Shohei Fujita
Explainable AI for Accelerated Microstructure Imaging: A SHAP-Guided Protocol on the Connectome 2.0 scanner
Sep 11, 2025Abstract:The diffusion MRI Neurite Exchange Imaging model offers a promising framework for probing gray matter microstructure by estimating parameters such as compartment sizes, diffusivities, and inter-compartmental water exchange time. However, existing protocols require long scan times. This study proposes a reduced acquisition scheme for the Connectome 2.0 scanner that preserves model accuracy while substantially shortening scan duration. We developed a data-driven framework using explainable artificial intelligence with a guided recursive feature elimination strategy to identify an optimal 8-feature subset from a 15-feature protocol. The performance of this optimized protocol was validated in vivo and benchmarked against the full acquisition and alternative reduction strategies. Parameter accuracy, preservation of anatomical contrast, and test-retest reproducibility were assessed. The reduced protocol yielded parameter estimates and cortical maps comparable to the full protocol, with low estimation errors in synthetic data and minimal impact on test-retest variability. Compared to theory-driven and heuristic reduction schemes, the optimized protocol demonstrated superior robustness, reducing the deviation in water exchange time estimates by over two-fold. In conclusion, this hybrid optimization framework enables viable imaging of neurite exchange in 14 minutes without loss of parameter fidelity. This approach supports the broader application of exchange-sensitive diffusion magnetic resonance imaging in neuroscience and clinical research, and offers a generalizable method for designing efficient acquisition protocols in biophysical parameter mapping.
MIMOSA: Multi-parametric Imaging using Multiple-echoes with Optimized Simultaneous Acquisition for highly-efficient quantitative MRI
Aug 13, 2025



Abstract:Purpose: To develop a new sequence, MIMOSA, for highly-efficient T1, T2, T2*, proton density (PD), and source separation quantitative susceptibility mapping (QSM). Methods: MIMOSA was developed based on 3D-quantification using an interleaved Look-Locker acquisition sequence with T2 preparation pulse (3D-QALAS) by combining 3D turbo Fast Low Angle Shot (FLASH) and multi-echo gradient echo acquisition modules with a spiral-like Cartesian trajectory to facilitate highly-efficient acquisition. Simulations were performed to optimize the sequence. Multi-contrast/-slice zero-shot self-supervised learning algorithm was employed for reconstruction. The accuracy of quantitative mapping was assessed by comparing MIMOSA with 3D-QALAS and reference techniques in both ISMRM/NIST phantom and in-vivo experiments. MIMOSA's acceleration capability was assessed at R = 3.3, 6.5, and 11.8 in in-vivo experiments, with repeatability assessed through scan-rescan studies. Beyond the 3T experiments, mesoscale quantitative mapping was performed at 750 um isotropic resolution at 7T. Results: Simulations demonstrated that MIMOSA achieved improved parameter estimation accuracy compared to 3D-QALAS. Phantom experiments indicated that MIMOSA exhibited better agreement with the reference techniques than 3D-QALAS. In-vivo experiments demonstrated that an acceleration factor of up to R = 11.8-fold can be achieved while preserving parameter estimation accuracy, with intra-class correlation coefficients of 0.998 (T1), 0.973 (T2), 0.947 (T2*), 0.992 (QSM), 0.987 (paramagnetic susceptibility), and 0.977 (diamagnetic susceptibility) in scan-rescan studies. Whole-brain T1, T2, T2*, PD, source separation QSM were obtained with 1 mm isotropic resolution in 3 min at 3T and 750 um isotropic resolution in 13 min at 7T. Conclusion: MIMOSA demonstrated potential for highly-efficient multi-parametric mapping.
A Tutorial on MRI Reconstruction: From Modern Methods to Clinical Implications
Jul 22, 2025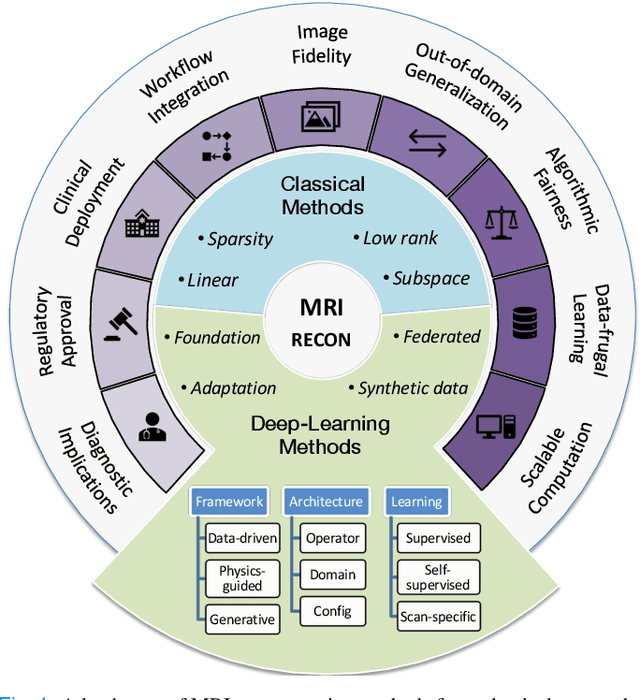
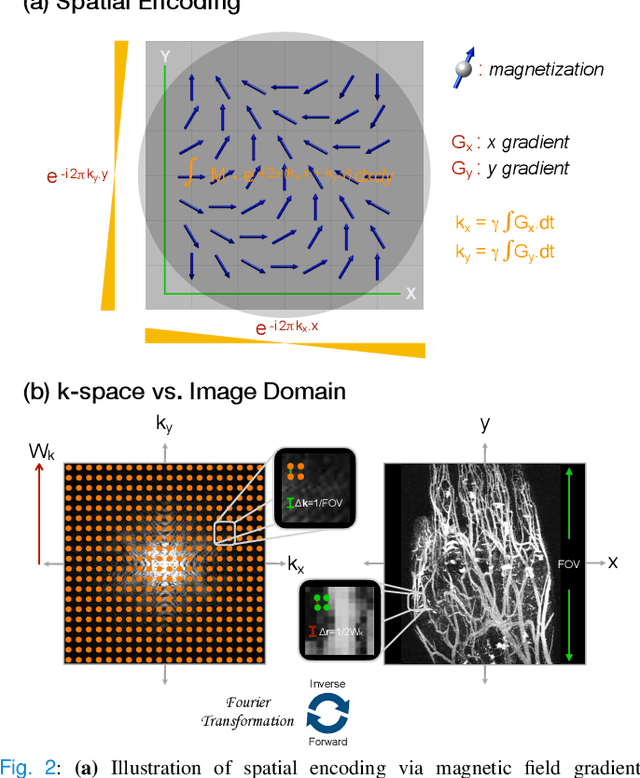
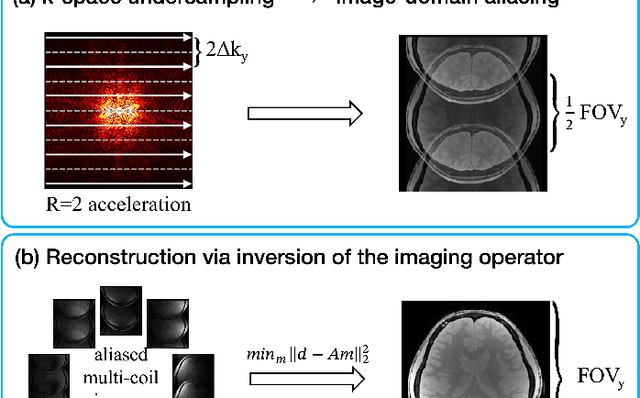
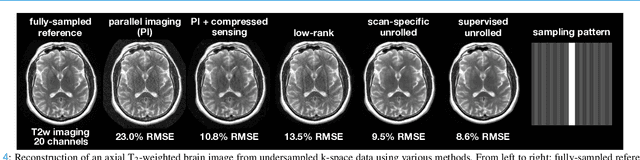
Abstract:MRI is an indispensable clinical tool, offering a rich variety of tissue contrasts to support broad diagnostic and research applications. Clinical exams routinely acquire multiple structural sequences that provide complementary information for differential diagnosis, while research protocols often incorporate advanced functional, diffusion, spectroscopic, and relaxometry sequences to capture multidimensional insights into tissue structure and composition. However, these capabilities come at the cost of prolonged scan times, which reduce patient throughput, increase susceptibility to motion artifacts, and may require trade-offs in image quality or diagnostic scope. Over the last two decades, advances in image reconstruction algorithms--alongside improvements in hardware and pulse sequence design--have made it possible to accelerate acquisitions while preserving diagnostic quality. Central to this progress is the ability to incorporate prior information to regularize the solutions to the reconstruction problem. In this tutorial, we overview the basics of MRI reconstruction and highlight state-of-the-art approaches, beginning with classical methods that rely on explicit hand-crafted priors, and then turning to deep learning methods that leverage a combination of learned and crafted priors to further push the performance envelope. We also explore the translational aspects and eventual clinical implications of these methods. We conclude by discussing future directions to address remaining challenges in MRI reconstruction. The tutorial is accompanied by a Python toolbox (https://github.com/tutorial-MRI-recon/tutorial) to demonstrate select methods discussed in the article.
Efficient MedSAMs: Segment Anything in Medical Images on Laptop
Dec 20, 2024


Abstract:Promptable segmentation foundation models have emerged as a transformative approach to addressing the diverse needs in medical images, but most existing models require expensive computing, posing a big barrier to their adoption in clinical practice. In this work, we organized the first international competition dedicated to promptable medical image segmentation, featuring a large-scale dataset spanning nine common imaging modalities from over 20 different institutions. The top teams developed lightweight segmentation foundation models and implemented an efficient inference pipeline that substantially reduced computational requirements while maintaining state-of-the-art segmentation accuracy. Moreover, the post-challenge phase advanced the algorithms through the design of performance booster and reproducibility tasks, resulting in improved algorithms and validated reproducibility of the winning solution. Furthermore, the best-performing algorithms have been incorporated into the open-source software with a user-friendly interface to facilitate clinical adoption. The data and code are publicly available to foster the further development of medical image segmentation foundation models and pave the way for impactful real-world applications.
PRIME: Phase Reversed Interleaved Multi-Echo acquisition enables highly accelerated distortion-free diffusion MRI
Sep 11, 2024


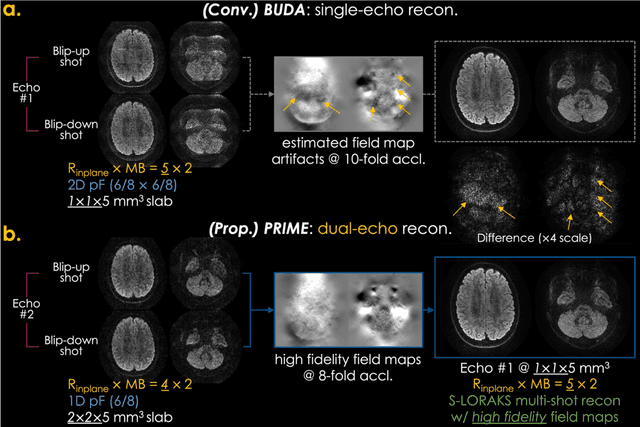
Abstract:Purpose: To develop and evaluate a new pulse sequence for highly accelerated distortion-free diffusion MRI (dMRI) by inserting an additional echo without prolonging TR, when generalized slice dithered enhanced resolution (gSlider) radiofrequency encoding is used for volumetric acquisition. Methods: A phase-reversed interleaved multi-echo acquisition (PRIME) was developed for rapid, high-resolution, and distortion-free dMRI, which includes two echoes where the first echo is for target diffusion-weighted imaging (DWI) acquisition with high-resolution and the second echo is acquired with either 1) lower-resolution for high-fidelity field map estimation, or 2) matching resolution to enable efficient diffusion relaxometry acquisitions. The sequence was evaluated on in vivo data acquired from healthy volunteers on clinical and Connectome 2.0 scanners. Results: In vivo experiments demonstrated that 1) high in-plane acceleration (Rin-plane of 5-fold with 2D partial Fourier) was achieved using the high-fidelity field maps estimated from the second echo, which was made at a lower resolution/acceleration to increase its SNR while matching the effective echo spacing of the first readout, 2) high-resolution diffusion relaxometry parameters were estimated from dual-echo PRIME data using a white matter model of multi-TE spherical mean technique (MTE-SMT), and 3) high-fidelity mesoscale DWI at 550 um isotropic resolution could be obtained in vivo by capitalizing on the high-performance gradients of the Connectome 2.0 scanner. Conclusion: The proposed PRIME sequence enabled highly accelerated, high-resolution, and distortion-free dMRI using an additional echo without prolonging scan time when gSlider encoding is utilized.
Zero-DeepSub: Zero-Shot Deep Subspace Reconstruction for Rapid Multiparametric Quantitative MRI Using 3D-QALAS
Jul 04, 2023Abstract:Purpose: To develop and evaluate methods for 1) reconstructing 3D-quantification using an interleaved Look-Locker acquisition sequence with T2 preparation pulse (3D-QALAS) time-series images using a low-rank subspace method, which enables accurate and rapid T1 and T2 mapping, and 2) improving the fidelity of subspace QALAS by combining scan-specific deep-learning-based reconstruction and subspace modeling. Methods: A low-rank subspace method for 3D-QALAS (i.e., subspace QALAS) and zero-shot deep-learning subspace method (i.e., Zero-DeepSub) were proposed for rapid and high fidelity T1 and T2 mapping and time-resolved imaging using 3D-QALAS. Using an ISMRM/NIST system phantom, the accuracy of the T1 and T2 maps estimated using the proposed methods was evaluated by comparing them with reference techniques. The reconstruction performance of the proposed subspace QALAS using Zero-DeepSub was evaluated in vivo and compared with conventional QALAS at high reduction factors of up to 9-fold. Results: Phantom experiments showed that subspace QALAS had good linearity with respect to the reference methods while reducing biases compared to conventional QALAS, especially for T2 maps. Moreover, in vivo results demonstrated that subspace QALAS had better g-factor maps and could reduce voxel blurring, noise, and artifacts compared to conventional QALAS and showed robust performance at up to 9-fold acceleration with Zero-DeepSub, which enabled whole-brain T1, T2, and PD mapping at 1 mm isotropic resolution within 2 min of scan time. Conclusion: The proposed subspace QALAS along with Zero-DeepSub enabled high fidelity and rapid whole-brain multiparametric quantification and time-resolved imaging.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge