Danail Stoyanov
*: shared first/last authors
TemporalDoRA: Temporal PEFT for Robust Surgical Video Question Answering
Mar 10, 2026Abstract:Surgical Video Question Answering (VideoQA) requires accurate temporal grounding while remaining robust to natural variation in how clinicians phrase questions, where linguistic bias can arise. Standard Parameter Efficient Fine Tuning (PEFT) methods adapt pretrained projections without explicitly modeling frame-to-frame interactions within the adaptation pathway, limiting their ability to exploit sparse temporal evidence. We introduce TemporalDoRA, a video-specific PEFT formulation that extends Weight-Decomposed Low-Rank Adaptation by (i) inserting lightweight temporal Multi-Head Attention (MHA) inside the low-rank bottleneck of the vision encoder and (ii) selectively applying weight decomposition only to the trainable low-rank branch rather than the full adapted weight. This design enables temporally-aware updates while preserving a frozen backbone and stable scaling. By mixing information across frames within the adaptation subspace, TemporalDoRA steers updates toward temporally consistent visual cues and improves robustness with minimal parameter overhead. To benchmark this setting, we present REAL-Colon-VQA, a colonoscopy VideoQA dataset with 6,424 clip--question pairs, including paired rephrased Out-of-Template questions to evaluate sensitivity to linguistic variation. TemporalDoRA improves Out-of-Template performance, and ablation studies confirm that temporal mixing inside the low-rank branch is the primary driver of these gains. We also validate on EndoVis18-VQA adapted to short clips and observe consistent improvements on the Out-of-Template split. Code and dataset available at~\href{https://anonymous.4open.science/r/TemporalDoRA-BFC8/}{Anonymous GitHub}.
Training-free Temporal Object Tracking in Surgical Videos
Mar 08, 2026Abstract:Purpose: In this paper, we present a novel approach for online object tracking in laparoscopic cholecystectomy (LC) surgical videos, targeting localisation and tracking of critical anatomical structures and instruments. Our method addresses the challenges of costly pixel-level annotations and label inconsistencies inherent in existing datasets. Methods: Leveraging the inherent object localisation capabilities of pre-trained text-to-image diffusion models, we extract representative features from surgical frames without any training or fine-tuning. Our tracking framework uses these features, along with cross-frame interactions via an affinity matrix inspired by query-key-value attention, to ensure temporal continuity in the tracking process. Results: Through a pilot study, we first demonstrate that diffusion features exhibit superior object localisation and consistent semantics across different decoder levels and temporal frames. Later, we perform extensive experiments to validate the effectiveness of our approach, showcasing its superiority over competitors for the task of temporal object tracking. Specifically, we achieve a per-pixel classification accuracy of 79.19%, mean Jaccard Score of 56.20%, and mean F-Score of 79.48% on the publicly available CholeSeg8K dataset. Conclusion: Our work not only introduces a novel application of text-to-image diffusion models but also contributes to advancing the field of surgical video analysis, offering a promising avenue for accurate and cost-effective temporal object tracking in minimally invasive surgery videos.
* Accepted in IPCAI 2025
Confidence-aware Monocular Depth Estimation for Minimally Invasive Surgery
Mar 03, 2026Abstract:Purpose: Monocular depth estimation (MDE) is vital for scene understanding in minimally invasive surgery (MIS). However, endoscopic video sequences are often contaminated by smoke, specular reflections, blur, and occlusions, limiting the accuracy of MDE models. In addition, current MDE models do not output depth confidence, which could be a valuable tool for improving their clinical reliability. Methods: We propose a novel confidence-aware MDE framework featuring three significant contributions: (i) Calibrated confidence targets: an ensemble of fine-tuned stereo matching models is used to capture disparity variance into pixel-wise confidence probabilities; (ii) Confidence-aware loss: Baseline MDE models are optimized with confidence-aware loss functions, utilizing pixel-wise confidence probabilities such that reliable pixels dominate training; and (iii) Inference-time confidence: a confidence estimation head is proposed with two convolution layers to predict per-pixel confidence at inference, enabling assessment of depth reliability. Results: Comprehensive experimental validation across internal and public datasets demonstrates that our framework improves depth estimation accuracy and can robustly quantify the prediction's confidence. On the internal clinical endoscopic dataset (StereoKP), we improve dense depth estimation accuracy by ~8% as compared to the baseline model. Conclusion: Our confidence-aware framework enables improved accuracy of MDE models in MIS, addressing challenges posed by noise and artifacts in pre-clinical and clinical data, and allows MDE models to provide confidence maps that may be used to improve their reliability for clinical applications.
FoundationPose-Initialized 3D-2D Liver Registration for Surgical Augmented Reality
Feb 19, 2026Abstract:Augmented reality can improve tumor localization in laparoscopic liver surgery. Existing registration pipelines typically depend on organ contours; deformable (non-rigid) alignment is often handled with finite-element (FE) models coupled to dimensionality-reduction or machine-learning components. We integrate laparoscopic depth maps with a foundation pose estimator for camera-liver pose estimation and replace FE-based deformation with non-rigid iterative closest point (NICP) to lower engineering/modeling complexity and expertise requirements. On real patient data, the depth-augmented foundation pose approach achieved 9.91 mm mean registration error in 3 cases. Combined rigid-NICP registration outperformed rigid-only registration, demonstrating NICP as an efficient substitute for finite-element deformable models. This pipeline achieves clinically relevant accuracy while offering a lightweight, engineering-friendly alternative to FE-based deformation.
A multi-centre, multi-device benchmark dataset for landmark-based comprehensive fetal biometry
Dec 19, 2025Abstract:Accurate fetal growth assessment from ultrasound (US) relies on precise biometry measured by manually identifying anatomical landmarks in standard planes. Manual landmarking is time-consuming, operator-dependent, and sensitive to variability across scanners and sites, limiting the reproducibility of automated approaches. There is a need for multi-source annotated datasets to develop artificial intelligence-assisted fetal growth assessment methods. To address this bottleneck, we present an open, multi-centre, multi-device benchmark dataset of fetal US images with expert anatomical landmark annotations for clinically used fetal biometric measurements. These measurements include head bi-parietal and occipito-frontal diameters, abdominal transverse and antero-posterior diameters, and femoral length. The dataset comprises 4,513 de-identified US images from 1,904 subjects acquired at three clinical sites using seven different US devices. We provide standardised, subject-disjoint train/test splits, evaluation code, and baseline results to enable fair and reproducible comparison of methods. Using an automatic biometry model, we quantify domain shift and demonstrate that training and evaluation confined to a single centre substantially overestimate performance relative to multi-centre testing. To the best of our knowledge, this is the first publicly available multi-centre, multi-device, landmark-annotated dataset that covers all primary fetal biometry measures, providing a robust benchmark for domain adaptation and multi-centre generalisation in fetal biometry and enabling more reliable AI-assisted fetal growth assessment across centres. All data, annotations, training code, and evaluation pipelines are made publicly available.
Learning from Single Timestamps: Complexity Estimation in Laparoscopic Cholecystectomy
Nov 06, 2025Abstract:Purpose: Accurate assessment of surgical complexity is essential in Laparoscopic Cholecystectomy (LC), where severe inflammation is associated with longer operative times and increased risk of postoperative complications. The Parkland Grading Scale (PGS) provides a clinically validated framework for stratifying inflammation severity; however, its automation in surgical videos remains largely unexplored, particularly in realistic scenarios where complete videos must be analyzed without prior manual curation. Methods: In this work, we introduce STC-Net, a novel framework for SingleTimestamp-based Complexity estimation in LC via the PGS, designed to operate under weak temporal supervision. Unlike prior methods limited to static images or manually trimmed clips, STC-Net operates directly on full videos. It jointly performs temporal localization and grading through a localization, window proposal, and grading module. We introduce a novel loss formulation combining hard and soft localization objectives and background-aware grading supervision. Results: Evaluated on a private dataset of 1,859 LC videos, STC-Net achieves an accuracy of 62.11% and an F1-score of 61.42%, outperforming non-localized baselines by over 10% in both metrics and highlighting the effectiveness of weak supervision for surgical complexity assessment. Conclusion: STC-Net demonstrates a scalable and effective approach for automated PGS-based surgical complexity estimation from full LC videos, making it promising for post-operative analysis and surgical training.
Federated Learning for Surgical Vision in Appendicitis Classification: Results of the FedSurg EndoVis 2024 Challenge
Oct 06, 2025Abstract:Purpose: The FedSurg challenge was designed to benchmark the state of the art in federated learning for surgical video classification. Its goal was to assess how well current methods generalize to unseen clinical centers and adapt through local fine-tuning while enabling collaborative model development without sharing patient data. Methods: Participants developed strategies to classify inflammation stages in appendicitis using a preliminary version of the multi-center Appendix300 video dataset. The challenge evaluated two tasks: generalization to an unseen center and center-specific adaptation after fine-tuning. Submitted approaches included foundation models with linear probing, metric learning with triplet loss, and various FL aggregation schemes (FedAvg, FedMedian, FedSAM). Performance was assessed using F1-score and Expected Cost, with ranking robustness evaluated via bootstrapping and statistical testing. Results: In the generalization task, performance across centers was limited. In the adaptation task, all teams improved after fine-tuning, though ranking stability was low. The ViViT-based submission achieved the strongest overall performance. The challenge highlighted limitations in generalization, sensitivity to class imbalance, and difficulties in hyperparameter tuning in decentralized training, while spatiotemporal modeling and context-aware preprocessing emerged as promising strategies. Conclusion: The FedSurg Challenge establishes the first benchmark for evaluating FL strategies in surgical video classification. Findings highlight the trade-off between local personalization and global robustness, and underscore the importance of architecture choice, preprocessing, and loss design. This benchmarking offers a reference point for future development of imbalance-aware, adaptive, and robust FL methods in clinical surgical AI.
Exploring Pre-training Across Domains for Few-Shot Surgical Skill Assessment
Sep 11, 2025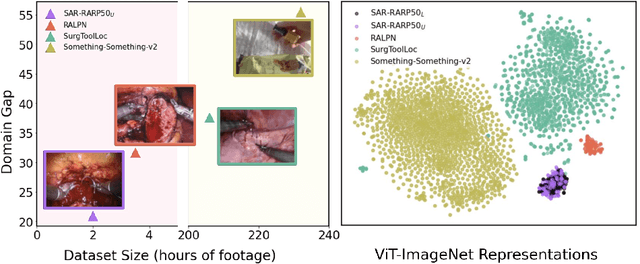

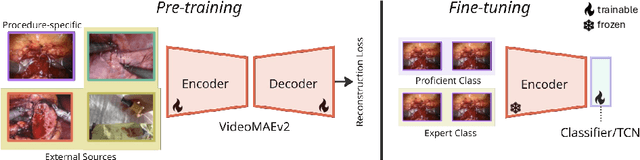
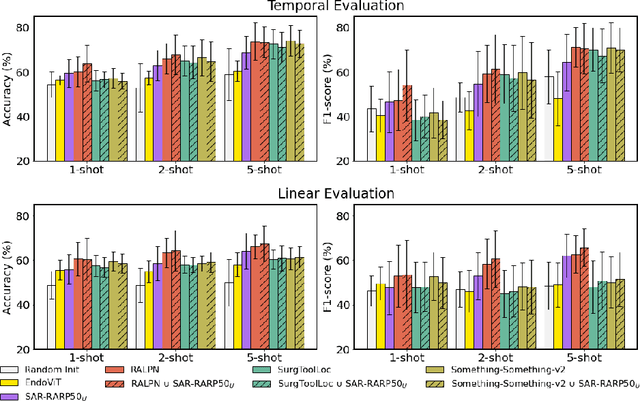
Abstract:Automated surgical skill assessment (SSA) is a central task in surgical computer vision. Developing robust SSA models is challenging due to the scarcity of skill annotations, which are time-consuming to produce and require expert consensus. Few-shot learning (FSL) offers a scalable alternative enabling model development with minimal supervision, though its success critically depends on effective pre-training. While widely studied for several surgical downstream tasks, pre-training has remained largely unexplored in SSA. In this work, we formulate SSA as a few-shot task and investigate how self-supervised pre-training strategies affect downstream few-shot SSA performance. We annotate a publicly available robotic surgery dataset with Objective Structured Assessment of Technical Skill (OSATS) scores, and evaluate various pre-training sources across three few-shot settings. We quantify domain similarity and analyze how domain gap and the inclusion of procedure-specific data into pre-training influence transferability. Our results show that small but domain-relevant datasets can outperform large scale, less aligned ones, achieving accuracies of 60.16%, 66.03%, and 73.65% in the 1-, 2-, and 5-shot settings, respectively. Moreover, incorporating procedure-specific data into pre-training with a domain-relevant external dataset significantly boosts downstream performance, with an average gain of +1.22% in accuracy and +2.28% in F1-score; however, applying the same strategy with less similar but large-scale sources can instead lead to performance degradation. Code and models are available at https://github.com/anastadimi/ssa-fsl.
Comparative validation of surgical phase recognition, instrument keypoint estimation, and instrument instance segmentation in endoscopy: Results of the PhaKIR 2024 challenge
Jul 22, 2025Abstract:Reliable recognition and localization of surgical instruments in endoscopic video recordings are foundational for a wide range of applications in computer- and robot-assisted minimally invasive surgery (RAMIS), including surgical training, skill assessment, and autonomous assistance. However, robust performance under real-world conditions remains a significant challenge. Incorporating surgical context - such as the current procedural phase - has emerged as a promising strategy to improve robustness and interpretability. To address these challenges, we organized the Surgical Procedure Phase, Keypoint, and Instrument Recognition (PhaKIR) sub-challenge as part of the Endoscopic Vision (EndoVis) challenge at MICCAI 2024. We introduced a novel, multi-center dataset comprising thirteen full-length laparoscopic cholecystectomy videos collected from three distinct medical institutions, with unified annotations for three interrelated tasks: surgical phase recognition, instrument keypoint estimation, and instrument instance segmentation. Unlike existing datasets, ours enables joint investigation of instrument localization and procedural context within the same data while supporting the integration of temporal information across entire procedures. We report results and findings in accordance with the BIAS guidelines for biomedical image analysis challenges. The PhaKIR sub-challenge advances the field by providing a unique benchmark for developing temporally aware, context-driven methods in RAMIS and offers a high-quality resource to support future research in surgical scene understanding.
StereoMamba: Real-time and Robust Intraoperative Stereo Disparity Estimation via Long-range Spatial Dependencies
Apr 24, 2025Abstract:Stereo disparity estimation is crucial for obtaining depth information in robot-assisted minimally invasive surgery (RAMIS). While current deep learning methods have made significant advancements, challenges remain in achieving an optimal balance between accuracy, robustness, and inference speed. To address these challenges, we propose the StereoMamba architecture, which is specifically designed for stereo disparity estimation in RAMIS. Our approach is based on a novel Feature Extraction Mamba (FE-Mamba) module, which enhances long-range spatial dependencies both within and across stereo images. To effectively integrate multi-scale features from FE-Mamba, we then introduce a novel Multidimensional Feature Fusion (MFF) module. Experiments against the state-of-the-art on the ex-vivo SCARED benchmark demonstrate that StereoMamba achieves superior performance on EPE of 2.64 px and depth MAE of 2.55 mm, the second-best performance on Bad2 of 41.49% and Bad3 of 26.99%, while maintaining an inference speed of 21.28 FPS for a pair of high-resolution images (1280*1024), striking the optimum balance between accuracy, robustness, and efficiency. Furthermore, by comparing synthesized right images, generated from warping left images using the generated disparity maps, with the actual right image, StereoMamba achieves the best average SSIM (0.8970) and PSNR (16.0761), exhibiting strong zero-shot generalization on the in-vivo RIS2017 and StereoMIS datasets.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge