Jeremy Balch
MANDARIN: Mixture-of-Experts Framework for Dynamic Delirium and Coma Prediction in ICU Patients: Development and Validation of an Acute Brain Dysfunction Prediction Model
Mar 08, 2025Abstract:Acute brain dysfunction (ABD) is a common, severe ICU complication, presenting as delirium or coma and leading to prolonged stays, increased mortality, and cognitive decline. Traditional screening tools like the Glasgow Coma Scale (GCS), Confusion Assessment Method (CAM), and Richmond Agitation-Sedation Scale (RASS) rely on intermittent assessments, causing delays and inconsistencies. In this study, we propose MANDARIN (Mixture-of-Experts Framework for Dynamic Delirium and Coma Prediction in ICU Patients), a 1.5M-parameter mixture-of-experts neural network to predict ABD in real-time among ICU patients. The model integrates temporal and static data from the ICU to predict the brain status in the next 12 to 72 hours, using a multi-branch approach to account for current brain status. The MANDARIN model was trained on data from 92,734 patients (132,997 ICU admissions) from 2 hospitals between 2008-2019 and validated externally on data from 11,719 patients (14,519 ICU admissions) from 15 hospitals and prospectively on data from 304 patients (503 ICU admissions) from one hospital in 2021-2024. Three datasets were used: the University of Florida Health (UFH) dataset, the electronic ICU Collaborative Research Database (eICU), and the Medical Information Mart for Intensive Care (MIMIC)-IV dataset. MANDARIN significantly outperforms the baseline neurological assessment scores (GCS, CAM, and RASS) for delirium prediction in both external (AUROC 75.5% CI: 74.2%-76.8% vs 68.3% CI: 66.9%-69.5%) and prospective (AUROC 82.0% CI: 74.8%-89.2% vs 72.7% CI: 65.5%-81.0%) cohorts, as well as for coma prediction (external AUROC 87.3% CI: 85.9%-89.0% vs 72.8% CI: 70.6%-74.9%, and prospective AUROC 93.4% CI: 88.5%-97.9% vs 67.7% CI: 57.7%-76.8%) with a 12-hour lead time. This tool has the potential to assist clinicians in decision-making by continuously monitoring the brain status of patients in the ICU.
Transparent AI: Developing an Explainable Interface for Predicting Postoperative Complications
Apr 18, 2024



Abstract:Given the sheer volume of surgical procedures and the significant rate of postoperative fatalities, assessing and managing surgical complications has become a critical public health concern. Existing artificial intelligence (AI) tools for risk surveillance and diagnosis often lack adequate interpretability, fairness, and reproducibility. To address this, we proposed an Explainable AI (XAI) framework designed to answer five critical questions: why, why not, how, what if, and what else, with the goal of enhancing the explainability and transparency of AI models. We incorporated various techniques such as Local Interpretable Model-agnostic Explanations (LIME), SHapley Additive exPlanations (SHAP), counterfactual explanations, model cards, an interactive feature manipulation interface, and the identification of similar patients to address these questions. We showcased an XAI interface prototype that adheres to this framework for predicting major postoperative complications. This initial implementation has provided valuable insights into the vast explanatory potential of our XAI framework and represents an initial step towards its clinical adoption.
A multi-cohort study on prediction of acute brain dysfunction states using selective state space models
Mar 11, 2024

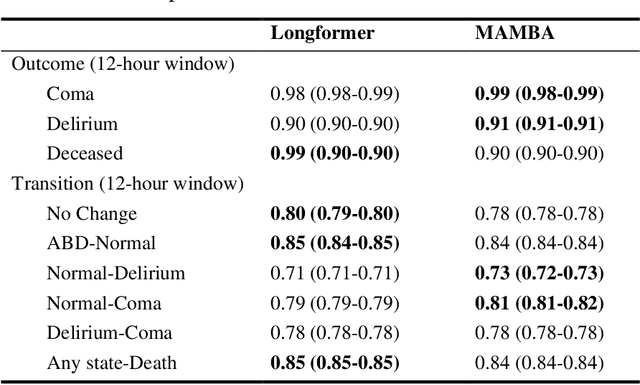

Abstract:Assessing acute brain dysfunction (ABD), including delirium and coma in the intensive care unit (ICU), is a critical challenge due to its prevalence and severe implications for patient outcomes. Current diagnostic methods rely on infrequent clinical observations, which can only determine a patient's ABD status after onset. Our research attempts to solve these problems by harnessing Electronic Health Records (EHR) data to develop automated methods for ABD prediction for patients in the ICU. Existing models solely predict a single state (e.g., either delirium or coma), require at least 24 hours of observation data to make predictions, do not dynamically predict fluctuating ABD conditions during ICU stay (typically a one-time prediction), and use small sample size, proprietary single-hospital datasets. Our research fills these gaps in the existing literature by dynamically predicting delirium, coma, and mortality for 12-hour intervals throughout an ICU stay and validating on two public datasets. Our research also introduces the concept of dynamically predicting critical transitions from non-ABD to ABD and between different ABD states in real time, which could be clinically more informative for the hospital staff. We compared the predictive performance of two state-of-the-art neural network models, the MAMBA selective state space model and the Longformer Transformer model. Using the MAMBA model, we achieved a mean area under the receiving operator characteristic curve (AUROC) of 0.95 on outcome prediction of ABD for 12-hour intervals. The model achieves a mean AUROC of 0.79 when predicting transitions between ABD states. Our study uses a curated dataset from the University of Florida Health Shands Hospital for internal validation and two publicly available datasets, MIMIC-IV and eICU, for external validation, demonstrating robustness across ICU stays from 203 hospitals and 140,945 patients.
Identifying acute illness phenotypes via deep temporal interpolation and clustering network on physiologic signatures
Jul 27, 2023Abstract:Initial hours of hospital admission impact clinical trajectory, but early clinical decisions often suffer due to data paucity. With clustering analysis for vital signs within six hours of admission, patient phenotypes with distinct pathophysiological signatures and outcomes may support early clinical decisions. We created a single-center, longitudinal EHR dataset for 75,762 adults admitted to a tertiary care center for 6+ hours. We proposed a deep temporal interpolation and clustering network to extract latent representations from sparse, irregularly sampled vital sign data and derived distinct patient phenotypes in a training cohort (n=41,502). Model and hyper-parameters were chosen based on a validation cohort (n=17,415). Test cohort (n=16,845) was used to analyze reproducibility and correlation with biomarkers. The training, validation, and testing cohorts had similar distributions of age (54-55 yrs), sex (55% female), race, comorbidities, and illness severity. Four clusters were identified. Phenotype A (18%) had most comorbid disease with higher rate of prolonged respiratory insufficiency, acute kidney injury, sepsis, and three-year mortality. Phenotypes B (33%) and C (31%) had diffuse patterns of mild organ dysfunction. Phenotype B had favorable short-term outcomes but second-highest three-year mortality. Phenotype C had favorable clinical outcomes. Phenotype D (17%) had early/persistent hypotension, high rate of early surgery, and substantial biomarker rate of inflammation but second-lowest three-year mortality. After comparing phenotypes' SOFA scores, clustering results did not simply repeat other acuity assessments. In a heterogeneous cohort, four phenotypes with distinct categories of disease and outcomes were identified by a deep temporal interpolation and clustering network. This tool may impact triage decisions and clinical decision-support under time constraints.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge