Yuan Bi
Feasibility of Augmented Reality-Guided Robotic Ultrasound with Cone-Beam CT Integration for Spine Procedures
Mar 23, 2026Abstract:Accurate needle placement in spine interventions is critical for effective pain management, yet it depends on reliable identification of anatomical landmarks and careful trajectory planning. Conventional imaging guidance often relies both on CT and X-ray fluoroscopy, exposing patients and staff to high dose of radiation while providing limited real-time 3D feedback. We present an optical see-through augmented reality (OST-AR)-guided robotic system for spine procedures that provides in situ visualization of spinal structures to support needle trajectory planning. We integrate a cone-beam CT (CBCT)-derived 3D spine model which is co-registered with live ultrasound, enabling users to combine global anatomical context with local, real-time imaging. We evaluated the system in a phantom user study involving two representative spine procedures: facet joint injection and lumbar puncture. Sixteen participants performed insertions under two visualization conditions: conventional screen vs. AR. Results show that AR significantly reduces execution time and across-task placement error, while also improving usability, trust, and spatial understanding and lowering cognitive workload. These findings demonstrate the feasibility of AR-guided robotic ultrasound for spine interventions, highlighting its potential to enhance accuracy, efficiency, and user experience in image-guided procedures.
From Scanning Guidelines to Action: A Robotic Ultrasound Agent with LLM-Based Reasoning
Mar 15, 2026Abstract:Robotic ultrasound offers advantages over free-hand scanning, including improved reproducibility and reduced operator dependency. In clinical practice, US acquisition relies heavily on the sonographer's experience and situational judgment. When transferring this process to robotic systems, such expertise is often encoded explicitly through fixed procedures and task-specific models, yielding pipelines that can be difficult to adapt to new scanning tasks. In this work, we propose a unified framework for autonomous robotic US scanning that leverages a LLM-based agent to interpret US scanning guidelines and execute scans by dynamically invoking a set of provided software tools. Instead of encoding fixed scanning procedures, the LLM agent retrieves and reasons over guideline steps from scanning handbooks and adapts its planning decisions based on observations and the current scanning state. This enables the system to handle variable and decision-dependent workflows, such as adjusting scanning strategies, repeating steps, or selecting the appropriate next tool call in response to image quality or anatomical findings. Because the reasoning underlying tool selection is also critical for transparent and trustworthy planning, we further fine tune the LLM agent using a RL based strategy to improve both its reasoning quality and the correctness of tool selection and parameterization, while maintaining robust generalization to unseen guidelines and related tasks. We first validate the approach via verbal execution on 10 US scanning guidelines, assessing reasoning as well as tool selection and parameterization, and showing the benefit of RL fine tuning. We then demonstrate real world feasibility on robotic scanning of the gallbladder, spine, and kidney. Overall, the framework follows diverse guidelines and enables reliable autonomous scanning across multiple anatomical targets within a unified system.
Robotic Ultrasound Makes CBCT Alive
Mar 10, 2026Abstract:Intraoperative Cone Beam Computed Tomography (CBCT) provides a reliable 3D anatomical context essential for interventional planning. However, its static nature fails to provide continuous monitoring of soft-tissue deformations induced by respiration, probe pressure, and surgical manipulation, leading to navigation discrepancies. We propose a deformation-aware CBCT updating framework that leverages robotic ultrasound as a dynamic proxy to infer tissue motion and update static CBCT slices in real time. Starting from calibration-initialized alignment with linear correlation of linear combination (LC2)-based rigid refinement, our method establishes accurate multimodal correspondence. To capture intraoperative dynamics, we introduce the ultrasound correlation UNet (USCorUNet), a lightweight network trained with optical flow-guided supervision to learn deformation-aware correlation representations, enabling accurate, real-time dense deformation field estimation from ultrasound streams. The inferred deformation is spatially regularized and transferred to the CBCT reference to produce deformation-consistent visualizations without repeated radiation exposure. We validate the proposed approach through deformation estimation and ultrasound-guided CBCT updating experiments. Results demonstrate real-time end-to-end CBCT slice updating and physically plausible deformation estimation, enabling dynamic refinement of static CBCT guidance during robotic ultrasound-assisted interventions. The source code is publicly available at https://github.com/anonymous-codebase/us-cbct-demo.
DAISS: Phase-Aware Imitation Learning for Dual-Arm Robotic Ultrasound-Guided Interventions
Mar 08, 2026Abstract:Imitation learning has shown strong potential for automating complex robotic manipulation. In medical robotics, ultrasound-guided needle insertion demands precise bimanual coordination, as clinicians must simultaneously manipulate an ultrasound probe to maintain an optimal acoustic view while steering an interventional needle. Automating this asymmetric workflow -- and reliably transferring expert strategies to robots -- remains highly challenging. In this paper, we present the Dual-Arm Interventional Surgical System (DAISS), a teleoperated platform that collects high-fidelity dual-arm demonstrations and learns a phase-aware imitation policy for ultrasound-guided interventions. To avoid constraining the operator's natural behavior, DAISS uses a flexible NDI-based leader interface for teleoperating two coordinated follower arms. To support robust execution under real-time ultrasound feedback, we develop a lightweight, data-efficient imitation policy. Specifically, the policy incorporates a phase-aware architecture and a dynamic mask loss tailored to asymmetric bimanual control. Conditioned on a planned trajectory, the network fuses real-time ultrasound with external visual observations to generate smooth, coordinated dual-arm motions. Experimental results show that DAISS can learn personalized expert strategies from limited demonstrations. Overall, these findings highlight the promise of phase-aware imitation-learning-driven dual-arm robots for improving precision and reducing cognitive workload in image-guided interventions.
Ultrasound-Guided Real-Time Spinal Motion Visualization for Spinal Instability Assessment
Feb 13, 2026Abstract:Purpose: Spinal instability is a widespread condition that causes pain, fatigue, and restricted mobility, profoundly affecting patients' quality of life. In clinical practice, the gold standard for diagnosis is dynamic X-ray imaging. However, X-ray provides only 2D motion information, while 3D modalities such as computed tomography (CT) or cone beam computed tomography (CBCT) cannot efficiently capture motion. Therefore, there is a need for a system capable of visualizing real-time 3D spinal motion while minimizing radiation exposure. Methods: We propose ultrasound as an auxiliary modality for 3D spine visualization. Due to acoustic limitations, ultrasound captures only the superficial spinal surface. Therefore, the partially compounded ultrasound volume is registered to preoperative 3D imaging. In this study, CBCT provides the neutral spine configuration, while robotic ultrasound acquisition is performed at maximal spinal bending. A kinematic model is applied to the CBCT-derived spine model for coarse registration, followed by ICP for fine registration, with kinematic parameters optimized based on the registration results. Real-time ultrasound motion tracking is then used to estimate continuous 3D spinal motion by interpolating between the neutral and maximally bent states. Results: The pipeline was evaluated on a bendable 3D-printed lumbar spine phantom. The registration error was $1.941 \pm 0.199$ mm and the interpolated spinal motion error was $2.01 \pm 0.309$ mm (median). Conclusion: The proposed robotic ultrasound framework enables radiation-reduced, real-time 3D visualization of spinal motion, offering a promising 3D alternative to conventional dynamic X-ray imaging for assessing spinal instability.
Intelligent Virtual Sonographer (IVS): Enhancing Physician-Robot-Patient Communication
Jul 17, 2025Abstract:The advancement and maturity of large language models (LLMs) and robotics have unlocked vast potential for human-computer interaction, particularly in the field of robotic ultrasound. While existing research primarily focuses on either patient-robot or physician-robot interaction, the role of an intelligent virtual sonographer (IVS) bridging physician-robot-patient communication remains underexplored. This work introduces a conversational virtual agent in Extended Reality (XR) that facilitates real-time interaction between physicians, a robotic ultrasound system(RUS), and patients. The IVS agent communicates with physicians in a professional manner while offering empathetic explanations and reassurance to patients. Furthermore, it actively controls the RUS by executing physician commands and transparently relays these actions to the patient. By integrating LLM-powered dialogue with speech-to-text, text-to-speech, and robotic control, our system enhances the efficiency, clarity, and accessibility of robotic ultrasound acquisition. This work constitutes a first step toward understanding how IVS can bridge communication gaps in physician-robot-patient interaction, providing more control and therefore trust into physician-robot interaction while improving patient experience and acceptance of robotic ultrasound.
UltraAD: Fine-Grained Ultrasound Anomaly Classification via Few-Shot CLIP Adaptation
Jun 24, 2025Abstract:Precise anomaly detection in medical images is critical for clinical decision-making. While recent unsupervised or semi-supervised anomaly detection methods trained on large-scale normal data show promising results, they lack fine-grained differentiation, such as benign vs. malignant tumors. Additionally, ultrasound (US) imaging is highly sensitive to devices and acquisition parameter variations, creating significant domain gaps in the resulting US images. To address these challenges, we propose UltraAD, a vision-language model (VLM)-based approach that leverages few-shot US examples for generalized anomaly localization and fine-grained classification. To enhance localization performance, the image-level token of query visual prototypes is first fused with learnable text embeddings. This image-informed prompt feature is then further integrated with patch-level tokens, refining local representations for improved accuracy. For fine-grained classification, a memory bank is constructed from few-shot image samples and corresponding text descriptions that capture anatomical and abnormality-specific features. During training, the stored text embeddings remain frozen, while image features are adapted to better align with medical data. UltraAD has been extensively evaluated on three breast US datasets, outperforming state-of-the-art methods in both lesion localization and fine-grained medical classification. The code will be released upon acceptance.
Does Machine Unlearning Truly Remove Model Knowledge? A Framework for Auditing Unlearning in LLMs
May 29, 2025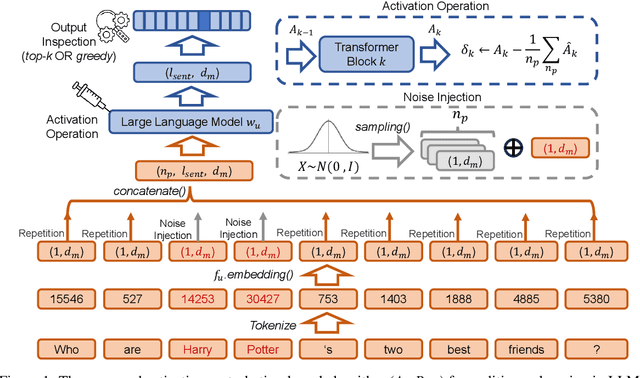
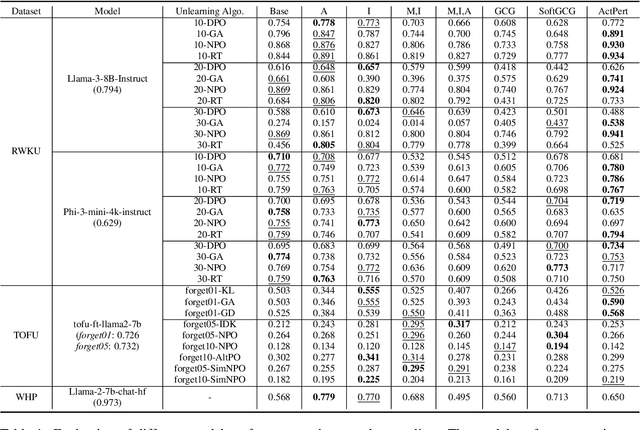
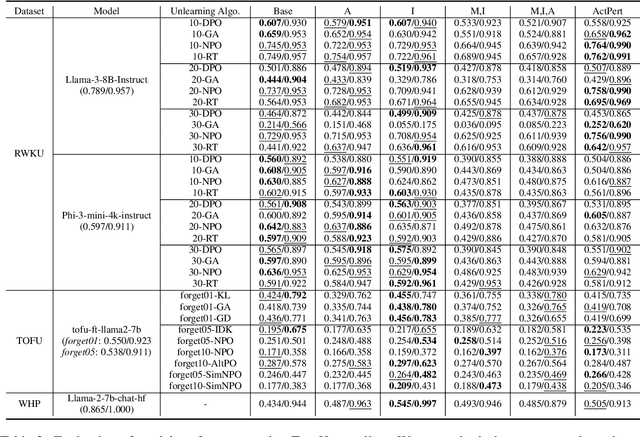
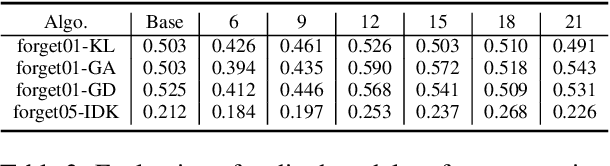
Abstract:In recent years, Large Language Models (LLMs) have achieved remarkable advancements, drawing significant attention from the research community. Their capabilities are largely attributed to large-scale architectures, which require extensive training on massive datasets. However, such datasets often contain sensitive or copyrighted content sourced from the public internet, raising concerns about data privacy and ownership. Regulatory frameworks, such as the General Data Protection Regulation (GDPR), grant individuals the right to request the removal of such sensitive information. This has motivated the development of machine unlearning algorithms that aim to remove specific knowledge from models without the need for costly retraining. Despite these advancements, evaluating the efficacy of unlearning algorithms remains a challenge due to the inherent complexity and generative nature of LLMs. In this work, we introduce a comprehensive auditing framework for unlearning evaluation, comprising three benchmark datasets, six unlearning algorithms, and five prompt-based auditing methods. By using various auditing algorithms, we evaluate the effectiveness and robustness of different unlearning strategies. To explore alternatives beyond prompt-based auditing, we propose a novel technique that leverages intermediate activation perturbations, addressing the limitations of auditing methods that rely solely on model inputs and outputs.
Robotic CBCT Meets Robotic Ultrasound
Feb 17, 2025Abstract:The multi-modality imaging system offers optimal fused images for safe and precise interventions in modern clinical practices, such as computed tomography - ultrasound (CT-US) guidance for needle insertion. However, the limited dexterity and mobility of current imaging devices hinder their integration into standardized workflows and the advancement toward fully autonomous intervention systems. In this paper, we present a novel clinical setup where robotic cone beam computed tomography (CBCT) and robotic US are pre-calibrated and dynamically co-registered, enabling new clinical applications. This setup allows registration-free rigid registration, facilitating multi-modal guided procedures in the absence of tissue deformation. First, a one-time pre-calibration is performed between the systems. To ensure a safe insertion path by highlighting critical vasculature on the 3D CBCT, SAM2 segments vessels from B-mode images, using the Doppler signal as an autonomously generated prompt. Based on the registration, the Doppler image or segmented vessel masks are then mapped onto the CBCT, creating an optimally fused image with comprehensive detail. To validate the system, we used a specially designed phantom, featuring lesions covered by ribs and multiple vessels with simulated moving flow. The mapping error between US and CBCT resulted in an average deviation of 1.72+-0.62 mm. A user study demonstrated the effectiveness of CBCT-US fusion for needle insertion guidance, showing significant improvements in time efficiency, accuracy, and success rate. Needle intervention performance improved by approximately 50% compared to the conventional US-guided workflow. We present the first robotic dual-modality imaging system designed to guide clinical applications. The results show significant performance improvements compared to traditional manual interventions.
Gaze-Guided Robotic Vascular Ultrasound Leveraging Human Intention Estimation
Feb 07, 2025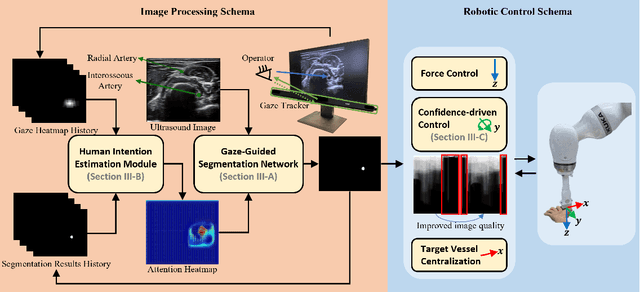
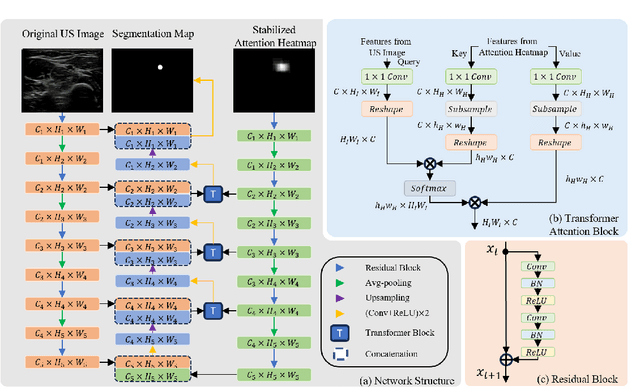
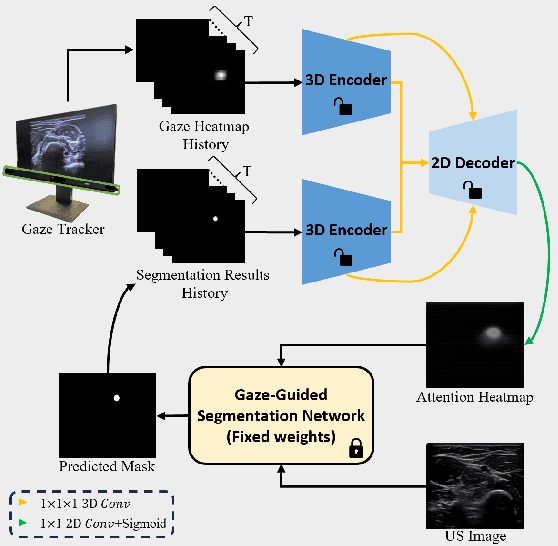
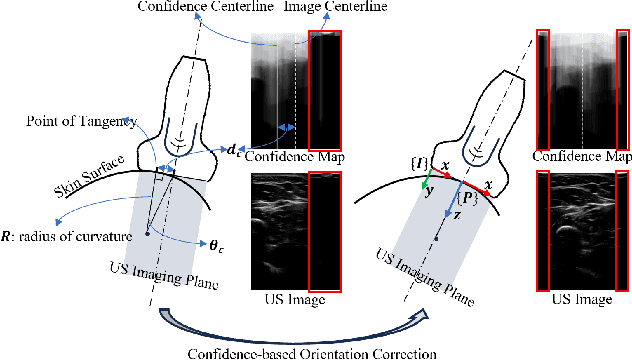
Abstract:Medical ultrasound has been widely used to examine vascular structure in modern clinical practice. However, traditional ultrasound examination often faces challenges related to inter- and intra-operator variation. The robotic ultrasound system (RUSS) appears as a potential solution for such challenges because of its superiority in stability and reproducibility. Given the complex anatomy of human vasculature, multiple vessels often appear in ultrasound images, or a single vessel bifurcates into branches, complicating the examination process. To tackle this challenge, this work presents a gaze-guided RUSS for vascular applications. A gaze tracker captures the eye movements of the operator. The extracted gaze signal guides the RUSS to follow the correct vessel when it bifurcates. Additionally, a gaze-guided segmentation network is proposed to enhance segmentation robustness by exploiting gaze information. However, gaze signals are often noisy, requiring interpretation to accurately discern the operator's true intentions. To this end, this study proposes a stabilization module to process raw gaze data. The inferred attention heatmap is utilized as a region proposal to aid segmentation and serve as a trigger signal when the operator needs to adjust the scanning target, such as when a bifurcation appears. To ensure appropriate contact between the probe and surface during scanning, an automatic ultrasound confidence-based orientation correction method is developed. In experiments, we demonstrated the efficiency of the proposed gaze-guided segmentation pipeline by comparing it with other methods. Besides, the performance of the proposed gaze-guided RUSS was also validated as a whole on a realistic arm phantom with an uneven surface.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge