physician refining their internal criteria, and (3)~a \emph{Tool Reliability Controller} that tracks per-tool trustworthiness. On ChestAgentBench, Evo-MedAgent raises multiple-choice question (MCQ) accuracy from 0.68 to 0.79 on GPT-5-mini, and from 0.76 to 0.87 on Gemini-3 Flash. With a strong base model, evolving memory improves performance more effectively than orchestrating external tools on qualitative diagnostic tasks. Because Evo-MedAgent requires no training, its per-case overhead is bounded by one additional retrieval pass and a single reflection call, making it deployable on top of any frozen model.
MedOpenClaw: Auditable Medical Imaging Agents Reasoning over Uncurated Full Studies
Mar 25, 2026Abstract:Currently, evaluating vision-language models (VLMs) in medical imaging tasks oversimplifies clinical reality by relying on pre-selected 2D images that demand significant manual labor to curate. This setup misses the core challenge of realworld diagnostics: a true clinical agent must actively navigate full 3D volumes across multiple sequences or modalities to gather evidence and ultimately support a final decision. To address this, we propose MEDOPENCLAW, an auditable runtime designed to let VLMs operate dynamically within standard medical tools or viewers (e.g., 3D Slicer). On top of this runtime, we introduce MEDFLOWBENCH, a full-study medical imaging benchmark covering multi-sequence brain MRI and lung CT/PET. It systematically evaluates medical agentic capabilities across viewer-only, tool-use, and open-method tracks. Initial results reveal a critical insight: while state-of-the-art LLMs/VLMs (e.g., Gemini 3.1 Pro and GPT-5.4) can successfully navigate the viewer to solve basic study-level tasks, their performance paradoxically degrades when given access to professional support tools due to a lack of precise spatial grounding. By bridging the gap between static-image perception and interactive clinical workflows, MEDOPENCLAW and MEDFLOWBENCH establish a reproducible foundation for developing auditable, full-study medical imaging agents.
Opportunistic Cardiac Health Assessment: Estimating Phenotypes from Localizer MRI through Multi-Modal Representations
Mar 13, 2026Abstract:Cardiovascular diseases are the leading cause of death. Cardiac phenotypes (CPs), e.g., ejection fraction, are the gold standard for assessing cardiac health, but they are derived from cine cardiac magnetic resonance imaging (CMR), which is costly and requires high spatio-temporal resolution. Every magnetic resonance (MR) examination begins with rapid and coarse localizers for scan planning, which are discarded thereafter. Despite non-diagnostic image quality and lack of temporal information, localizers can provide valuable structural information rapidly. In addition to imaging, patient-level information, including demographics and lifestyle, influence the cardiac health assessment. Electrocardiograms (ECGs) are inexpensive, routinely ordered in clinical practice, and capture the temporal activity of the heart. Here, we introduce C-TRIP (Cardiac Tri-modal Representations for Imaging Phenotypes), a multi-modal framework that aligns localizer MRI, ECG signals, and tabular metadata to learn a robust latent space and predict CPs using localizer images as an opportunistic alternative to CMR. By combining these three modalities, we leverage cheap spatial and temporal information from localizers, and ECG, respectively while benefiting from patient-specific context provided by tabular data. Our pipeline consists of three stages. First, encoders are trained independently to learn uni-modal representations. The second stage fuses the pre-trained encoders to unify the latent space. The final stage uses the enriched representation space for CP prediction, with inference performed exclusively on localizer MRI. Proposed C-TRIP yields accurate functional CPs, and high correlations for structural CPs. Since localizers are inherently rapid and low-cost, our C-TRIP framework could enable better accessibility for CP estimation.
No Image, No Problem: End-to-End Multi-Task Cardiac Analysis from Undersampled k-Space
Mar 10, 2026Abstract:Conventional clinical CMR pipelines rely on a sequential "reconstruct-then-analyze" paradigm, forcing an ill-posed intermediate step that introduces avoidable artifacts and information bottlenecks. This creates a fundamental mathematical paradox: it attempts to recover high-dimensional pixel arrays (i.e., images) from undersampled k-space, rather than directly extracting the low-dimensional physiological labels actually required for diagnosis. To unlock the direct diagnostic potential of k-space, we propose k-MTR (k-space Multi-Task Representation), a k-space representation learning framework that aligns undersampled k-space data and fully-sampled images into a shared semantic manifold. Leveraging a large-scale controlled simulation of 42,000 subjects, k-MTR forces the k-space encoder to restore anatomical information lost to undersampling directly within the latent space, bypassing the explicit inverse problem for downstream analysis. We demonstrate that this latent alignment enables the dense latent space embedded with high-level physiological semantics directly from undersampled frequencies. Across continuous phenotype regression, disease classification, and anatomical segmentation, k-MTR achieves highly competitive performance against state-of-the-art image-domain baselines. By showcasing that precise spatial geometries and multi-task features can be successfully recovered directly from the k-space representations, k-MTR provides a robust architectural blueprint for task-aware cardiac MRI workflows.
Reconstruction-free segmentation from undersampled k-space using transformers
Nov 05, 2025Abstract:Motivation: High acceleration factors place a limit on MRI image reconstruction. This limit is extended to segmentation models when treating these as subsequent independent processes. Goal: Our goal is to produce segmentations directly from sparse k-space measurements without the need for intermediate image reconstruction. Approach: We employ a transformer architecture to encode global k-space information into latent features. The produced latent vectors condition queried coordinates during decoding to generate segmentation class probabilities. Results: The model is able to produce better segmentations across high acceleration factors than image-based segmentation baselines. Impact: Cardiac segmentation directly from undersampled k-space samples circumvents the need for an intermediate image reconstruction step. This allows the potential to assess myocardial structure and function on higher acceleration factors than methods that rely on images as input.
AortaDiff: A Unified Multitask Diffusion Framework For Contrast-Free AAA Imaging
Oct 01, 2025Abstract:While contrast-enhanced CT (CECT) is standard for assessing abdominal aortic aneurysms (AAA), the required iodinated contrast agents pose significant risks, including nephrotoxicity, patient allergies, and environmental harm. To reduce contrast agent use, recent deep learning methods have focused on generating synthetic CECT from non-contrast CT (NCCT) scans. However, most adopt a multi-stage pipeline that first generates images and then performs segmentation, which leads to error accumulation and fails to leverage shared semantic and anatomical structures. To address this, we propose a unified deep learning framework that generates synthetic CECT images from NCCT scans while simultaneously segmenting the aortic lumen and thrombus. Our approach integrates conditional diffusion models (CDM) with multi-task learning, enabling end-to-end joint optimization of image synthesis and anatomical segmentation. Unlike previous multitask diffusion models, our approach requires no initial predictions (e.g., a coarse segmentation mask), shares both encoder and decoder parameters across tasks, and employs a semi-supervised training strategy to learn from scans with missing segmentation labels, a common constraint in real-world clinical data. We evaluated our method on a cohort of 264 patients, where it consistently outperformed state-of-the-art single-task and multi-stage models. For image synthesis, our model achieved a PSNR of 25.61 dB, compared to 23.80 dB from a single-task CDM. For anatomical segmentation, it improved the lumen Dice score to 0.89 from 0.87 and the challenging thrombus Dice score to 0.53 from 0.48 (nnU-Net). These segmentation enhancements led to more accurate clinical measurements, reducing the lumen diameter MAE to 4.19 mm from 5.78 mm and the thrombus area error to 33.85% from 41.45% when compared to nnU-Net. Code is available at https://github.com/yuxuanou623/AortaDiff.git.
Beyond Distillation: Pushing the Limits of Medical LLM Reasoning with Minimalist Rule-Based RL
May 23, 2025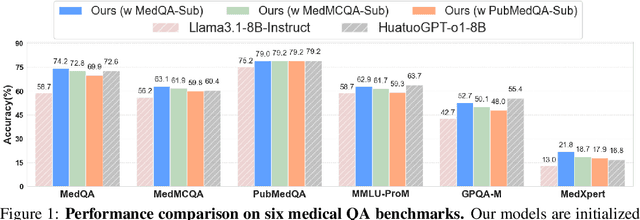
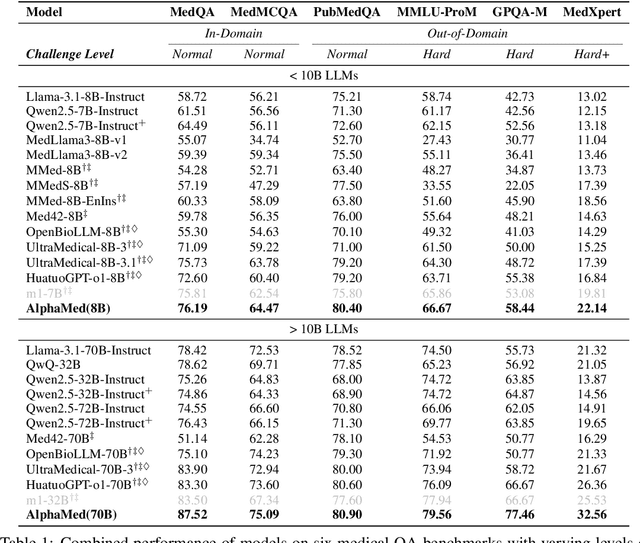
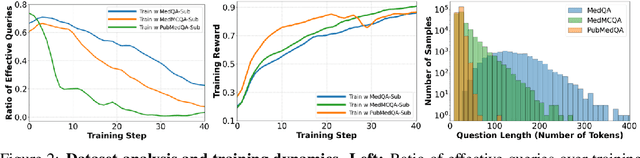
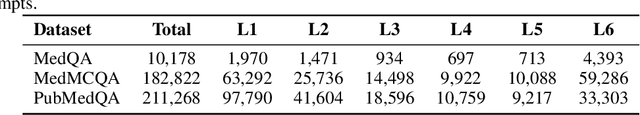
Abstract:Improving performance on complex tasks and enabling interpretable decision making in large language models (LLMs), especially for clinical applications, requires effective reasoning. Yet this remains challenging without supervised fine-tuning (SFT) on costly chain-of-thought (CoT) data distilled from closed-source models (e.g., GPT-4o). In this work, we present AlphaMed, the first medical LLM to show that reasoning capability can emerge purely through reinforcement learning (RL), using minimalist rule-based rewards on public multiple-choice QA datasets, without relying on SFT or distilled CoT data. AlphaMed achieves state-of-the-art results on six medical QA benchmarks, outperforming models trained with conventional SFT+RL pipelines. On challenging benchmarks (e.g., MedXpert), AlphaMed even surpasses larger or closed-source models such as DeepSeek-V3-671B and Claude-3.5-Sonnet. To understand the factors behind this success, we conduct a comprehensive data-centric analysis guided by three questions: (i) Can minimalist rule-based RL incentivize reasoning without distilled CoT supervision? (ii) How do dataset quantity and diversity impact reasoning? (iii) How does question difficulty shape the emergence and generalization of reasoning? Our findings show that dataset informativeness is a key driver of reasoning performance, and that minimalist RL on informative, multiple-choice QA data is effective at inducing reasoning without CoT supervision. We also observe divergent trends across benchmarks, underscoring limitations in current evaluation and the need for more challenging, reasoning-oriented medical QA benchmarks.
Towards Cardiac MRI Foundation Models: Comprehensive Visual-Tabular Representations for Whole-Heart Assessment and Beyond
Apr 18, 2025Abstract:Cardiac magnetic resonance imaging is the gold standard for non-invasive cardiac assessment, offering rich spatio-temporal views of the cardiac anatomy and physiology. Patient-level health factors, such as demographics, metabolic, and lifestyle, are known to substantially influence cardiovascular health and disease risk, yet remain uncaptured by CMR alone. To holistically understand cardiac health and to enable the best possible interpretation of an individual's disease risk, CMR and patient-level factors must be jointly exploited within an integrated framework. Recent multi-modal approaches have begun to bridge this gap, yet they often rely on limited spatio-temporal data and focus on isolated clinical tasks, thereby hindering the development of a comprehensive representation for cardiac health evaluation. To overcome these limitations, we introduce ViTa, a step toward foundation models that delivers a comprehensive representation of the heart and a precise interpretation of individual disease risk. Leveraging data from 42,000 UK Biobank participants, ViTa integrates 3D+T cine stacks from short-axis and long-axis views, enabling a complete capture of the cardiac cycle. These imaging data are then fused with detailed tabular patient-level factors, enabling context-aware insights. This multi-modal paradigm supports a wide spectrum of downstream tasks, including cardiac phenotype and physiological feature prediction, segmentation, and classification of cardiac and metabolic diseases within a single unified framework. By learning a shared latent representation that bridges rich imaging features and patient context, ViTa moves beyond traditional, task-specific models toward a universal, patient-specific understanding of cardiac health, highlighting its potential to advance clinical utility and scalability in cardiac analysis.
MedVLM-R1: Incentivizing Medical Reasoning Capability of Vision-Language Models (VLMs) via Reinforcement Learning
Feb 26, 2025
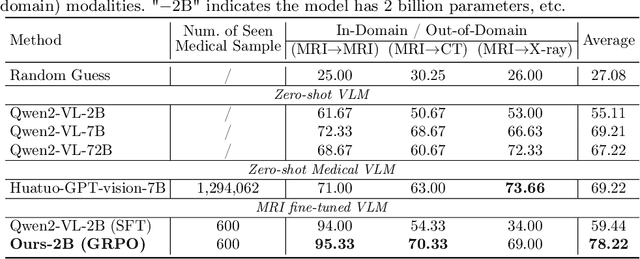
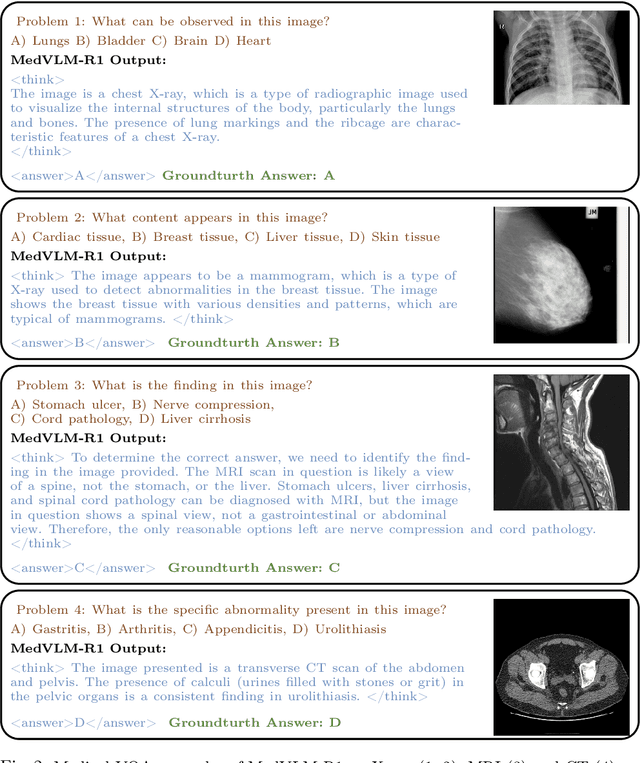
Abstract:Reasoning is a critical frontier for advancing medical image analysis, where transparency and trustworthiness play a central role in both clinician trust and regulatory approval. Although Medical Visual Language Models (VLMs) show promise for radiological tasks, most existing VLMs merely produce final answers without revealing the underlying reasoning. To address this gap, we introduce MedVLM-R1, a medical VLM that explicitly generates natural language reasoning to enhance transparency and trustworthiness. Instead of relying on supervised fine-tuning (SFT), which often suffers from overfitting to training distributions and fails to foster genuine reasoning, MedVLM-R1 employs a reinforcement learning framework that incentivizes the model to discover human-interpretable reasoning paths without using any reasoning references. Despite limited training data (600 visual question answering samples) and model parameters (2B), MedVLM-R1 boosts accuracy from 55.11% to 78.22% across MRI, CT, and X-ray benchmarks, outperforming larger models trained on over a million samples. It also demonstrates robust domain generalization under out-of-distribution tasks. By unifying medical image analysis with explicit reasoning, MedVLM-R1 marks a pivotal step toward trustworthy and interpretable AI in clinical practice.
TimeFlow: Longitudinal Brain Image Registration and Aging Progression Analysis
Jan 15, 2025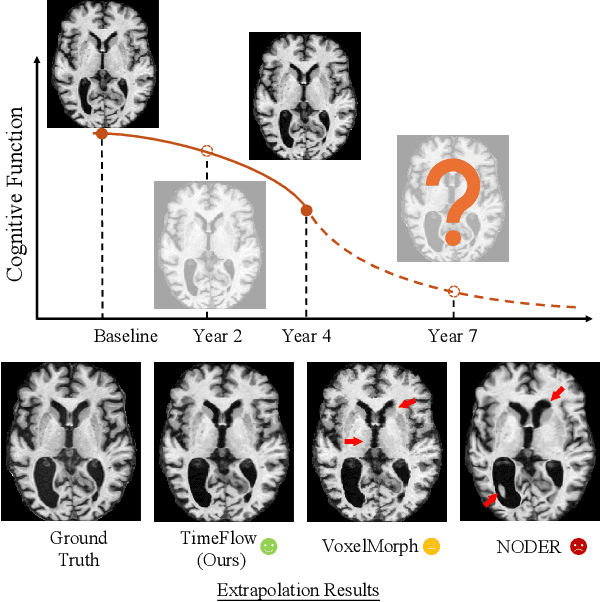
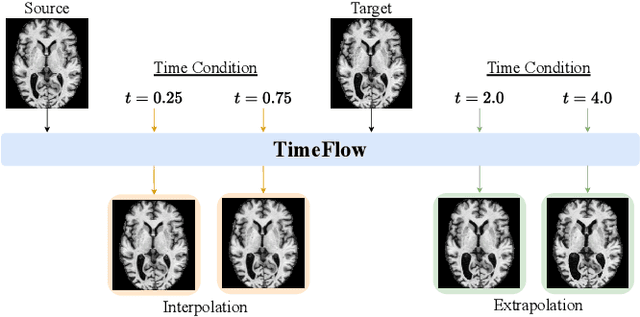
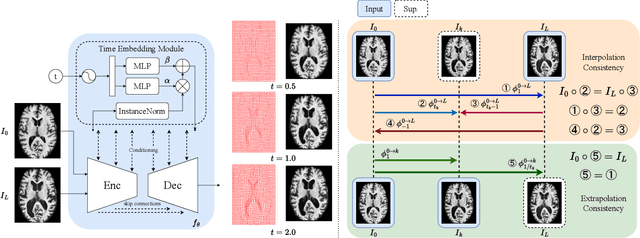
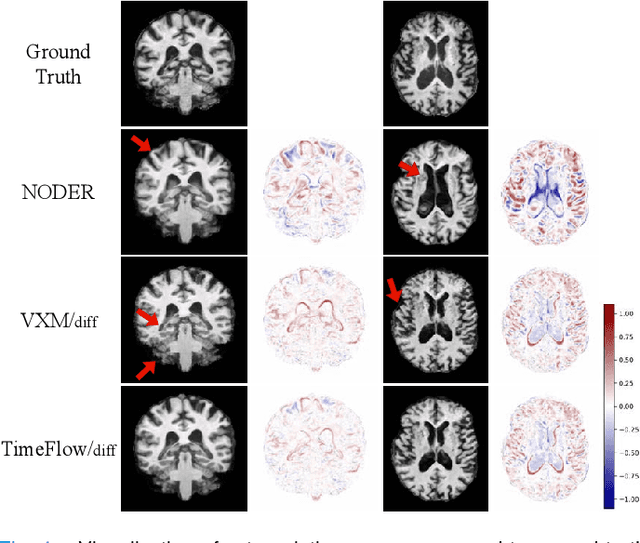
Abstract:Predicting future brain states is crucial for understanding healthy aging and neurodegenerative diseases. Longitudinal brain MRI registration, a cornerstone for such analyses, has long been limited by its inability to forecast future developments, reliance on extensive, dense longitudinal data, and the need to balance registration accuracy with temporal smoothness. In this work, we present \emph{TimeFlow}, a novel framework for longitudinal brain MRI registration that overcomes all these challenges. Leveraging a U-Net architecture with temporal conditioning inspired by diffusion models, TimeFlow enables accurate longitudinal registration and facilitates prospective analyses through future image prediction. Unlike traditional methods that depend on explicit smoothness regularizers and dense sequential data, TimeFlow achieves temporal consistency and continuity without these constraints. Experimental results highlight its superior performance in both future timepoint prediction and registration accuracy compared to state-of-the-art methods. Additionally, TimeFlow supports novel biological brain aging analyses, effectively differentiating neurodegenerative conditions from healthy aging. It eliminates the need for segmentation, thereby avoiding the challenges of non-trivial annotation and inconsistent segmentation errors. TimeFlow paves the way for accurate, data-efficient, and annotation-free prospective analyses of brain aging and chronic diseases.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge