Lidia Al-Zogbi
From Monocular Vision to Autonomous Action: Guiding Tumor Resection via 3D Reconstruction
Mar 20, 2025Abstract:Surgical automation requires precise guidance and understanding of the scene. Current methods in the literature rely on bulky depth cameras to create maps of the anatomy, however this does not translate well to space-limited clinical applications. Monocular cameras are small and allow minimally invasive surgeries in tight spaces but additional processing is required to generate 3D scene understanding. We propose a 3D mapping pipeline that uses only RGB images to create segmented point clouds of the target anatomy. To ensure the most precise reconstruction, we compare different structure from motion algorithms' performance on mapping the central airway obstructions, and test the pipeline on a downstream task of tumor resection. In several metrics, including post-procedure tissue model evaluation, our pipeline performs comparably to RGB-D cameras and, in some cases, even surpasses their performance. These promising results demonstrate that automation guidance can be achieved in minimally invasive procedures with monocular cameras. This study is a step toward the complete autonomy of surgical robots.
Sensorless Remote Center of Motion Misalignment Estimation
Mar 17, 2025

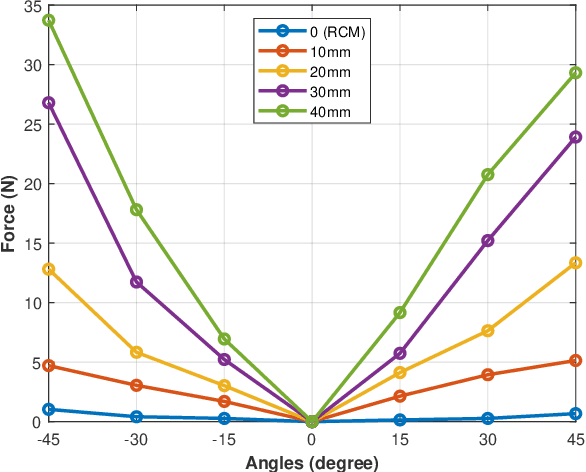

Abstract:Laparoscopic surgery constrains instrument motion around a fixed pivot point at the incision into a patient to minimize tissue trauma. Surgical robots achieve this through either hardware to software-based remote center of motion (RCM) constraints. However, accurate RCM alignment is difficult due to manual trocar placement, patient motion, and tissue deformation. Misalignment between the robot's RCM point and the patient incision site can cause unsafe forces at the incision site. This paper presents a sensorless force estimation-based framework for dynamically assessing and optimizing RCM misalignment in robotic surgery. Our experiments demonstrate that misalignment exceeding 20 mm can generate large enough forces to potentially damage tissue, emphasizing the need for precise RCM positioning. For misalignment $D\geq $ 20 mm, our optimization algorithm estimates the RCM offset with an absolute error within 5 mm. Accurate RCM misalignment estimation is a step toward automated RCM misalignment compensation, enhancing safety and reducing tissue damage in robotic-assisted laparoscopic surgery.
Robotic Ultrasound-Guided Femoral Artery Reconstruction of Anatomically-Representative Phantoms
Mar 09, 2025Abstract:Femoral artery access is essential for numerous clinical procedures, including diagnostic angiography, therapeutic catheterization, and emergency interventions. Despite its critical role, successful vascular access remains challenging due to anatomical variability, overlying adipose tissue, and the need for precise ultrasound (US) guidance. Errors in needle placement can lead to severe complications, restricting the procedure to highly skilled clinicians in controlled hospital settings. While robotic systems have shown promise in addressing these challenges through autonomous scanning and vessel reconstruction, clinical translation remains limited due to reliance on simplified phantom models that fail to capture human anatomical complexity. In this work, we present a method for autonomous robotic US scanning of bifurcated femoral arteries, and validate it on five vascular phantoms created from real patient computed tomography (CT) data. Additionally, we introduce a video-based deep learning US segmentation network tailored for vascular imaging, enabling improved 3D arterial reconstruction. The proposed network achieves a Dice score of 89.21% and an Intersection over Union of 80.54% on a newly developed vascular dataset. The quality of the reconstructed artery centerline is evaluated against ground truth CT data, demonstrating an average L2 deviation of 0.91+/-0.70 mm, with an average Hausdorff distance of 4.36+/-1.11mm. This study is the first to validate an autonomous robotic system for US scanning of the femoral artery on a diverse set of patient-specific phantoms, introducing a more advanced framework for evaluating robotic performance in vascular imaging and intervention.
Shape Manipulation of Bevel-Tip Needles for Prostate Biopsy Procedures: A Comparison of Two Resolved-Rate Controllers
Feb 05, 2024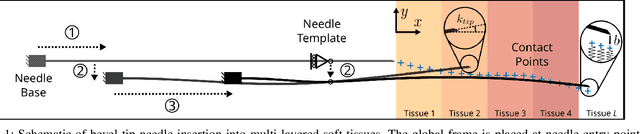
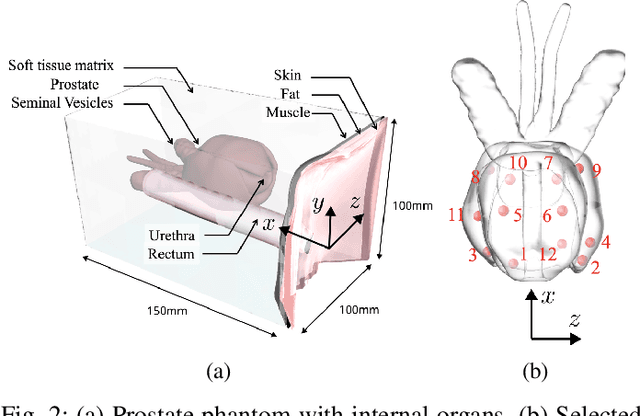
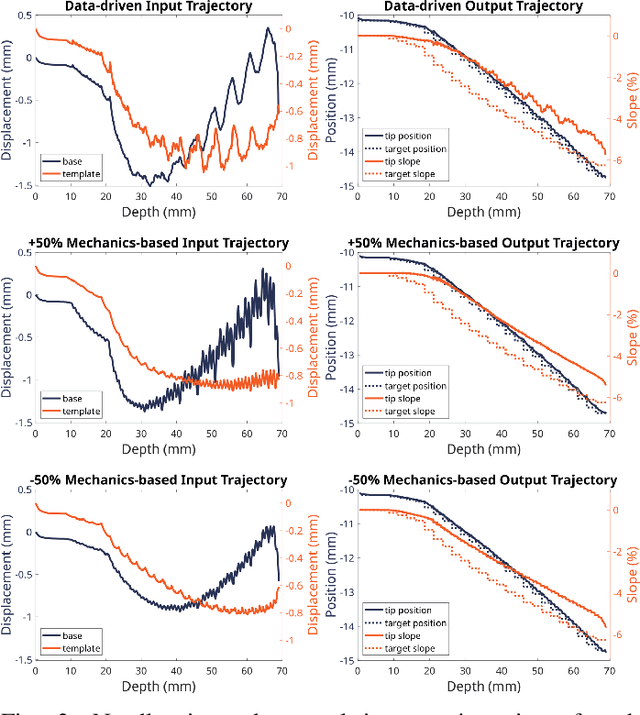

Abstract:Prostate cancer diagnosis continues to encounter challenges, often due to imprecise needle placement in standard biopsies. Several control strategies have been developed to compensate for needle tip prediction inaccuracies, however none were compared against each other, and it is unclear whether any of them can be safely and universally applied in clinical settings. This paper compares the performance of two resolved-rate controllers, derived from a mechanics-based and a data-driven approach, for bevel-tip needle control using needle shape manipulation through a template. We demonstrate for a simulated 12-core biopsy procedure under model parameter uncertainty that the mechanics-based controller can better reach desired targets when only the final goal configuration is presented even with uncertainty on model parameters estimation, and that providing a feasible needle path is crucial in ensuring safe surgical outcomes when either controller is used for needle shape manipulation.
Bevel-Tip Needle Deflection Modeling, Simulation, and Validation in Multi-Layer Tissues
Nov 29, 2023


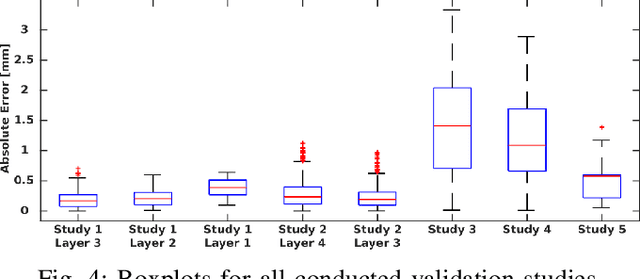
Abstract:Percutaneous needle insertions are commonly performed for diagnostic and therapeutic purposes as an effective alternative to more invasive surgical procedures. However, the outcome of needle-based approaches relies heavily on the accuracy of needle placement, which remains a challenge even with robot assistance and medical imaging guidance due to needle deflection caused by contact with soft tissues. In this paper, we present a novel mechanics-based 2D bevel-tip needle model that can account for the effect of nonlinear strain-dependent behavior of biological soft tissues under compression. Real-time finite element simulation allows multiple control inputs along the length of the needle with full three-degree-of-freedom (DOF) planar needle motions. Cross-validation studies using custom-designed multi-layer tissue phantoms as well as heterogeneous chicken breast tissues result in less than 1mm in-plane errors for insertions reaching depths of up to 61 mm, demonstrating the validity and generalizability of the proposed method.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge