Francesco La Rosa
Structural-Based Uncertainty in Deep Learning Across Anatomical Scales: Analysis in White Matter Lesion Segmentation
Nov 15, 2023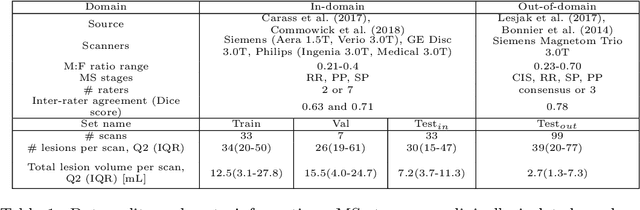


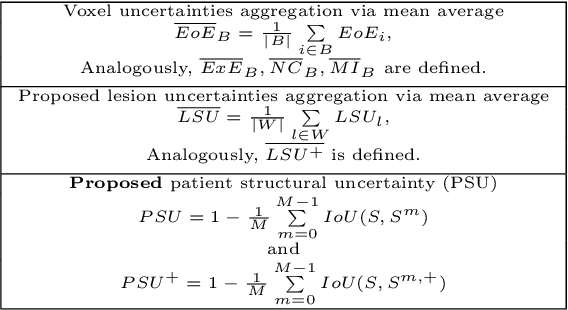
Abstract:This paper explores uncertainty quantification (UQ) as an indicator of the trustworthiness of automated deep-learning (DL) tools in the context of white matter lesion (WML) segmentation from magnetic resonance imaging (MRI) scans of multiple sclerosis (MS) patients. Our study focuses on two principal aspects of uncertainty in structured output segmentation tasks. Firstly, we postulate that a good uncertainty measure should indicate predictions likely to be incorrect with high uncertainty values. Second, we investigate the merit of quantifying uncertainty at different anatomical scales (voxel, lesion, or patient). We hypothesize that uncertainty at each scale is related to specific types of errors. Our study aims to confirm this relationship by conducting separate analyses for in-domain and out-of-domain settings. Our primary methodological contributions are (i) the development of novel measures for quantifying uncertainty at lesion and patient scales, derived from structural prediction discrepancies, and (ii) the extension of an error retention curve analysis framework to facilitate the evaluation of UQ performance at both lesion and patient scales. The results from a multi-centric MRI dataset of 172 patients demonstrate that our proposed measures more effectively capture model errors at the lesion and patient scales compared to measures that average voxel-scale uncertainty values. We provide the UQ protocols code at https://github.com/Medical-Image-Analysis-Laboratory/MS_WML_uncs.
Biomedical image analysis competitions: The state of current participation practice
Dec 16, 2022Abstract:The number of international benchmarking competitions is steadily increasing in various fields of machine learning (ML) research and practice. So far, however, little is known about the common practice as well as bottlenecks faced by the community in tackling the research questions posed. To shed light on the status quo of algorithm development in the specific field of biomedical imaging analysis, we designed an international survey that was issued to all participants of challenges conducted in conjunction with the IEEE ISBI 2021 and MICCAI 2021 conferences (80 competitions in total). The survey covered participants' expertise and working environments, their chosen strategies, as well as algorithm characteristics. A median of 72% challenge participants took part in the survey. According to our results, knowledge exchange was the primary incentive (70%) for participation, while the reception of prize money played only a minor role (16%). While a median of 80 working hours was spent on method development, a large portion of participants stated that they did not have enough time for method development (32%). 25% perceived the infrastructure to be a bottleneck. Overall, 94% of all solutions were deep learning-based. Of these, 84% were based on standard architectures. 43% of the respondents reported that the data samples (e.g., images) were too large to be processed at once. This was most commonly addressed by patch-based training (69%), downsampling (37%), and solving 3D analysis tasks as a series of 2D tasks. K-fold cross-validation on the training set was performed by only 37% of the participants and only 50% of the participants performed ensembling based on multiple identical models (61%) or heterogeneous models (39%). 48% of the respondents applied postprocessing steps.
Novel structural-scale uncertainty measures and error retention curves: application to multiple sclerosis
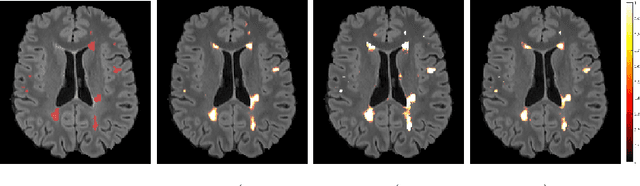
Nov 11, 2022Abstract:This paper focuses on the uncertainty estimation for white matter lesions (WML) segmentation in magnetic resonance imaging (MRI). On one side, voxel-scale segmentation errors cause the erroneous delineation of the lesions; on the other side, lesion-scale detection errors lead to wrong lesion counts. Both of these factors are clinically relevant for the assessment of multiple sclerosis patients. This work aims to compare the ability of different voxel- and lesion-scale uncertainty measures to capture errors related to segmentation and lesion detection, respectively. Our main contributions are (i) proposing new measures of lesion-scale uncertainty that do not utilise voxel-scale uncertainties; (ii) extending an error retention curves analysis framework for evaluation of lesion-scale uncertainty measures. Our results obtained on the multi-center testing set of 58 patients demonstrate that the proposed lesion-scale measure achieves the best performance among the analysed measures. All code implementations are provided at https://github.com/NataliiaMolch/MS_WML_uncs
Shifts 2.0: Extending The Dataset of Real Distributional Shifts
Jun 30, 2022



Abstract:Distributional shift, or the mismatch between training and deployment data, is a significant obstacle to the usage of machine learning in high-stakes industrial applications, such as autonomous driving and medicine. This creates a need to be able to assess how robustly ML models generalize as well as the quality of their uncertainty estimates. Standard ML baseline datasets do not allow these properties to be assessed, as the training, validation and test data are often identically distributed. Recently, a range of dedicated benchmarks have appeared, featuring both distributionally matched and shifted data. Among these benchmarks, the Shifts dataset stands out in terms of the diversity of tasks as well as the data modalities it features. While most of the benchmarks are heavily dominated by 2D image classification tasks, Shifts contains tabular weather forecasting, machine translation, and vehicle motion prediction tasks. This enables the robustness properties of models to be assessed on a diverse set of industrial-scale tasks and either universal or directly applicable task-specific conclusions to be reached. In this paper, we extend the Shifts Dataset with two datasets sourced from industrial, high-risk applications of high societal importance. Specifically, we consider the tasks of segmentation of white matter Multiple Sclerosis lesions in 3D magnetic resonance brain images and the estimation of power consumption in marine cargo vessels. Both tasks feature ubiquitous distributional shifts and a strict safety requirement due to the high cost of errors. These new datasets will allow researchers to further explore robust generalization and uncertainty estimation in new situations. In this work, we provide a description of the dataset and baseline results for both tasks.
Cortical lesions, central vein sign, and paramagnetic rim lesions in multiple sclerosis: emerging machine learning techniques and future avenues
Jan 19, 2022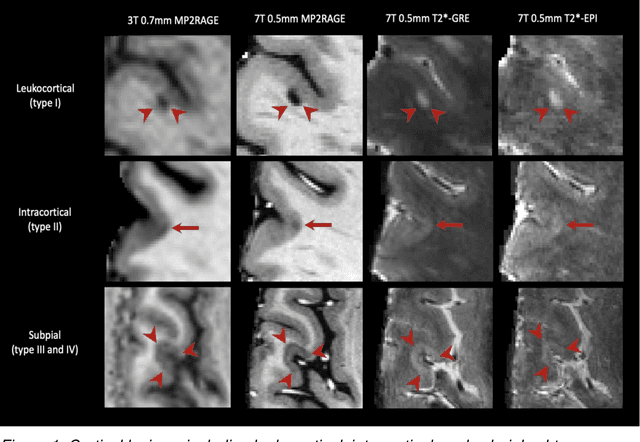
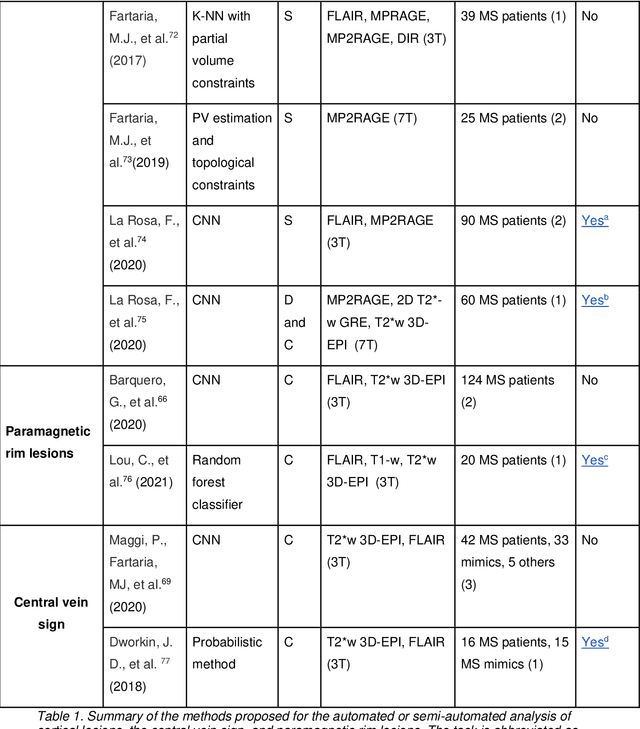
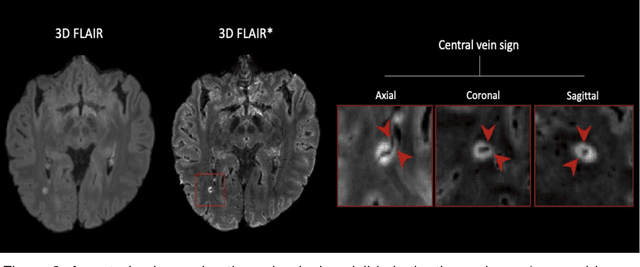
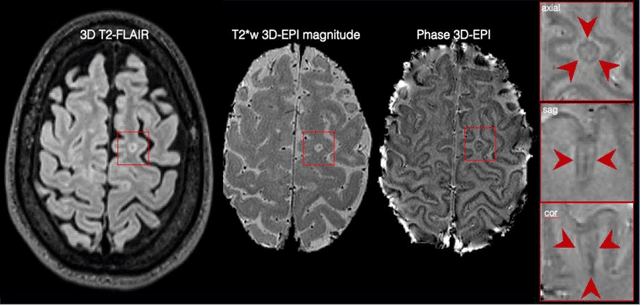
Abstract:The current multiple sclerosis (MS) diagnostic criteria lack specificity, and this may lead to misdiagnosis, which remains an issue in present-day clinical practice. In addition, conventional biomarkers only moderately correlate with MS disease progression. Recently, advanced MS lesional imaging biomarkers such as cortical lesions (CL), the central vein sign (CVS), and paramagnetic rim lesions (PRL), visible in specialized magnetic resonance imaging (MRI) sequences, have shown higher specificity in differential diagnosis. Moreover, studies have shown that CL and PRL are potential prognostic biomarkers, the former correlating with cognitive impairments and the latter with early disability progression. As machine learning-based methods have achieved extraordinary performance in the assessment of conventional imaging biomarkers, such as white matter lesion segmentation, several automated or semi-automated methods have been proposed for CL, CVS, and PRL as well. In the present review, we first introduce these advanced MS imaging biomarkers and their imaging methods. Subsequently, we describe the corresponding machine learning-based methods that were used to tackle these clinical questions, putting them into context with respect to the challenges they are still facing, including non-standardized MRI protocols, limited datasets, and moderate inter-rater variability. We conclude by presenting the current limitations that prevent their broader deployment and suggesting future research directions.
Automated Detection of Cortical Lesions in Multiple Sclerosis Patients with 7T MRI
Aug 15, 2020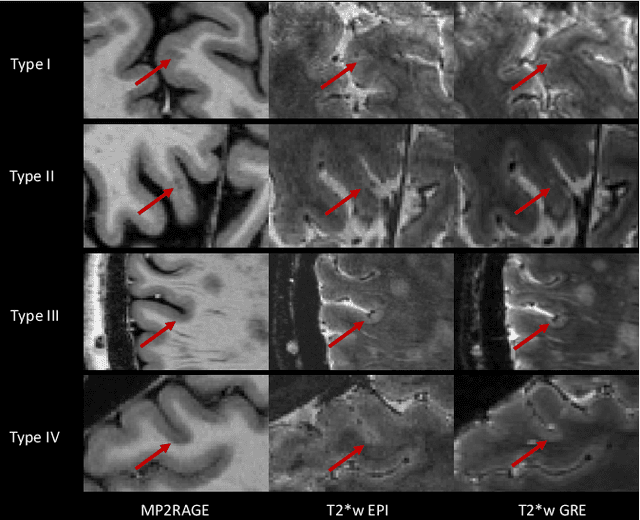
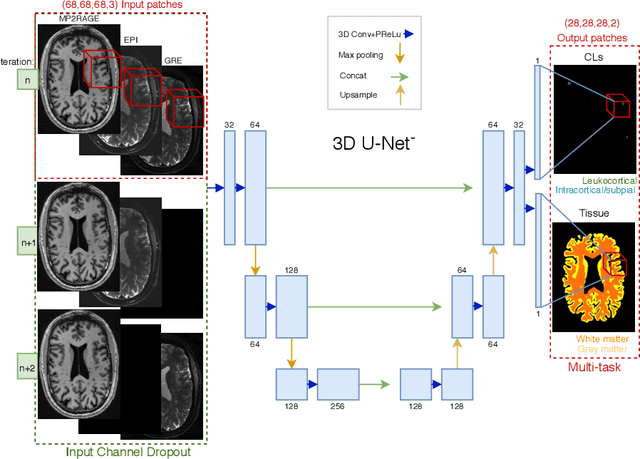
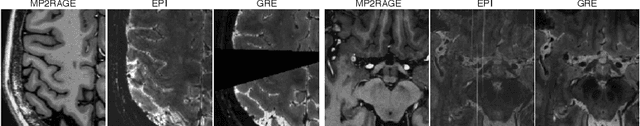
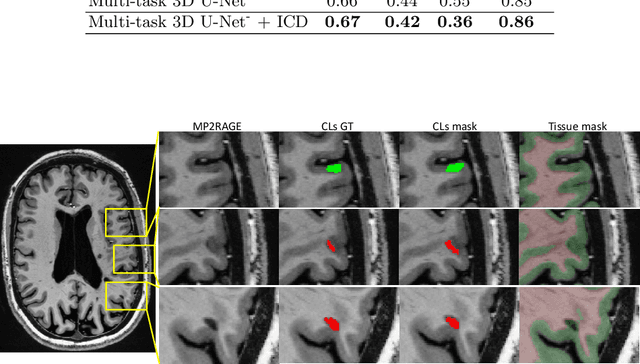
Abstract:The automated detection of cortical lesions (CLs) in patients with multiple sclerosis (MS) is a challenging task that, despite its clinical relevance, has received very little attention. Accurate detection of the small and scarce lesions requires specialized sequences and high or ultra-high field MRI. For supervised training based on multimodal structural MRI at 7T, two experts generated ground truth segmentation masks of 60 patients with 2014 CLs. We implemented a simplified 3D U-Net with three resolution levels (3D U-Net-). By increasing the complexity of the task (adding brain tissue segmentation), while randomly dropping input channels during training, we improved the performance compared to the baseline. Considering a minimum lesion size of 0.75 {\mu}L, we achieved a lesion-wise cortical lesion detection rate of 67% and a false positive rate of 42%. However, 393 (24%) of the lesions reported as false positives were post-hoc confirmed as potential or definite lesions by an expert. This indicates the potential of the proposed method to support experts in the tedious process of CL manual segmentation.
Shallow vs deep learning architectures for white matter lesion segmentation in the early stages of multiple sclerosis
Sep 10, 2018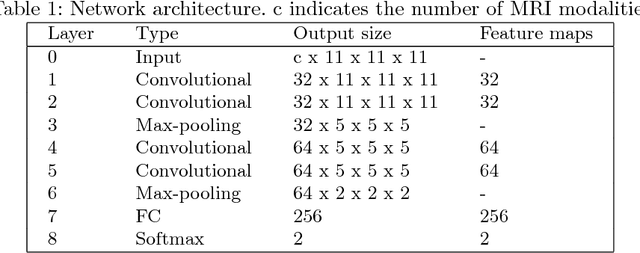
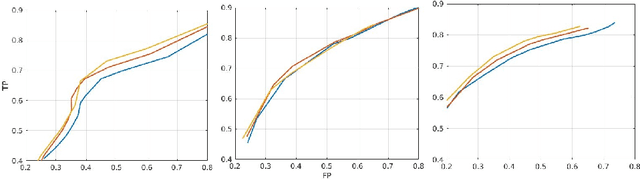
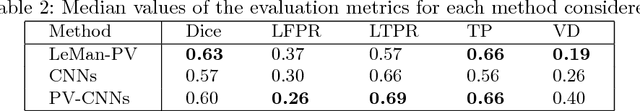
Abstract:In this work, we present a comparison of a shallow and a deep learning architecture for the automated segmentation of white matter lesions in MR images of multiple sclerosis patients. In particular, we train and test both methods on early stage disease patients, to verify their performance in challenging conditions, more similar to a clinical setting than what is typically provided in multiple sclerosis segmentation challenges. Furthermore, we evaluate a prototype naive combination of the two methods, which refines the final segmentation. All methods were trained on 32 patients, and the evaluation was performed on a pure test set of 73 cases. Results show low lesion-wise false positives (30%) for the deep learning architecture, whereas the shallow architecture yields the best Dice coefficient (63%) and volume difference (19%). Combining both shallow and deep architectures further improves the lesion-wise metrics (69% and 26% lesion-wise true and false positive rate, respectively).
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge