Jiayu Lei
AutoRG-Brain: Grounded Report Generation for Brain MRI
Jul 26, 2024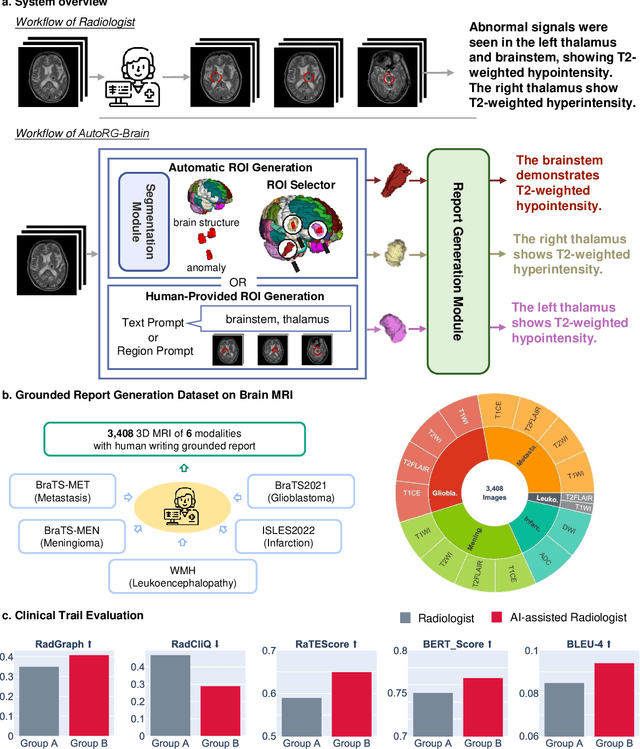
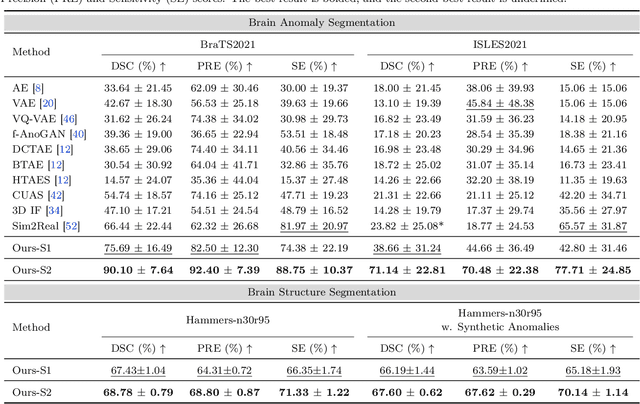
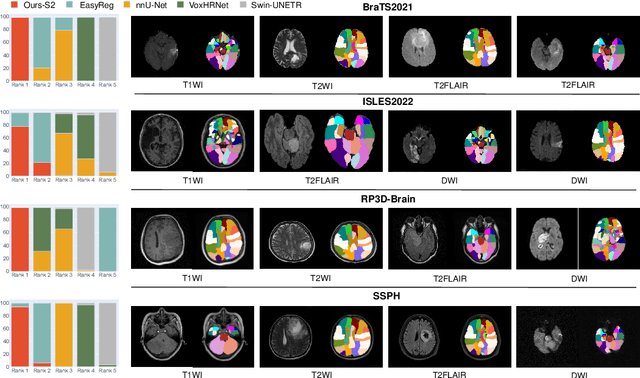
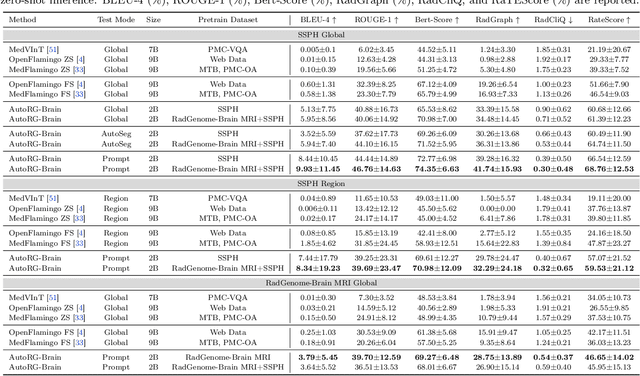
Abstract:Radiologists are tasked with interpreting a large number of images in a daily base, with the responsibility of generating corresponding reports. This demanding workload elevates the risk of human error, potentially leading to treatment delays, increased healthcare costs, revenue loss, and operational inefficiencies. To address these challenges, we initiate a series of work on grounded Automatic Report Generation (AutoRG), starting from the brain MRI interpretation system, which supports the delineation of brain structures, the localization of anomalies, and the generation of well-organized findings. We make contributions from the following aspects, first, on dataset construction, we release a comprehensive dataset encompassing segmentation masks of anomaly regions and manually authored reports, termed as RadGenome-Brain MRI. This data resource is intended to catalyze ongoing research and development in the field of AI-assisted report generation systems. Second, on system design, we propose AutoRG-Brain, the first brain MRI report generation system with pixel-level grounded visual clues. Third, for evaluation, we conduct quantitative assessments and human evaluations of brain structure segmentation, anomaly localization, and report generation tasks to provide evidence of its reliability and accuracy. This system has been integrated into real clinical scenarios, where radiologists were instructed to write reports based on our generated findings and anomaly segmentation masks. The results demonstrate that our system enhances the report-writing skills of junior doctors, aligning their performance more closely with senior doctors, thereby boosting overall productivity.
RadGenome-Chest CT: A Grounded Vision-Language Dataset for Chest CT Analysis
Apr 25, 2024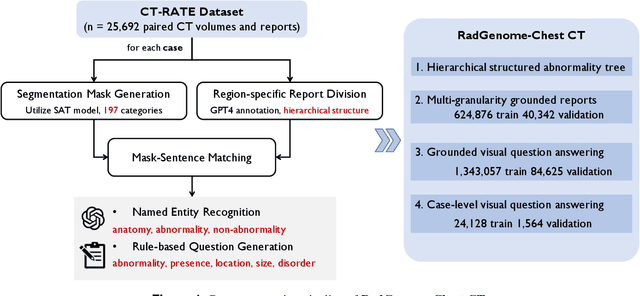


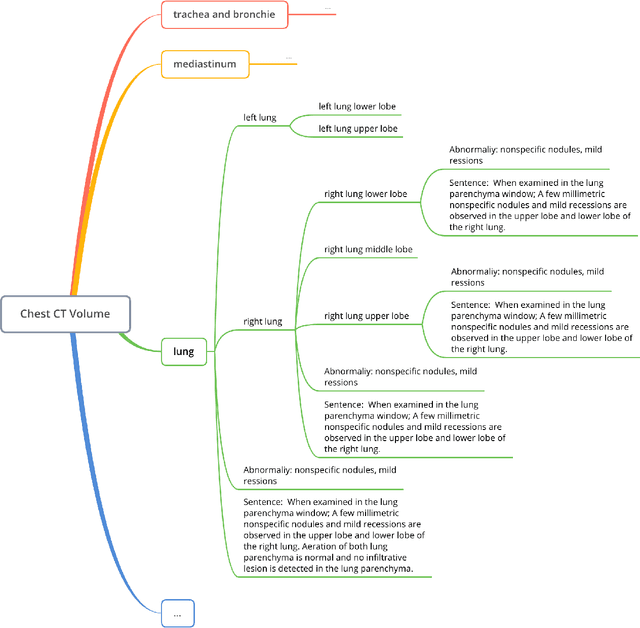
Abstract:Developing generalist foundation model has recently attracted tremendous attention among researchers in the field of AI for Medicine (AI4Medicine). A pivotal insight in developing these models is their reliance on dataset scaling, which emphasizes the requirements on developing open-source medical image datasets that incorporate diverse supervision signals across various imaging modalities. In this paper, we introduce RadGenome-Chest CT, a comprehensive, large-scale, region-guided 3D chest CT interpretation dataset based on CT-RATE. Specifically, we leverage the latest powerful universal segmentation and large language models, to extend the original datasets (over 25,692 non-contrast 3D chest CT volume and reports from 20,000 patients) from the following aspects: (i) organ-level segmentation masks covering 197 categories, which provide intermediate reasoning visual clues for interpretation; (ii) 665 K multi-granularity grounded reports, where each sentence of the report is linked to the corresponding anatomical region of CT volume in the form of a segmentation mask; (iii) 1.3 M grounded VQA pairs, where questions and answers are all linked with reference segmentation masks, enabling models to associate visual evidence with textual explanations. All grounded reports and VQA pairs in the validation set have gone through manual verification to ensure dataset quality. We believe that RadGenome-Chest CT can significantly advance the development of multimodal medical foundation models, by training to generate texts based on given segmentation regions, which is unattainable with previous relevant datasets. We will release all segmentation masks, grounded reports, and VQA pairs to facilitate further research and development in this field.
Can GPT-4V Serve Medical Applications? Case Studies on GPT-4V for Multimodal Medical Diagnosis
Oct 17, 2023Abstract:Driven by the large foundation models, the development of artificial intelligence has witnessed tremendous progress lately, leading to a surge of general interest from the public. In this study, we aim to assess the performance of OpenAI's newest model, GPT-4V(ision), specifically in the realm of multimodal medical diagnosis. Our evaluation encompasses 17 human body systems, including Central Nervous System, Head and Neck, Cardiac, Chest, Hematology, Hepatobiliary, Gastrointestinal, Urogenital, Gynecology, Obstetrics, Breast, Musculoskeletal, Spine, Vascular, Oncology, Trauma, Pediatrics, with images taken from 8 modalities used in daily clinic routine, e.g., X-ray, Computed Tomography (CT), Magnetic Resonance Imaging (MRI), Positron Emission Tomography (PET), Digital Subtraction Angiography (DSA), Mammography, Ultrasound, and Pathology. We probe the GPT-4V's ability on multiple clinical tasks with or without patent history provided, including imaging modality and anatomy recognition, disease diagnosis, report generation, disease localisation. Our observation shows that, while GPT-4V demonstrates proficiency in distinguishing between medical image modalities and anatomy, it faces significant challenges in disease diagnosis and generating comprehensive reports. These findings underscore that while large multimodal models have made significant advancements in computer vision and natural language processing, it remains far from being used to effectively support real-world medical applications and clinical decision-making. All images used in this report can be found in https://github.com/chaoyi-wu/GPT-4V_Medical_Evaluation.
UniBrain: Universal Brain MRI Diagnosis with Hierarchical Knowledge-enhanced Pre-training
Sep 13, 2023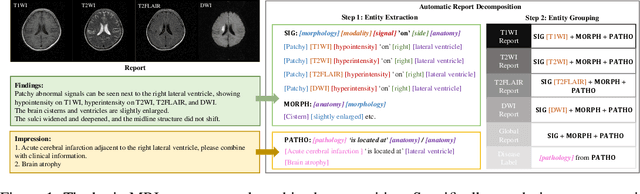
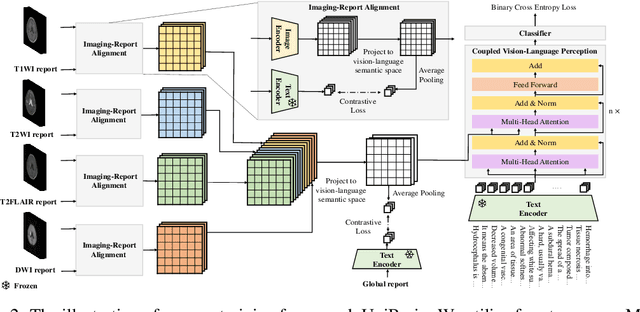
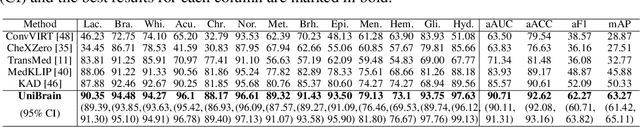
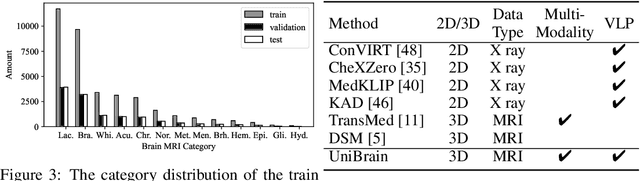
Abstract:Magnetic resonance imaging~(MRI) have played a crucial role in brain disease diagnosis, with which a range of computer-aided artificial intelligence methods have been proposed. However, the early explorations usually focus on the limited types of brain diseases in one study and train the model on the data in a small scale, yielding the bottleneck of generalization. Towards a more effective and scalable paradigm, we propose a hierarchical knowledge-enhanced pre-training framework for the universal brain MRI diagnosis, termed as UniBrain. Specifically, UniBrain leverages a large-scale dataset of 24,770 imaging-report pairs from routine diagnostics. Different from previous pre-training techniques for the unitary vision or textual feature, or with the brute-force alignment between vision and language information, we leverage the unique characteristic of report information in different granularity to build a hierarchical alignment mechanism, which strengthens the efficiency in feature learning. Our UniBrain is validated on three real world datasets with severe class imbalance and the public BraTS2019 dataset. It not only consistently outperforms all state-of-the-art diagnostic methods by a large margin and provides a superior grounding performance but also shows comparable performance compared to expert radiologists on certain disease types.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge