Chinmay Prabhakar
Graph Energy Matching: Transport-Aligned Energy-Based Modeling for Graph Generation
Mar 24, 2026Abstract:Energy-based models for discrete domains, such as graphs, explicitly capture relative likelihoods, naturally enabling composable probabilistic inference tasks like conditional generation or enforcing constraints at test-time. However, discrete energy-based models typically struggle with efficient and high-quality sampling, as off-support regions often contain spurious local minima, trapping samplers and causing training instabilities. This has historically resulted in a fidelity gap relative to discrete diffusion models. We introduce Graph Energy Matching (GEM), a generative framework for graphs that closes this fidelity gap. Motivated by the transport map optimization perspective of the Jordan-Kinderlehrer-Otto (JKO) scheme, GEM learns a permutation-invariant potential energy that simultaneously provides transport-aligned guidance from noise toward data and refines samples within regions of high data likelihood. Further, we introduce a sampling protocol that leverages an energy-based switch to seamlessly bridge: (i) rapid, gradient-guided transport toward high-probability regions to (ii) a mixing regime for exploration of the learned graph distribution. On molecular graph benchmarks, GEM matches or exceeds strong discrete diffusion baselines. Beyond sample quality, explicit modeling of relative likelihood enables targeted exploration at inference time, facilitating compositional generation, property-constrained sampling, and geodesic interpolation between graphs.
VesselTok: Tokenizing Vessel-like 3D Biomedical Graph Representations for Reconstruction and Generation
Mar 19, 2026Abstract:Spatial graphs provide a lightweight and elegant representation of curvilinear anatomical structures such as blood vessels, lung airways, and neuronal networks. Accurately modeling these graphs is crucial in clinical and (bio-)medical research. However, the high spatial resolution of large networks drastically increases their complexity, resulting in significant computational challenges. In this work, we aim to tackle these challenges by proposing VesselTok, a framework that approaches spatially dense graphs from a parametric shape perspective to learn latent representations (tokens). VesselTok leverages centerline points with a pseudo radius to effectively encode tubular geometry. Specifically, we learn a novel latent representation conditioned on centerline points to encode neural implicit representations of vessel-like, tubular structures. We demonstrate VesselTok's performance across diverse anatomies, including lung airways, lung vessels, and brain vessels, highlighting its ability to robustly encode complex topologies. To prove the effectiveness of VesselTok's learnt latent representations, we show that they (i) generalize to unseen anatomies, (ii) support generative modeling of plausible anatomical graphs, and (iii) transfer effectively to downstream inverse problems, such as link prediction.
VariViT: A Vision Transformer for Variable Image Sizes
Feb 16, 2026Abstract:Vision Transformers (ViTs) have emerged as the state-of-the-art architecture in representation learning, leveraging self-attention mechanisms to excel in various tasks. ViTs split images into fixed-size patches, constraining them to a predefined size and necessitating pre-processing steps like resizing, padding, or cropping. This poses challenges in medical imaging, particularly with irregularly shaped structures like tumors. A fixed bounding box crop size produces input images with highly variable foreground-to-background ratios. Resizing medical images can degrade information and introduce artefacts, impacting diagnosis. Hence, tailoring variable-sized crops to regions of interest can enhance feature representation capabilities. Moreover, large images are computationally expensive, and smaller sizes risk information loss, presenting a computation-accuracy tradeoff. We propose VariViT, an improved ViT model crafted to handle variable image sizes while maintaining a consistent patch size. VariViT employs a novel positional embedding resizing scheme for a variable number of patches. We also implement a new batching strategy within VariViT to reduce computational complexity, resulting in faster training and inference times. In our evaluations on two 3D brain MRI datasets, VariViT surpasses vanilla ViTs and ResNet in glioma genotype prediction and brain tumor classification. It achieves F1-scores of 75.5% and 76.3%, respectively, learning more discriminative features. Our proposed batching strategy reduces computation time by up to 30% compared to conventional architectures. These findings underscore the efficacy of VariViT in image representation learning. Our code can be found here: https://github.com/Aswathi-Varma/varivit
ProGiDiff: Prompt-Guided Diffusion-Based Medical Image Segmentation
Jan 22, 2026Abstract:Widely adopted medical image segmentation methods, although efficient, are primarily deterministic and remain poorly amenable to natural language prompts. Thus, they lack the capability to estimate multiple proposals, human interaction, and cross-modality adaptation. Recently, text-to-image diffusion models have shown potential to bridge the gap. However, training them from scratch requires a large dataset-a limitation for medical image segmentation. Furthermore, they are often limited to binary segmentation and cannot be conditioned on a natural language prompt. To this end, we propose a novel framework called ProGiDiff that leverages existing image generation models for medical image segmentation purposes. Specifically, we propose a ControlNet-style conditioning mechanism with a custom encoder, suitable for image conditioning, to steer a pre-trained diffusion model to output segmentation masks. It naturally extends to a multi-class setting simply by prompting the target organ. Our experiment on organ segmentation from CT images demonstrates strong performance compared to previous methods and could greatly benefit from an expert-in-the-loop setting to leverage multiple proposals. Importantly, we demonstrate that the learned conditioning mechanism can be easily transferred through low-rank, few-shot adaptation to segment MR images.
3D Vessel Graph Generation Using Denoising Diffusion
Jul 08, 2024



Abstract:Blood vessel networks, represented as 3D graphs, help predict disease biomarkers, simulate blood flow, and aid in synthetic image generation, relevant in both clinical and pre-clinical settings. However, generating realistic vessel graphs that correspond to an anatomy of interest is challenging. Previous methods aimed at generating vessel trees mostly in an autoregressive style and could not be applied to vessel graphs with cycles such as capillaries or specific anatomical structures such as the Circle of Willis. Addressing this gap, we introduce the first application of \textit{denoising diffusion models} in 3D vessel graph generation. Our contributions include a novel, two-stage generation method that sequentially denoises node coordinates and edges. We experiment with two real-world vessel datasets, consisting of microscopic capillaries and major cerebral vessels, and demonstrate the generalizability of our method for producing diverse, novel, and anatomically plausible vessel graphs.
Self-pruning Graph Neural Network for Predicting Inflammatory Disease Activity in Multiple Sclerosis from Brain MR Images
Aug 31, 2023



Abstract:Multiple Sclerosis (MS) is a severe neurological disease characterized by inflammatory lesions in the central nervous system. Hence, predicting inflammatory disease activity is crucial for disease assessment and treatment. However, MS lesions can occur throughout the brain and vary in shape, size and total count among patients. The high variance in lesion load and locations makes it challenging for machine learning methods to learn a globally effective representation of whole-brain MRI scans to assess and predict disease. Technically it is non-trivial to incorporate essential biomarkers such as lesion load or spatial proximity. Our work represents the first attempt to utilize graph neural networks (GNN) to aggregate these biomarkers for a novel global representation. We propose a two-stage MS inflammatory disease activity prediction approach. First, a 3D segmentation network detects lesions, and a self-supervised algorithm extracts their image features. Second, the detected lesions are used to build a patient graph. The lesions act as nodes in the graph and are initialized with image features extracted in the first stage. Finally, the lesions are connected based on their spatial proximity and the inflammatory disease activity prediction is formulated as a graph classification task. Furthermore, we propose a self-pruning strategy to auto-select the most critical lesions for prediction. Our proposed method outperforms the existing baseline by a large margin (AUCs of 0.67 vs. 0.61 and 0.66 vs. 0.60 for one-year and two-year inflammatory disease activity, respectively). Finally, our proposed method enjoys inherent explainability by assigning an importance score to each lesion for the overall prediction. Code is available at https://github.com/chinmay5/ms_ida.git
Link Prediction for Flow-Driven Spatial Networks
Mar 25, 2023


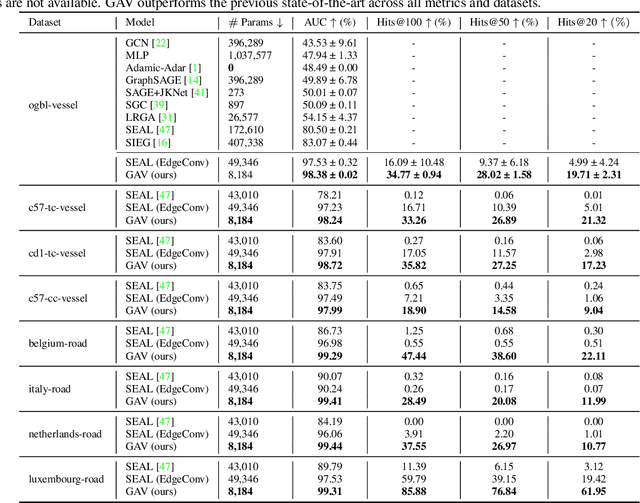
Abstract:Link prediction algorithms predict the existence of connections between nodes in network-structured data and are typically applied to refine the connectivity among nodes by proposing meaningful new links. In this work, we focus on link prediction for flow-driven spatial networks, which are embedded in a Euclidean space and relate to physical exchange and transportation processes (e.g., blood flow in vessels or traffic flow in road networks). To this end, we propose the Graph Attentive Vectors (GAV) link prediction framework. GAV models simplified dynamics of physical flow in spatial networks via an attentive, neighborhood-aware message-passing paradigm, updating vector embeddings in a constrained manner. We evaluate GAV on eight flow-driven spatial networks given by whole-brain vessel graphs and road networks. GAV demonstrates superior performances across all datasets and metrics and outperforms the current state-of-the-art on the ogbl-vessel benchmark by more than 18% (98.38 vs. 83.07 AUC).
ViT-AE++: Improving Vision Transformer Autoencoder for Self-supervised Medical Image Representations
Jan 18, 2023Abstract:Self-supervised learning has attracted increasing attention as it learns data-driven representation from data without annotations. Vision transformer-based autoencoder (ViT-AE) by He et al. (2021) is a recent self-supervised learning technique that employs a patch-masking strategy to learn a meaningful latent space. In this paper, we focus on improving ViT-AE (nicknamed ViT-AE++) for a more effective representation of both 2D and 3D medical images. We propose two new loss functions to enhance the representation during the training stage. The first loss term aims to improve self-reconstruction by considering the structured dependencies and hence indirectly improving the representation. The second loss term leverages contrastive loss to directly optimize the representation from two randomly masked views. As an independent contribution, we extended ViT-AE++ to a 3D fashion for volumetric medical images. We extensively evaluate ViT-AE++ on both natural images and medical images, demonstrating consistent improvement over vanilla ViT-AE and its superiority over other contrastive learning approaches.
A Domain-specific Perceptual Metric via Contrastive Self-supervised Representation: Applications on Natural and Medical Images
Dec 03, 2022



Abstract:Quantifying the perceptual similarity of two images is a long-standing problem in low-level computer vision. The natural image domain commonly relies on supervised learning, e.g., a pre-trained VGG, to obtain a latent representation. However, due to domain shift, pre-trained models from the natural image domain might not apply to other image domains, such as medical imaging. Notably, in medical imaging, evaluating the perceptual similarity is exclusively performed by specialists trained extensively in diverse medical fields. Thus, medical imaging remains devoid of task-specific, objective perceptual measures. This work answers the question: Is it necessary to rely on supervised learning to obtain an effective representation that could measure perceptual similarity, or is self-supervision sufficient? To understand whether recent contrastive self-supervised representation (CSR) may come to the rescue, we start with natural images and systematically evaluate CSR as a metric across numerous contemporary architectures and tasks and compare them with existing methods. We find that in the natural image domain, CSR behaves on par with the supervised one on several perceptual tests as a metric, and in the medical domain, CSR better quantifies perceptual similarity concerning the experts' ratings. We also demonstrate that CSR can significantly improve image quality in two image synthesis tasks. Finally, our extensive results suggest that perceptuality is an emergent property of CSR, which can be adapted to many image domains without requiring annotations.
Differentially Private Graph Classification with GNNs
Feb 08, 2022



Abstract:Graph Neural Networks (GNNs) have established themselves as the state-of-the-art models for many machine learning applications such as the analysis of social networks, protein interactions and molecules. Several among these datasets contain privacy-sensitive data. Machine learning with differential privacy is a promising technique to allow deriving insight from sensitive data while offering formal guarantees of privacy protection. However, the differentially private training of GNNs has so far remained under-explored due to the challenges presented by the intrinsic structural connectivity of graphs. In this work, we introduce differential privacy for graph-level classification, one of the key applications of machine learning on graphs. Our method is applicable to deep learning on multi-graph datasets and relies on differentially private stochastic gradient descent (DP-SGD). We show results on a variety of synthetic and public datasets and evaluate the impact of different GNN architectures and training hyperparameters on model performance for differentially private graph classification. Finally, we apply explainability techniques to assess whether similar representations are learned in the private and non-private settings and establish robust baselines for future work in this area.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge