Nicolo Savioli
Box2Poly: Memory-Efficient Polygon Prediction of Arbitrarily Shaped and Rotated Text
Sep 20, 2023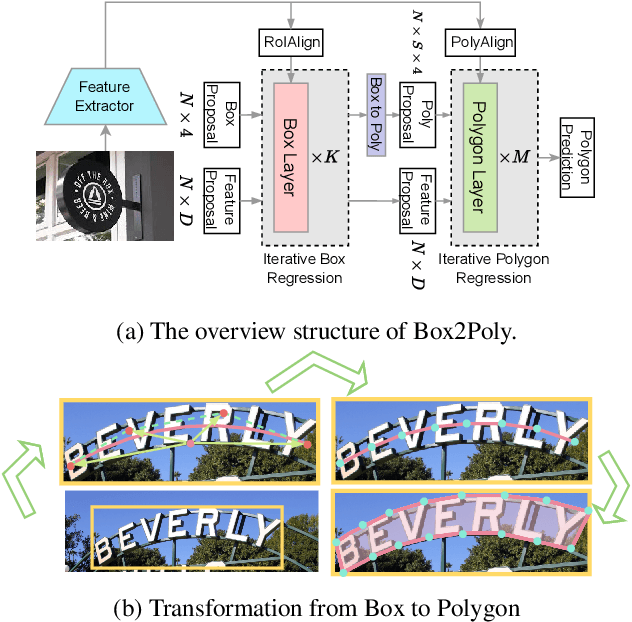

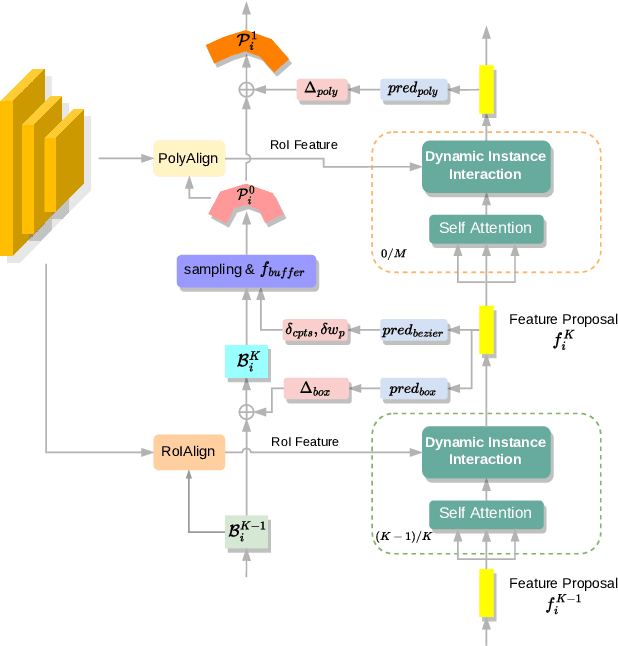

Abstract:Recently, Transformer-based text detection techniques have sought to predict polygons by encoding the coordinates of individual boundary vertices using distinct query features. However, this approach incurs a significant memory overhead and struggles to effectively capture the intricate relationships between vertices belonging to the same instance. Consequently, irregular text layouts often lead to the prediction of outlined vertices, diminishing the quality of results. To address these challenges, we present an innovative approach rooted in Sparse R-CNN: a cascade decoding pipeline for polygon prediction. Our method ensures precision by iteratively refining polygon predictions, considering both the scale and location of preceding results. Leveraging this stabilized regression pipeline, even employing just a single feature vector to guide polygon instance regression yields promising detection results. Simultaneously, the leverage of instance-level feature proposal substantially enhances memory efficiency (>50% less vs. the state-of-the-art method DPText-DETR) and reduces inference speed (>40% less vs. DPText-DETR) with minor performance drop on benchmarks.
Joint Semi-supervised 3D Super-Resolution and Segmentation with Mixed Adversarial Gaussian Domain Adaptation
Jul 16, 2021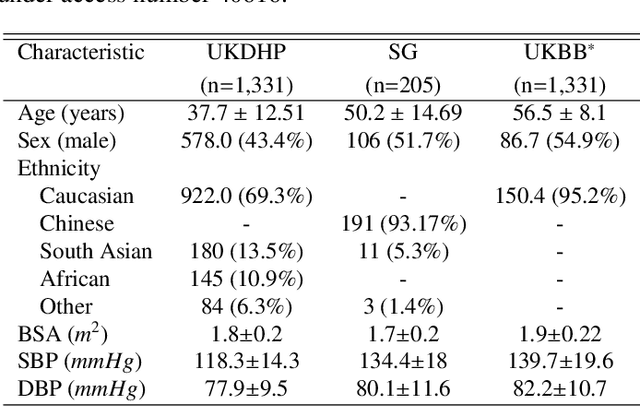
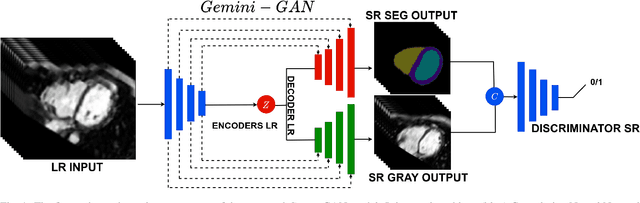
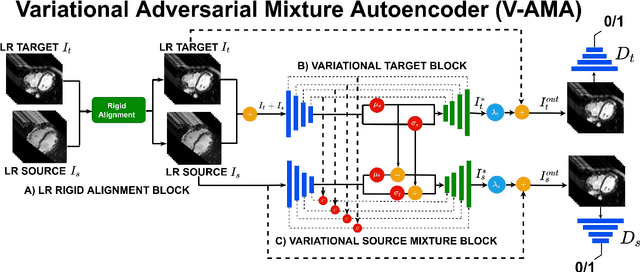
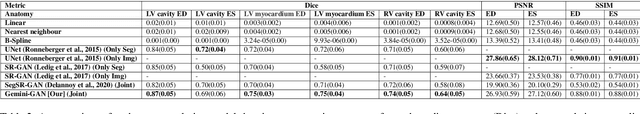
Abstract:Optimising the analysis of cardiac structure and function requires accurate 3D representations of shape and motion. However, techniques such as cardiac magnetic resonance imaging are conventionally limited to acquiring contiguous cross-sectional slices with low through-plane resolution and potential inter-slice spatial misalignment. Super-resolution in medical imaging aims to increase the resolution of images but is conventionally trained on features from low resolution datasets and does not super-resolve corresponding segmentations. Here we propose a semi-supervised multi-task generative adversarial network (Gemini-GAN) that performs joint super-resolution of the images and their labels using a ground truth of high resolution 3D cines and segmentations, while an unsupervised variational adversarial mixture autoencoder (V-AMA) is used for continuous domain adaptation. Our proposed approach is extensively evaluated on two transnational multi-ethnic populations of 1,331 and 205 adults respectively, delivering an improvement on state of the art methods in terms of Dice index, peak signal to noise ratio, and structural similarity index measure. This framework also exceeds the performance of state of the art generative domain adaptation models on external validation (Dice index 0.81 vs 0.74 for the left ventricle). This demonstrates how joint super-resolution and segmentation, trained on 3D ground-truth data with cross-domain generalization, enables robust precision phenotyping in diverse populations.
Joint Motion Correction and Super Resolution for Cardiac Segmentation via Latent Optimisation
Jul 08, 2021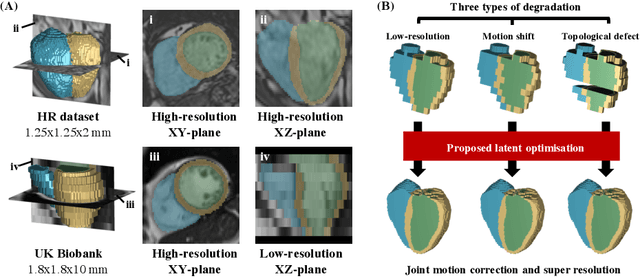

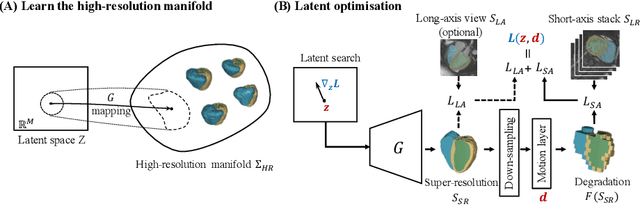
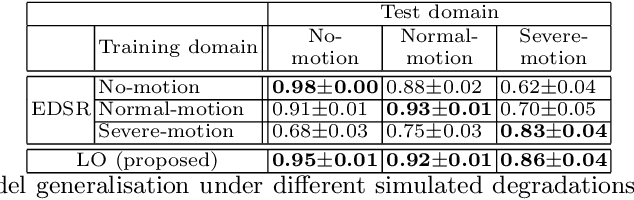
Abstract:In cardiac magnetic resonance (CMR) imaging, a 3D high-resolution segmentation of the heart is essential for detailed description of its anatomical structures. However, due to the limit of acquisition duration and respiratory/cardiac motion, stacks of multi-slice 2D images are acquired in clinical routine. The segmentation of these images provides a low-resolution representation of cardiac anatomy, which may contain artefacts caused by motion. Here we propose a novel latent optimisation framework that jointly performs motion correction and super resolution for cardiac image segmentations. Given a low-resolution segmentation as input, the framework accounts for inter-slice motion in cardiac MR imaging and super-resolves the input into a high-resolution segmentation consistent with input. A multi-view loss is incorporated to leverage information from both short-axis view and long-axis view of cardiac imaging. To solve the inverse problem, iterative optimisation is performed in a latent space, which ensures the anatomical plausibility. This alleviates the need of paired low-resolution and high-resolution images for supervised learning. Experiments on two cardiac MR datasets show that the proposed framework achieves high performance, comparable to state-of-the-art super-resolution approaches and with better cross-domain generalisability and anatomical plausibility.
A Global Benchmark of Algorithms for Segmenting Late Gadolinium-Enhanced Cardiac Magnetic Resonance Imaging
May 07, 2020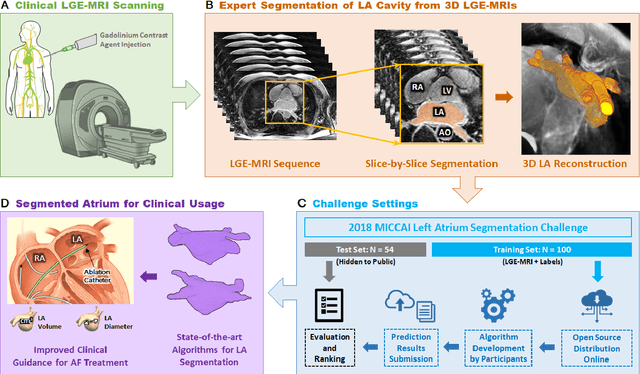
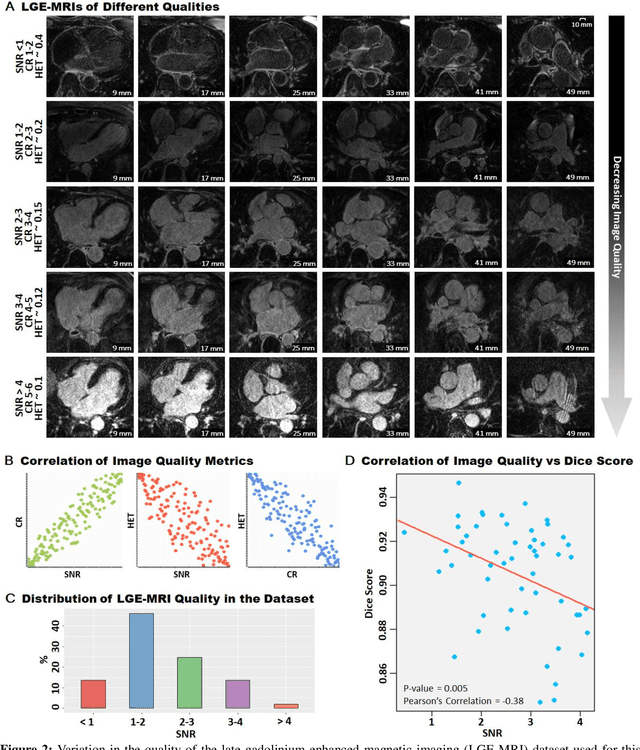
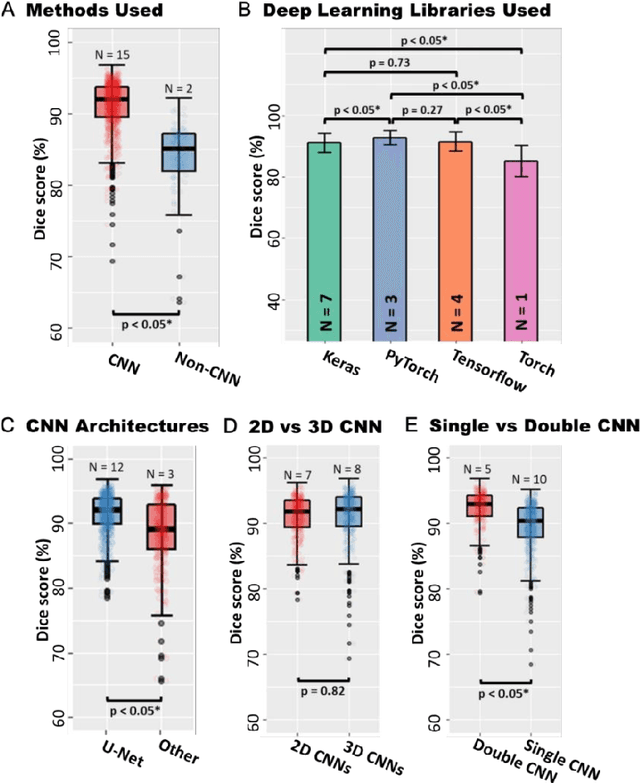
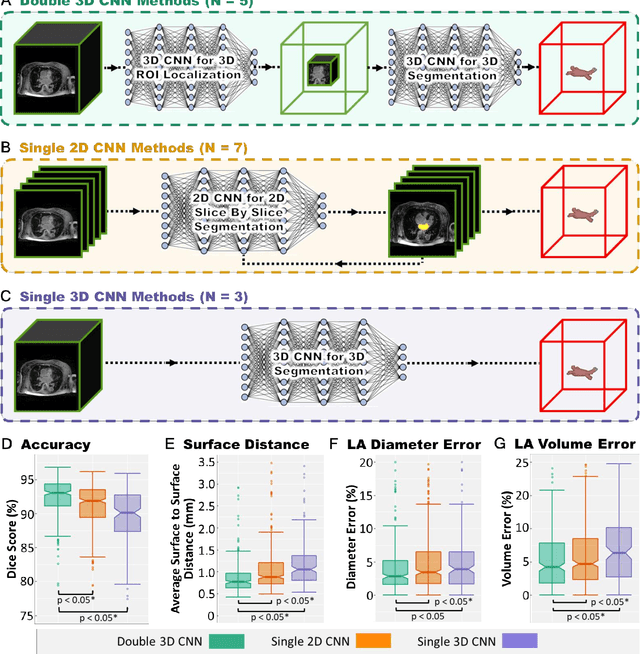
Abstract:Segmentation of cardiac images, particularly late gadolinium-enhanced magnetic resonance imaging (LGE-MRI) widely used for visualizing diseased cardiac structures, is a crucial first step for clinical diagnosis and treatment. However, direct segmentation of LGE-MRIs is challenging due to its attenuated contrast. Since most clinical studies have relied on manual and labor-intensive approaches, automatic methods are of high interest, particularly optimized machine learning approaches. To address this, we organized the "2018 Left Atrium Segmentation Challenge" using 154 3D LGE-MRIs, currently the world's largest cardiac LGE-MRI dataset, and associated labels of the left atrium segmented by three medical experts, ultimately attracting the participation of 27 international teams. In this paper, extensive analysis of the submitted algorithms using technical and biological metrics was performed by undergoing subgroup analysis and conducting hyper-parameter analysis, offering an overall picture of the major design choices of convolutional neural networks (CNNs) and practical considerations for achieving state-of-the-art left atrium segmentation. Results show the top method achieved a dice score of 93.2% and a mean surface to a surface distance of 0.7 mm, significantly outperforming prior state-of-the-art. Particularly, our analysis demonstrated that double, sequentially used CNNs, in which a first CNN is used for automatic region-of-interest localization and a subsequent CNN is used for refined regional segmentation, achieved far superior results than traditional methods and pipelines containing single CNNs. This large-scale benchmarking study makes a significant step towards much-improved segmentation methods for cardiac LGE-MRIs, and will serve as an important benchmark for evaluating and comparing the future works in the field.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge