Hatice Ceylan Koydemir
Virtual Gram staining of label-free bacteria using darkfield microscopy and deep learning
Jul 17, 2024



Abstract:Gram staining has been one of the most frequently used staining protocols in microbiology for over a century, utilized across various fields, including diagnostics, food safety, and environmental monitoring. Its manual procedures make it vulnerable to staining errors and artifacts due to, e.g., operator inexperience and chemical variations. Here, we introduce virtual Gram staining of label-free bacteria using a trained deep neural network that digitally transforms darkfield images of unstained bacteria into their Gram-stained equivalents matching brightfield image contrast. After a one-time training effort, the virtual Gram staining model processes an axial stack of darkfield microscopy images of label-free bacteria (never seen before) to rapidly generate Gram staining, bypassing several chemical steps involved in the conventional staining process. We demonstrated the success of the virtual Gram staining workflow on label-free bacteria samples containing Escherichia coli and Listeria innocua by quantifying the staining accuracy of the virtual Gram staining model and comparing the chromatic and morphological features of the virtually stained bacteria against their chemically stained counterparts. This virtual bacteria staining framework effectively bypasses the traditional Gram staining protocol and its challenges, including stain standardization, operator errors, and sensitivity to chemical variations.
DUBLINE: A Deep Unfolding Network for B-line Detection in Lung Ultrasound Images
Nov 11, 2023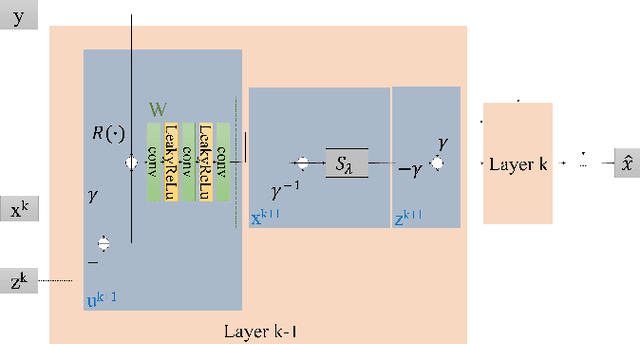
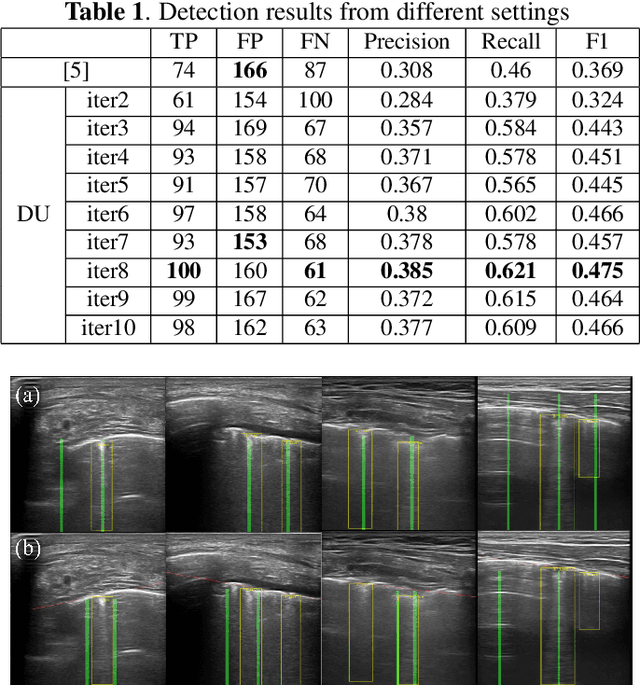

Abstract:In the context of lung ultrasound, the detection of B-lines, which are indicative of interstitial lung disease and pulmonary edema, plays a pivotal role in clinical diagnosis. Current methods still rely on visual inspection by experts. Vision-based automatic B-line detection methods have been developed, but their performance has yet to improve in terms of both accuracy and computational speed. This paper presents a novel approach to posing B-line detection as an inverse problem via deep unfolding of the Alternating Direction Method of Multipliers (ADMM). It tackles the challenges of data labelling and model training in lung ultrasound image analysis by harnessing the capabilities of deep neural networks and model-based methods. Our objective is to substantially enhance diagnostic accuracy while ensuring efficient real-time capabilities. The results show that the proposed method runs more than 90 times faster than the traditional model-based method and achieves an F1 score that is 10.6% higher.
Stain-free, rapid, and quantitative viral plaque assay using deep learning and holography
Jun 30, 2022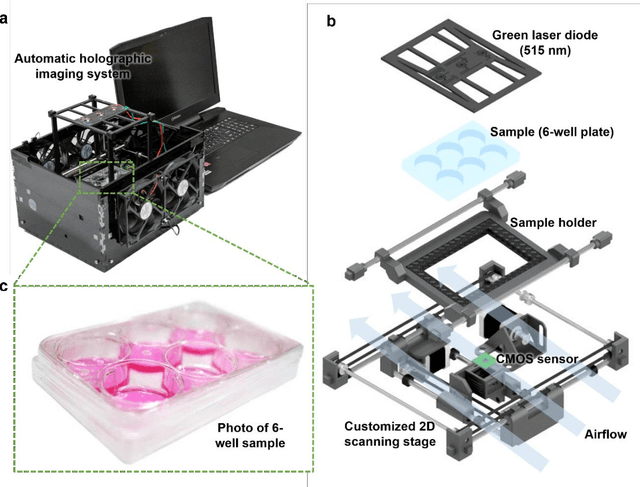
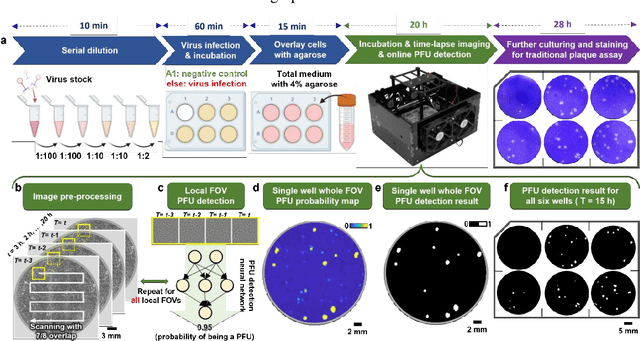
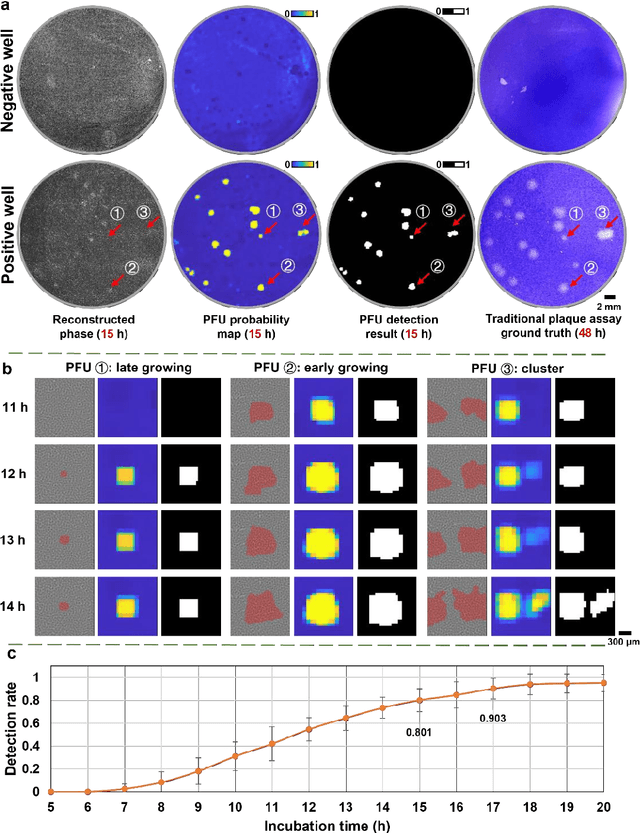

Abstract:Plaque assay is the gold standard method for quantifying the concentration of replication-competent lytic virions. Expediting and automating viral plaque assays will significantly benefit clinical diagnosis, vaccine development, and the production of recombinant proteins or antiviral agents. Here, we present a rapid and stain-free quantitative viral plaque assay using lensfree holographic imaging and deep learning. This cost-effective, compact, and automated device significantly reduces the incubation time needed for traditional plaque assays while preserving their advantages over other virus quantification methods. This device captures ~0.32 Giga-pixel/hour phase information of the objects per test well, covering an area of ~30x30 mm^2, in a label-free manner, eliminating staining entirely. We demonstrated the success of this computational method using Vero E6 cells and vesicular stomatitis virus. Using a neural network, this stain-free device automatically detected the first cell lysing events due to the viral replication as early as 5 hours after the incubation, and achieved >90% detection rate for the plaque-forming units (PFUs) with 100% specificity in <20 hours, providing major time savings compared to the traditional plaque assays that take ~48 hours or more. This data-driven plaque assay also offers the capability of quantifying the infected area of the cell monolayer, performing automated counting and quantification of PFUs and virus-infected areas over a 10-fold larger dynamic range of virus concentration than standard viral plaque assays. This compact, low-cost, automated PFU quantification device can be broadly used in virology research, vaccine development, and clinical applications
Deep Learning-enabled Detection and Classification of Bacterial Colonies using a Thin Film Transistor (TFT) Image Sensor
May 07, 2022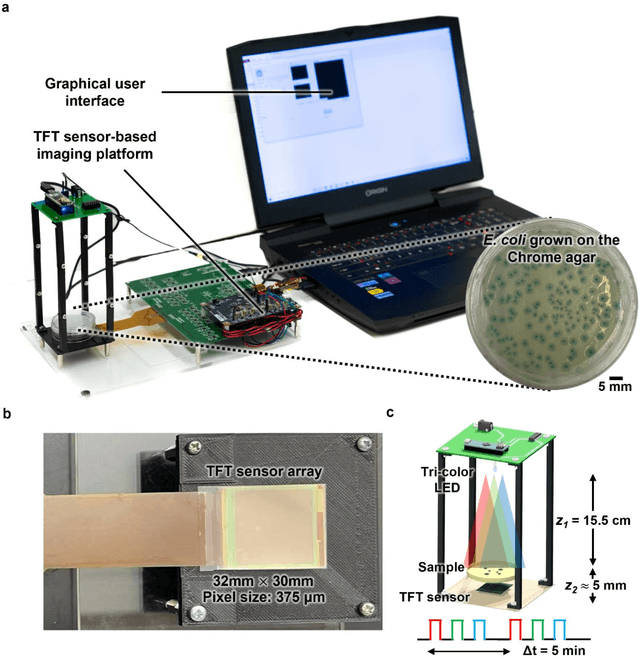
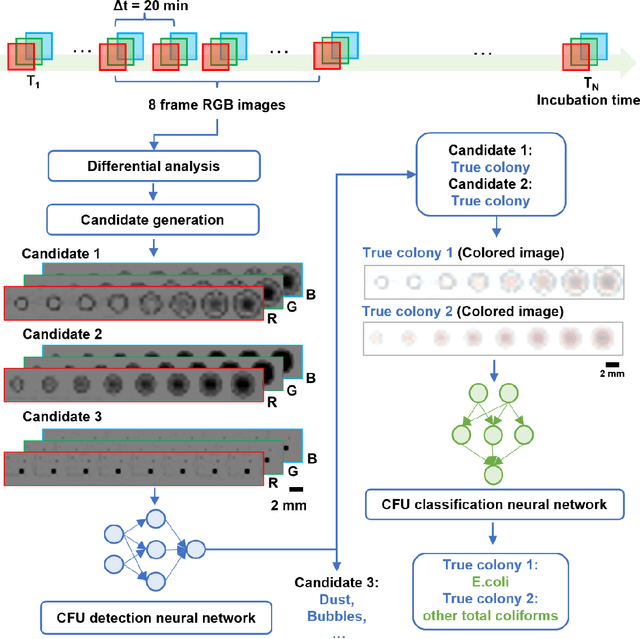

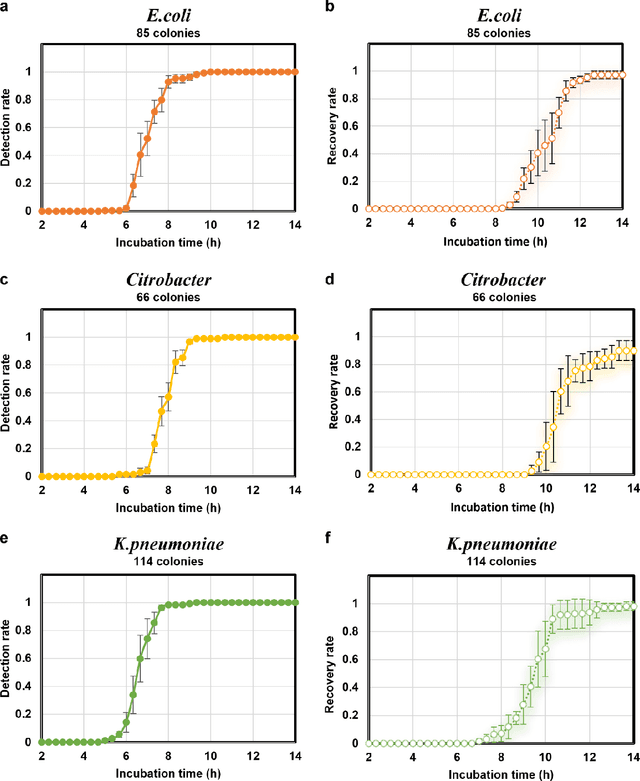
Abstract:Early detection and identification of pathogenic bacteria such as Escherichia coli (E. coli) is an essential task for public health. The conventional culture-based methods for bacterial colony detection usually take >24 hours to get the final read-out. Here, we demonstrate a bacterial colony-forming-unit (CFU) detection system exploiting a thin-film-transistor (TFT)-based image sensor array that saves ~12 hours compared to the Environmental Protection Agency (EPA)-approved methods. To demonstrate the efficacy of this CFU detection system, a lensfree imaging modality was built using the TFT image sensor with a sample field-of-view of ~10 cm^2. Time-lapse images of bacterial colonies cultured on chromogenic agar plates were automatically collected at 5-minute intervals. Two deep neural networks were used to detect and count the growing colonies and identify their species. When blindly tested with 265 colonies of E. coli and other coliform bacteria (i.e., Citrobacter and Klebsiella pneumoniae), our system reached an average CFU detection rate of 97.3% at 9 hours of incubation and an average recovery rate of 91.6% at ~12 hours. This TFT-based sensor can be applied to various microbiological detection methods. Due to the large scalability, ultra-large field-of-view, and low cost of the TFT-based image sensors, this platform can be integrated with each agar plate to be tested and disposed of after the automated CFU count. The imaging field-of-view of this platform can be cost-effectively increased to >100 cm^2 to provide a massive throughput for CFU detection using, e.g., roll-to-roll manufacturing of TFTs as used in the flexible display industry.
Early-detection and classification of live bacteria using time-lapse coherent imaging and deep learning
Jan 29, 2020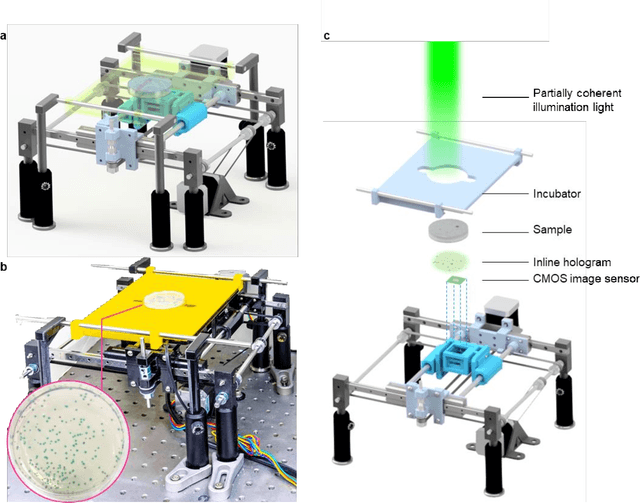
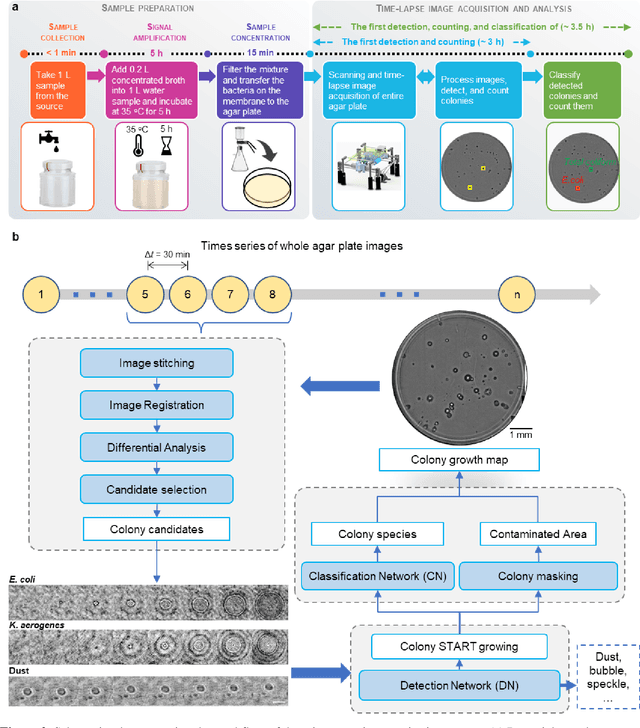
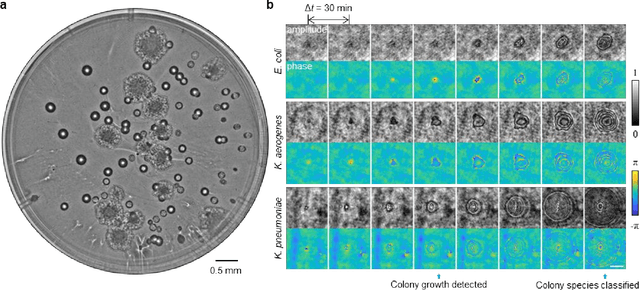
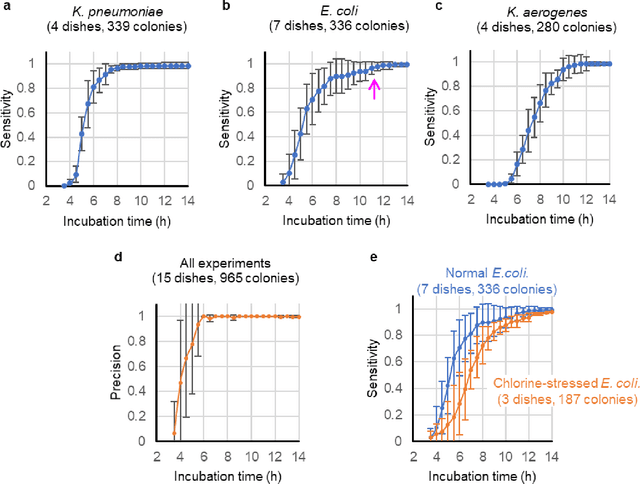
Abstract:We present a computational live bacteria detection system that periodically captures coherent microscopy images of bacterial growth inside a 60 mm diameter agar-plate and analyzes these time-lapsed holograms using deep neural networks for rapid detection of bacterial growth and classification of the corresponding species. The performance of our system was demonstrated by rapid detection of Escherichia coli and total coliform bacteria (i.e., Klebsiella aerogenes and Klebsiella pneumoniae subsp. pneumoniae) in water samples. These results were confirmed against gold-standard culture-based results, shortening the detection time of bacterial growth by >12 h as compared to the Environmental Protection Agency (EPA)-approved analytical methods. Our experiments further confirmed that this method successfully detects 90% of bacterial colonies within 7-10 h (and >95% within 12 h) with a precision of 99.2-100%, and correctly identifies their species in 7.6-12 h with 80% accuracy. Using pre-incubation of samples in growth media, our system achieved a limit of detection (LOD) of ~1 colony forming unit (CFU)/L within 9 h of total test time. This computational bacteria detection and classification platform is highly cost-effective (~$0.6 per test) and high-throughput with a scanning speed of 24 cm2/min over the entire plate surface, making it highly suitable for integration with the existing analytical methods currently used for bacteria detection on agar plates. Powered by deep learning, this automated and cost-effective live bacteria detection platform can be transformative for a wide range of applications in microbiology by significantly reducing the detection time, also automating the identification of colonies, without labeling or the need for an expert.
Deep learning enhanced mobile-phone microscopy
Dec 12, 2017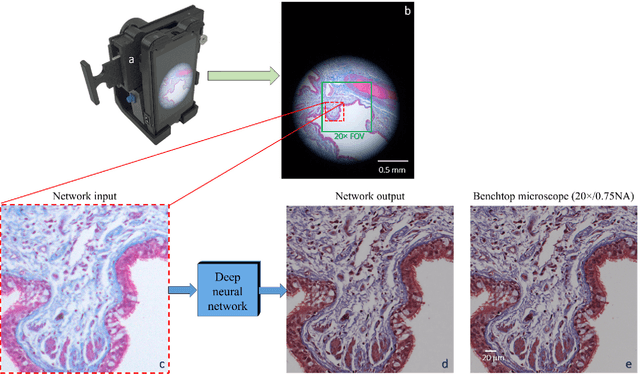
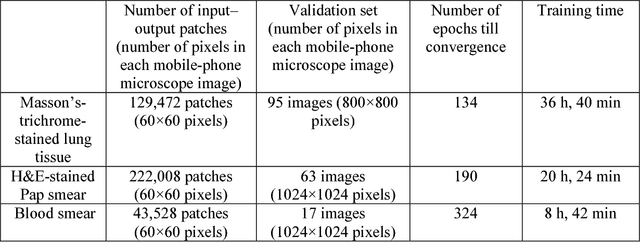
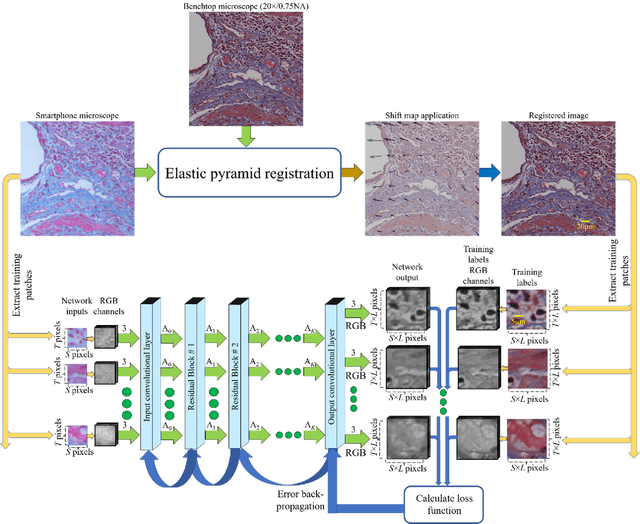
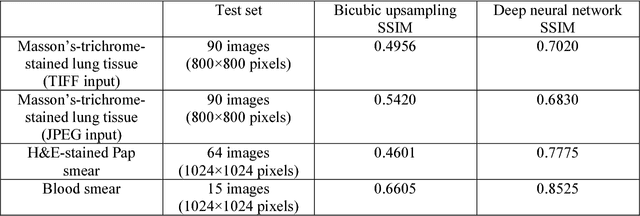
Abstract:Mobile-phones have facilitated the creation of field-portable, cost-effective imaging and sensing technologies that approach laboratory-grade instrument performance. However, the optical imaging interfaces of mobile-phones are not designed for microscopy and produce spatial and spectral distortions in imaging microscopic specimens. Here, we report on the use of deep learning to correct such distortions introduced by mobile-phone-based microscopes, facilitating the production of high-resolution, denoised and colour-corrected images, matching the performance of benchtop microscopes with high-end objective lenses, also extending their limited depth-of-field. After training a convolutional neural network, we successfully imaged various samples, including blood smears, histopathology tissue sections, and parasites, where the recorded images were highly compressed to ease storage and transmission for telemedicine applications. This method is applicable to other low-cost, aberrated imaging systems, and could offer alternatives for costly and bulky microscopes, while also providing a framework for standardization of optical images for clinical and biomedical applications.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge