Mark Mühlau
Optimizing Rank for High-Fidelity Implicit Neural Representations
Dec 16, 2025Abstract:Implicit Neural Representations (INRs) based on vanilla Multi-Layer Perceptrons (MLPs) are widely believed to be incapable of representing high-frequency content. This has directed research efforts towards architectural interventions, such as coordinate embeddings or specialized activation functions, to represent high-frequency signals. In this paper, we challenge the notion that the low-frequency bias of vanilla MLPs is an intrinsic, architectural limitation to learn high-frequency content, but instead a symptom of stable rank degradation during training. We empirically demonstrate that regulating the network's rank during training substantially improves the fidelity of the learned signal, rendering even simple MLP architectures expressive. Extensive experiments show that using optimizers like Muon, with high-rank, near-orthogonal updates, consistently enhances INR architectures even beyond simple ReLU MLPs. These substantial improvements hold across a diverse range of domains, including natural and medical images, and novel view synthesis, with up to 9 dB PSNR improvements over the previous state-of-the-art. Our project page, which includes code and experimental results, is available at: (https://muon-inrs.github.io).
Self-pruning Graph Neural Network for Predicting Inflammatory Disease Activity in Multiple Sclerosis from Brain MR Images
Aug 31, 2023



Abstract:Multiple Sclerosis (MS) is a severe neurological disease characterized by inflammatory lesions in the central nervous system. Hence, predicting inflammatory disease activity is crucial for disease assessment and treatment. However, MS lesions can occur throughout the brain and vary in shape, size and total count among patients. The high variance in lesion load and locations makes it challenging for machine learning methods to learn a globally effective representation of whole-brain MRI scans to assess and predict disease. Technically it is non-trivial to incorporate essential biomarkers such as lesion load or spatial proximity. Our work represents the first attempt to utilize graph neural networks (GNN) to aggregate these biomarkers for a novel global representation. We propose a two-stage MS inflammatory disease activity prediction approach. First, a 3D segmentation network detects lesions, and a self-supervised algorithm extracts their image features. Second, the detected lesions are used to build a patient graph. The lesions act as nodes in the graph and are initialized with image features extracted in the first stage. Finally, the lesions are connected based on their spatial proximity and the inflammatory disease activity prediction is formulated as a graph classification task. Furthermore, we propose a self-pruning strategy to auto-select the most critical lesions for prediction. Our proposed method outperforms the existing baseline by a large margin (AUCs of 0.67 vs. 0.61 and 0.66 vs. 0.60 for one-year and two-year inflammatory disease activity, respectively). Finally, our proposed method enjoys inherent explainability by assigning an importance score to each lesion for the overall prediction. Code is available at https://github.com/chinmay5/ms_ida.git
A Lightweight Causal Model for Interpretable Subject-level Prediction
Jun 19, 2023



Abstract:Recent years have seen a growing interest in methods for predicting a variable of interest, such as a subject's diagnosis, from medical images. Methods based on discriminative modeling excel at making accurate predictions, but are challenged in their ability to explain their decisions in anatomically meaningful terms. In this paper, we propose a simple technique for single-subject prediction that is inherently interpretable. It augments the generative models used in classical human brain mapping techniques, in which cause-effect relations can be encoded, with a multivariate noise model that captures dominant spatial correlations. Experiments demonstrate that the resulting model can be efficiently inverted to make accurate subject-level predictions, while at the same time offering intuitive causal explanations of its inner workings. The method is easy to use: training is fast for typical training set sizes, and only a single hyperparameter needs to be set by the user. Our code is available at https://github.com/chiara-mauri/Interpretable-subject-level-prediction.
Multi-contrast MRI Super-resolution via Implicit Neural Representations
Mar 27, 2023Abstract:Clinical routine and retrospective cohorts commonly include multi-parametric Magnetic Resonance Imaging; however, they are mostly acquired in different anisotropic 2D views due to signal-to-noise-ratio and scan-time constraints. Thus acquired views suffer from poor out-of-plane resolution and affect downstream volumetric image analysis that typically requires isotropic 3D scans. Combining different views of multi-contrast scans into high-resolution isotropic 3D scans is challenging due to the lack of a large training cohort, which calls for a subject-specific framework.This work proposes a novel solution to this problem leveraging Implicit Neural Representations (INR). Our proposed INR jointly learns two different contrasts of complementary views in a continuous spatial function and benefits from exchanging anatomical information between them. Trained within minutes on a single commodity GPU, our model provides realistic super-resolution across different pairs of contrasts in our experiments with three datasets. Using Mutual Information (MI) as a metric, we find that our model converges to an optimum MI amongst sequences, achieving anatomically faithful reconstruction. Code is available at: https://github.com/jqmcginnis/multi_contrast_inr.
An Open-Source Tool for Longitudinal Whole-Brain and White Matter Lesion Segmentation
Jul 10, 2022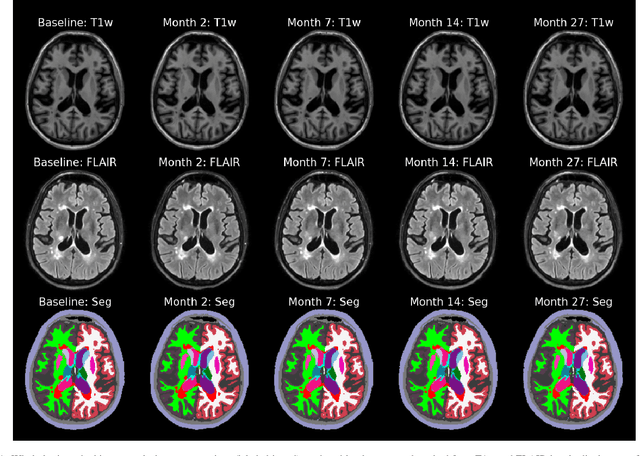
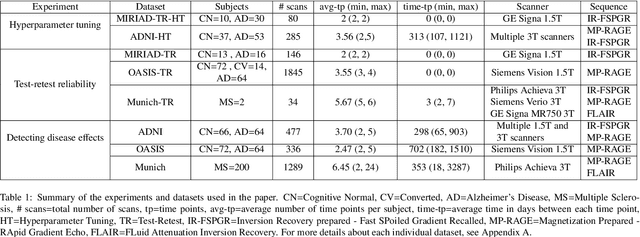
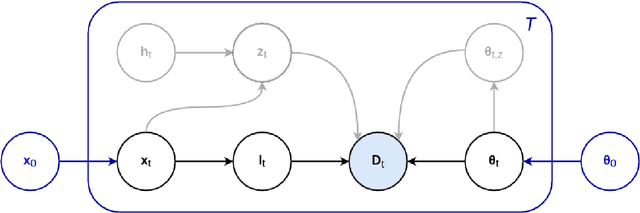
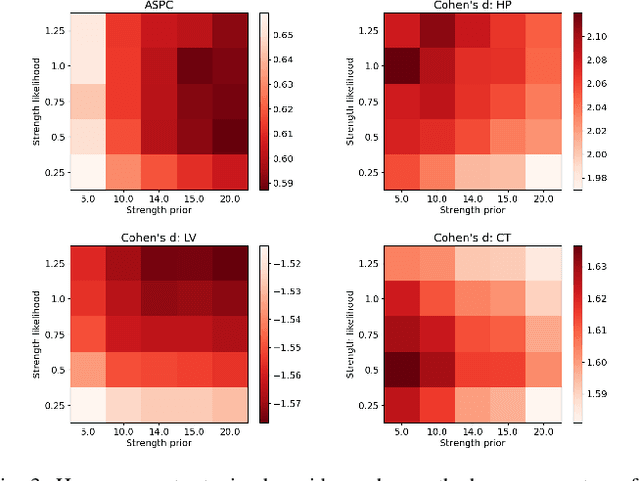
Abstract:In this paper we describe and validate a longitudinal method for whole-brain segmentation of longitudinal MRI scans. It builds upon an existing whole-brain segmentation method that can handle multi-contrast data and robustly analyze images with white matter lesions. This method is here extended with subject-specific latent variables that encourage temporal consistency between its segmentation results, enabling it to better track subtle morphological changes in dozens of neuroanatomical structures and white matter lesions. We validate the proposed method on multiple datasets of control subjects and patients suffering from Alzheimer's disease and multiple sclerosis, and compare its results against those obtained with its original cross-sectional formulation and two benchmark longitudinal methods. The results indicate that the method attains a higher test-retest reliability, while being more sensitive to longitudinal disease effect differences between patient groups. An implementation is publicly available as part of the open-source neuroimaging package FreeSurfer.
A Longitudinal Method for Simultaneous Whole-Brain and Lesion Segmentation in Multiple Sclerosis
Sep 15, 2020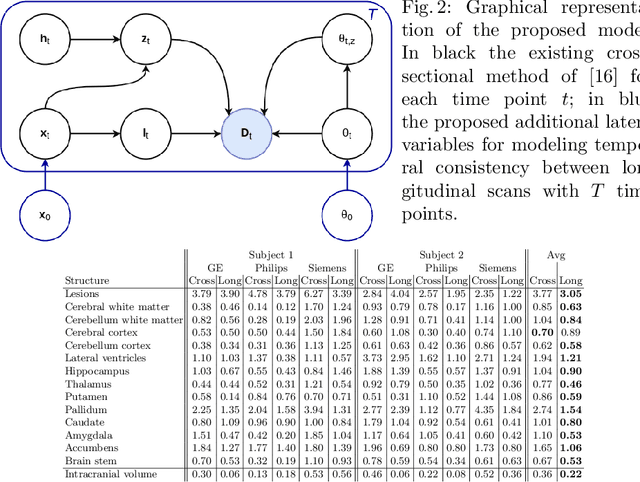
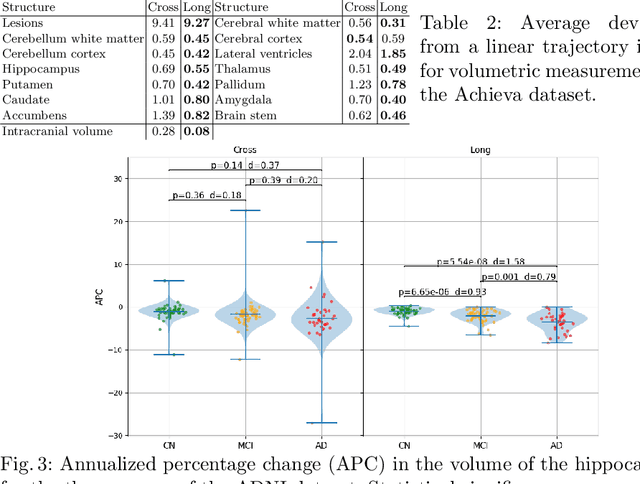
Abstract:In this paper we propose a novel method for the segmentation of longitudinal brain MRI scans of patients suffering from Multiple Sclerosis. The method builds upon an existing cross-sectional method for simultaneous whole-brain and lesion segmentation, introducing subject-specific latent variables to encourage temporal consistency between longitudinal scans. It is very generally applicable, as it does not make any prior assumptions on the scanner, the MRI protocol, or the number and timing of longitudinal follow-up scans. Preliminary experiments on three longitudinal datasets indicate that the proposed method produces more reliable segmentations and detects disease effects better than the cross-sectional method it is based upon.
A Contrast-Adaptive Method for Simultaneous Whole-Brain and Lesion Segmentation in Multiple Sclerosis
May 11, 2020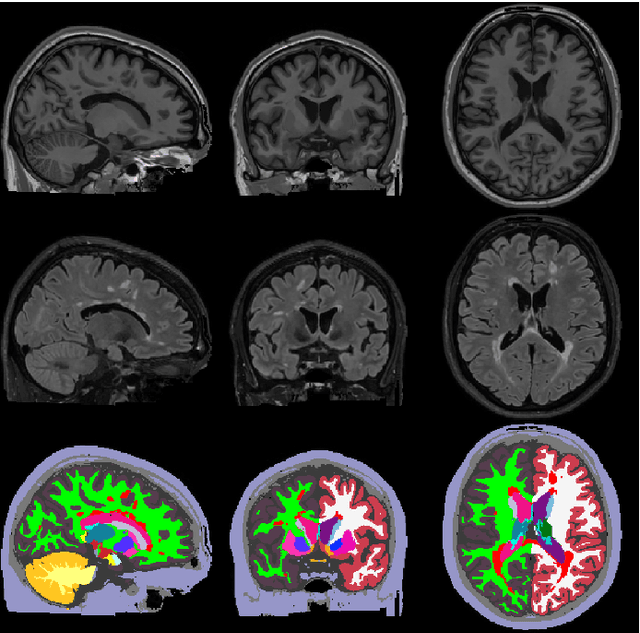
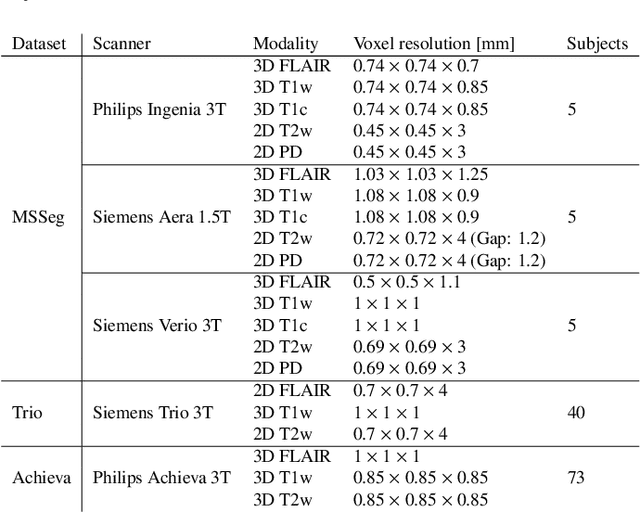
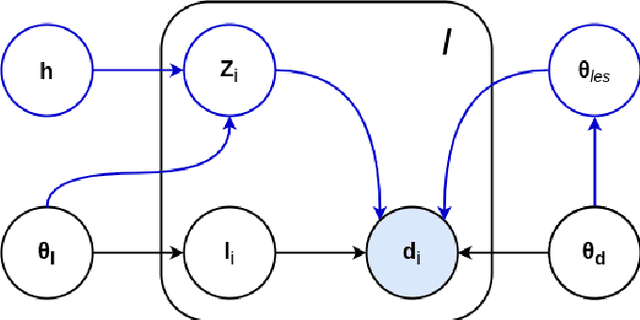
Abstract:Here we present a method for the simultaneous segmentation of white matter lesions and normal-appearing neuroanatomical structures from multi-contrast brain MRI scans of multiple sclerosis patients. The method integrates a novel model for white matter lesions into a previously validated generative model for whole-brain segmentation. By using separate models for the shape of anatomical structures and their appearance in MRI, the algorithm can adapt to data acquired with different scanners and imaging protocols without retraining. We validate the method using three disparate datasets, showing state-of-the-art performance in white matter lesion segmentation while simultaneously segmenting dozens of other brain structures. We further demonstrate that the contrast-adaptive method can also be applied robustly to MRI scans of healthy controls, and replicate previously documented atrophy patterns in deep gray matter structures in MS. The algorithm is publicly available as part of the open-source neuroimaging package FreeSurfer.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge