Virginia Fernandez
King's College London
DIST-CLIP: Arbitrary Metadata and Image Guided MRI Harmonization via Disentangled Anatomy-Contrast Representations
Dec 08, 2025Abstract:Deep learning holds immense promise for transforming medical image analysis, yet its clinical generalization remains profoundly limited. A major barrier is data heterogeneity. This is particularly true in Magnetic Resonance Imaging, where scanner hardware differences, diverse acquisition protocols, and varying sequence parameters introduce substantial domain shifts that obscure underlying biological signals. Data harmonization methods aim to reduce these instrumental and acquisition variability, but existing approaches remain insufficient. When applied to imaging data, image-based harmonization approaches are often restricted by the need for target images, while existing text-guided methods rely on simplistic labels that fail to capture complex acquisition details or are typically restricted to datasets with limited variability, failing to capture the heterogeneity of real-world clinical environments. To address these limitations, we propose DIST-CLIP (Disentangled Style Transfer with CLIP Guidance), a unified framework for MRI harmonization that flexibly uses either target images or DICOM metadata for guidance. Our framework explicitly disentangles anatomical content from image contrast, with the contrast representations being extracted using pre-trained CLIP encoders. These contrast embeddings are then integrated into the anatomical content via a novel Adaptive Style Transfer module. We trained and evaluated DIST-CLIP on diverse real-world clinical datasets, and showed significant improvements in performance when compared against state-of-the-art methods in both style translation fidelity and anatomical preservation, offering a flexible solution for style transfer and standardizing MRI data. Our code and weights will be made publicly available upon publication.
Diffusion-Based Quality Control of Medical Image Segmentations across Organs
Nov 12, 2025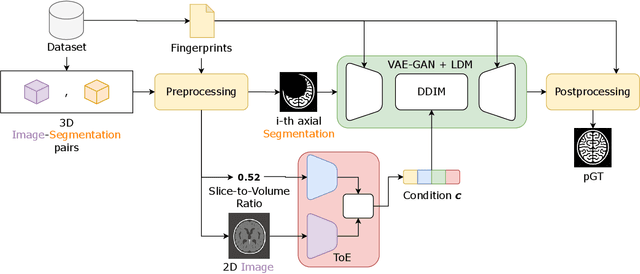
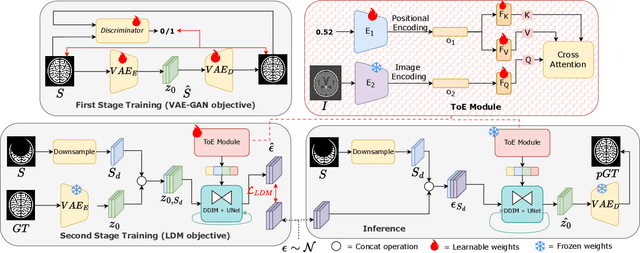
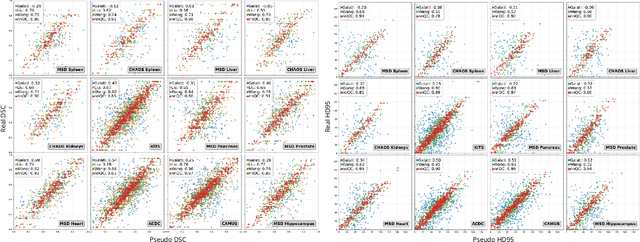
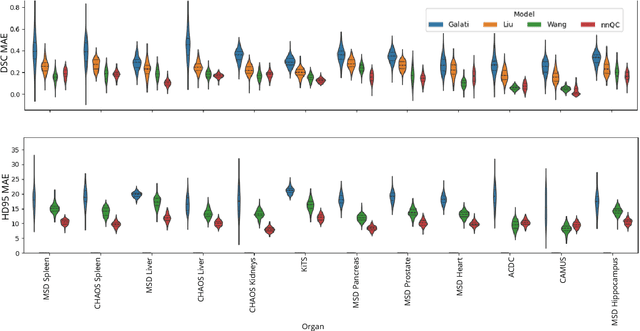
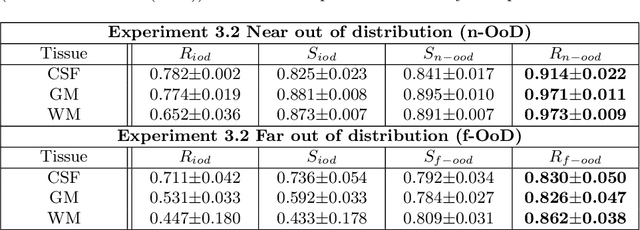
Abstract:Medical image segmentation using deep learning (DL) has enabled the development of automated analysis pipelines for large-scale population studies. However, state-of-the-art DL methods are prone to hallucinations, which can result in anatomically implausible segmentations. With manual correction impractical at scale, automated quality control (QC) techniques have to address the challenge. While promising, existing QC methods are organ-specific, limiting their generalizability and usability beyond their original intended task. To overcome this limitation, we propose no-new Quality Control (nnQC), a robust QC framework based on a diffusion-generative paradigm that self-adapts to any input organ dataset. Central to nnQC is a novel Team of Experts (ToE) architecture, where two specialized experts independently encode 3D spatial awareness, represented by the relative spatial position of an axial slice, and anatomical information derived from visual features from the original image. A weighted conditional module dynamically combines the pair of independent embeddings, or opinions to condition the sampling mechanism within a diffusion process, enabling the generation of a spatially aware pseudo-ground truth for predicting QC scores. Within its framework, nnQC integrates fingerprint adaptation to ensure adaptability across organs, datasets, and imaging modalities. We evaluated nnQC on seven organs using twelve publicly available datasets. Our results demonstrate that nnQC consistently outperforms state-of-the-art methods across all experiments, including cases where segmentation masks are highly degraded or completely missing, confirming its versatility and effectiveness across different organs.
A methodology for clinically driven interactive segmentation evaluation
Oct 10, 2025Abstract:Interactive segmentation is a promising strategy for building robust, generalisable algorithms for volumetric medical image segmentation. However, inconsistent and clinically unrealistic evaluation hinders fair comparison and misrepresents real-world performance. We propose a clinically grounded methodology for defining evaluation tasks and metrics, and built a software framework for constructing standardised evaluation pipelines. We evaluate state-of-the-art algorithms across heterogeneous and complex tasks and observe that (i) minimising information loss when processing user interactions is critical for model robustness, (ii) adaptive-zooming mechanisms boost robustness and speed convergence, (iii) performance drops if validation prompting behaviour/budgets differ from training, (iv) 2D methods perform well with slab-like images and coarse targets, but 3D context helps with large or irregularly shaped targets, (v) performance of non-medical-domain models (e.g. SAM2) degrades with poor contrast and complex shapes.
A 3D generative model of pathological multi-modal MR images and segmentations
Nov 08, 2023Abstract:Generative modelling and synthetic data can be a surrogate for real medical imaging datasets, whose scarcity and difficulty to share can be a nuisance when delivering accurate deep learning models for healthcare applications. In recent years, there has been an increased interest in using these models for data augmentation and synthetic data sharing, using architectures such as generative adversarial networks (GANs) or diffusion models (DMs). Nonetheless, the application of synthetic data to tasks such as 3D magnetic resonance imaging (MRI) segmentation remains limited due to the lack of labels associated with the generated images. Moreover, many of the proposed generative MRI models lack the ability to generate arbitrary modalities due to the absence of explicit contrast conditioning. These limitations prevent the user from adjusting the contrast and content of the images and obtaining more generalisable data for training task-specific models. In this work, we propose brainSPADE3D, a 3D generative model for brain MRI and associated segmentations, where the user can condition on specific pathological phenotypes and contrasts. The proposed joint imaging-segmentation generative model is shown to generate high-fidelity synthetic images and associated segmentations, with the ability to combine pathologies. We demonstrate how the model can alleviate issues with segmentation model performance when unexpected pathologies are present in the data.
Generative AI for Medical Imaging: extending the MONAI Framework
Jul 27, 2023Abstract:Recent advances in generative AI have brought incredible breakthroughs in several areas, including medical imaging. These generative models have tremendous potential not only to help safely share medical data via synthetic datasets but also to perform an array of diverse applications, such as anomaly detection, image-to-image translation, denoising, and MRI reconstruction. However, due to the complexity of these models, their implementation and reproducibility can be difficult. This complexity can hinder progress, act as a use barrier, and dissuade the comparison of new methods with existing works. In this study, we present MONAI Generative Models, a freely available open-source platform that allows researchers and developers to easily train, evaluate, and deploy generative models and related applications. Our platform reproduces state-of-art studies in a standardised way involving different architectures (such as diffusion models, autoregressive transformers, and GANs), and provides pre-trained models for the community. We have implemented these models in a generalisable fashion, illustrating that their results can be extended to 2D or 3D scenarios, including medical images with different modalities (like CT, MRI, and X-Ray data) and from different anatomical areas. Finally, we adopt a modular and extensible approach, ensuring long-term maintainability and the extension of current applications for future features.
Privacy Distillation: Reducing Re-identification Risk of Multimodal Diffusion Models
Jun 02, 2023



Abstract:Knowledge distillation in neural networks refers to compressing a large model or dataset into a smaller version of itself. We introduce Privacy Distillation, a framework that allows a text-to-image generative model to teach another model without exposing it to identifiable data. Here, we are interested in the privacy issue faced by a data provider who wishes to share their data via a multimodal generative model. A question that immediately arises is ``How can a data provider ensure that the generative model is not leaking identifiable information about a patient?''. Our solution consists of (1) training a first diffusion model on real data (2) generating a synthetic dataset using this model and filtering it to exclude images with a re-identifiability risk (3) training a second diffusion model on the filtered synthetic data only. We showcase that datasets sampled from models trained with privacy distillation can effectively reduce re-identification risk whilst maintaining downstream performance.
Transfer Learning for Fine-grained Classification Using Semi-supervised Learning and Visual Transformers
May 17, 2023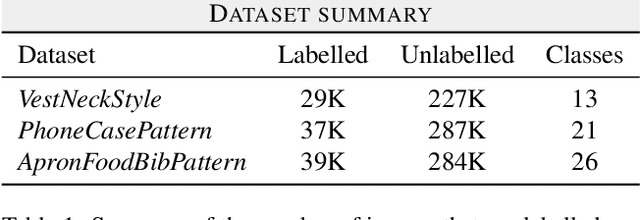

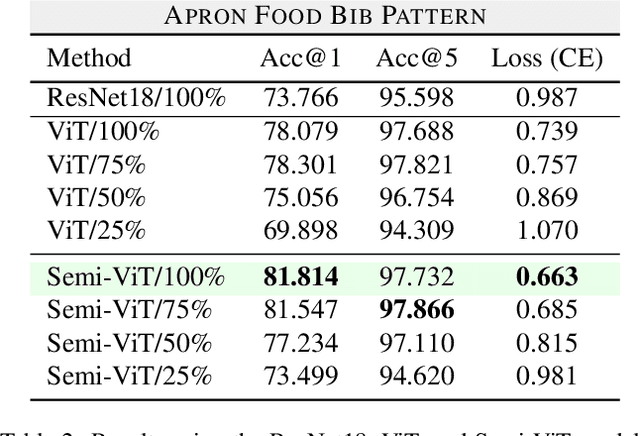
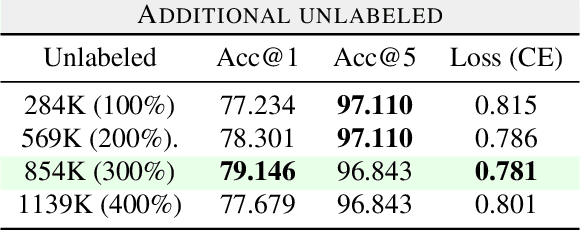
Abstract:Fine-grained classification is a challenging task that involves identifying subtle differences between objects within the same category. This task is particularly challenging in scenarios where data is scarce. Visual transformers (ViT) have recently emerged as a powerful tool for image classification, due to their ability to learn highly expressive representations of visual data using self-attention mechanisms. In this work, we explore Semi-ViT, a ViT model fine tuned using semi-supervised learning techniques, suitable for situations where we have lack of annotated data. This is particularly common in e-commerce, where images are readily available but labels are noisy, nonexistent, or expensive to obtain. Our results demonstrate that Semi-ViT outperforms traditional convolutional neural networks (CNN) and ViTs, even when fine-tuned with limited annotated data. These findings indicate that Semi-ViTs hold significant promise for applications that require precise and fine-grained classification of visual data.
Can segmentation models be trained with fully synthetically generated data?
Sep 17, 2022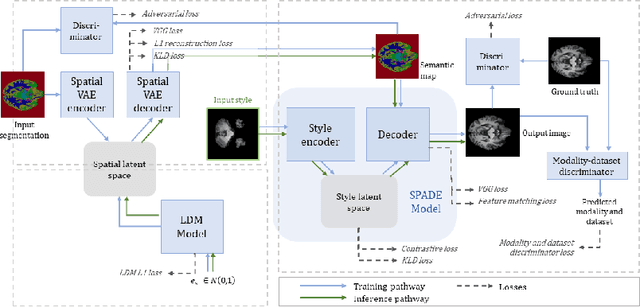

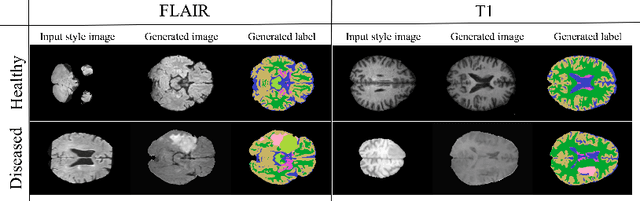
Abstract:In order to achieve good performance and generalisability, medical image segmentation models should be trained on sizeable datasets with sufficient variability. Due to ethics and governance restrictions, and the costs associated with labelling data, scientific development is often stifled, with models trained and tested on limited data. Data augmentation is often used to artificially increase the variability in the data distribution and improve model generalisability. Recent works have explored deep generative models for image synthesis, as such an approach would enable the generation of an effectively infinite amount of varied data, addressing the generalisability and data access problems. However, many proposed solutions limit the user's control over what is generated. In this work, we propose brainSPADE, a model which combines a synthetic diffusion-based label generator with a semantic image generator. Our model can produce fully synthetic brain labels on-demand, with or without pathology of interest, and then generate a corresponding MRI image of an arbitrary guided style. Experiments show that brainSPADE synthetic data can be used to train segmentation models with performance comparable to that of models trained on real data.
Brain Imaging Generation with Latent Diffusion Models
Sep 15, 2022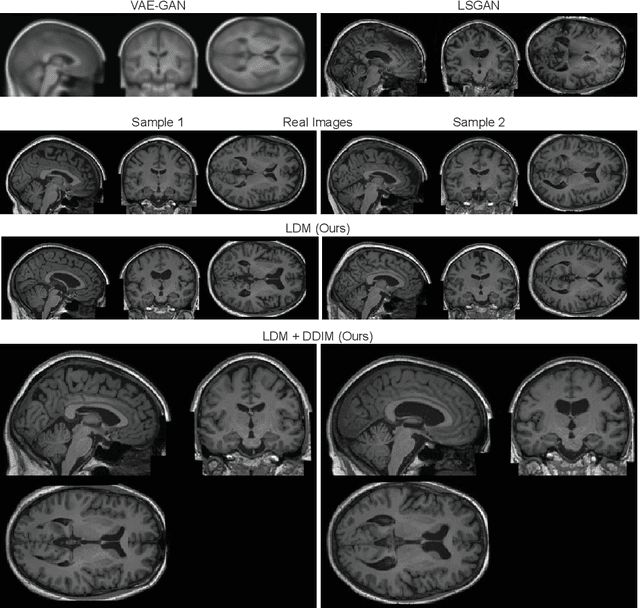
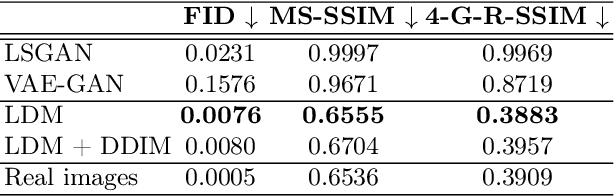
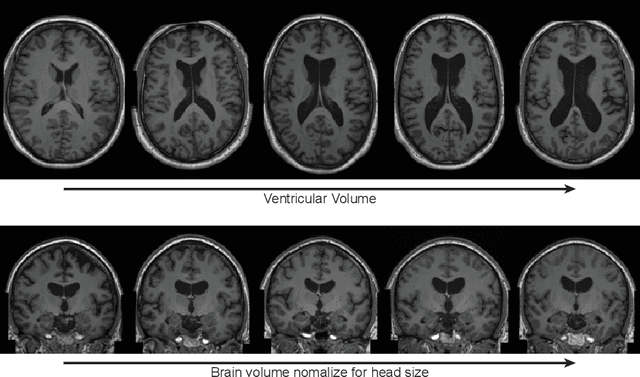
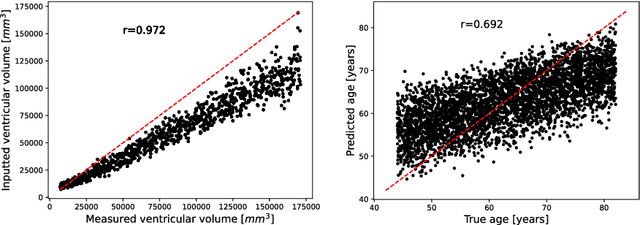
Abstract:Deep neural networks have brought remarkable breakthroughs in medical image analysis. However, due to their data-hungry nature, the modest dataset sizes in medical imaging projects might be hindering their full potential. Generating synthetic data provides a promising alternative, allowing to complement training datasets and conducting medical image research at a larger scale. Diffusion models recently have caught the attention of the computer vision community by producing photorealistic synthetic images. In this study, we explore using Latent Diffusion Models to generate synthetic images from high-resolution 3D brain images. We used T1w MRI images from the UK Biobank dataset (N=31,740) to train our models to learn about the probabilistic distribution of brain images, conditioned on covariables, such as age, sex, and brain structure volumes. We found that our models created realistic data, and we could use the conditioning variables to control the data generation effectively. Besides that, we created a synthetic dataset with 100,000 brain images and made it openly available to the scientific community.
Morphology-preserving Autoregressive 3D Generative Modelling of the Brain
Sep 07, 2022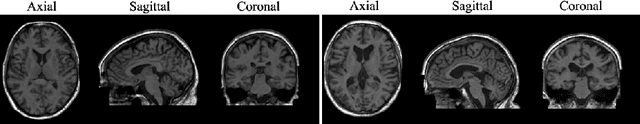
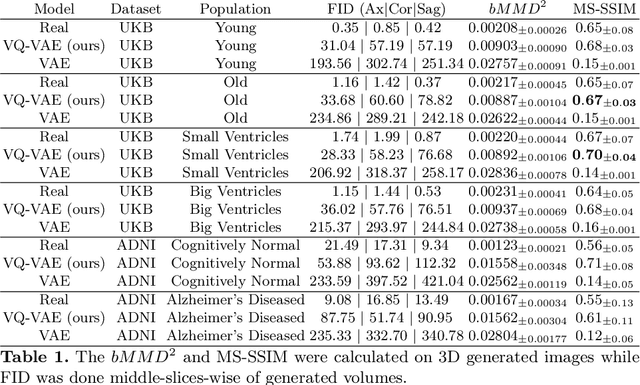
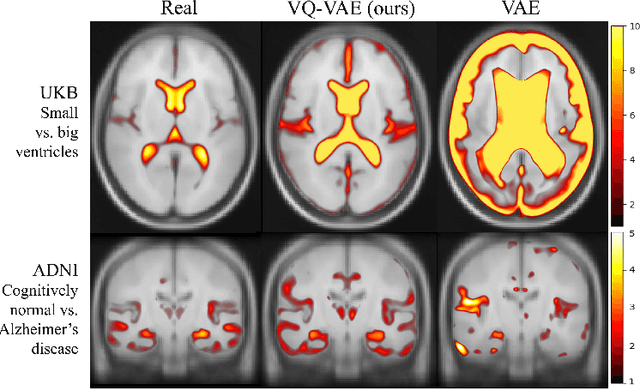
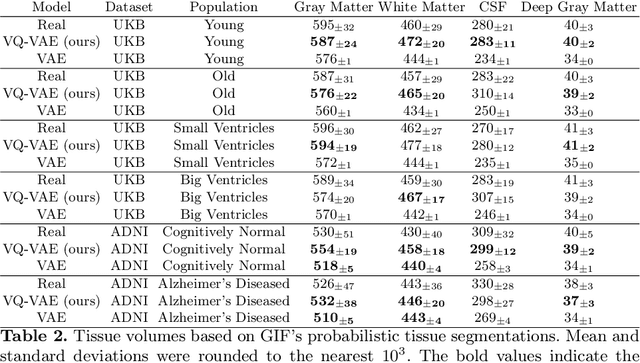
Abstract:Human anatomy, morphology, and associated diseases can be studied using medical imaging data. However, access to medical imaging data is restricted by governance and privacy concerns, data ownership, and the cost of acquisition, thus limiting our ability to understand the human body. A possible solution to this issue is the creation of a model able to learn and then generate synthetic images of the human body conditioned on specific characteristics of relevance (e.g., age, sex, and disease status). Deep generative models, in the form of neural networks, have been recently used to create synthetic 2D images of natural scenes. Still, the ability to produce high-resolution 3D volumetric imaging data with correct anatomical morphology has been hampered by data scarcity and algorithmic and computational limitations. This work proposes a generative model that can be scaled to produce anatomically correct, high-resolution, and realistic images of the human brain, with the necessary quality to allow further downstream analyses. The ability to generate a potentially unlimited amount of data not only enables large-scale studies of human anatomy and pathology without jeopardizing patient privacy, but also significantly advances research in the field of anomaly detection, modality synthesis, learning under limited data, and fair and ethical AI. Code and trained models are available at: https://github.com/AmigoLab/SynthAnatomy.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge