Hooman Vaseli
ControlEchoSynth: Boosting Ejection Fraction Estimation Models via Controlled Video Diffusion
Aug 25, 2025



Abstract:Synthetic data generation represents a significant advancement in boosting the performance of machine learning (ML) models, particularly in fields where data acquisition is challenging, such as echocardiography. The acquisition and labeling of echocardiograms (echo) for heart assessment, crucial in point-of-care ultrasound (POCUS) settings, often encounter limitations due to the restricted number of echo views available, typically captured by operators with varying levels of experience. This study proposes a novel approach for enhancing clinical diagnosis accuracy by synthetically generating echo views. These views are conditioned on existing, real views of the heart, focusing specifically on the estimation of ejection fraction (EF), a critical parameter traditionally measured from biplane apical views. By integrating a conditional generative model, we demonstrate an improvement in EF estimation accuracy, providing a comparative analysis with traditional methods. Preliminary results indicate that our synthetic echoes, when used to augment existing datasets, not only enhance EF estimation but also show potential in advancing the development of more robust, accurate, and clinically relevant ML models. This approach is anticipated to catalyze further research in synthetic data applications, paving the way for innovative solutions in medical imaging diagnostics.
Reliable Multi-View Learning with Conformal Prediction for Aortic Stenosis Classification in Echocardiography
Sep 15, 2024Abstract:The fundamental problem with ultrasound-guided diagnosis is that the acquired images are often 2-D cross-sections of a 3-D anatomy, potentially missing important anatomical details. This limitation leads to challenges in ultrasound echocardiography, such as poor visualization of heart valves or foreshortening of ventricles. Clinicians must interpret these images with inherent uncertainty, a nuance absent in machine learning's one-hot labels. We propose Re-Training for Uncertainty (RT4U), a data-centric method to introduce uncertainty to weakly informative inputs in the training set. This simple approach can be incorporated to existing state-of-the-art aortic stenosis classification methods to further improve their accuracy. When combined with conformal prediction techniques, RT4U can yield adaptively sized prediction sets which are guaranteed to contain the ground truth class to a high accuracy. We validate the effectiveness of RT4U on three diverse datasets: a public (TMED-2) and a private AS dataset, along with a CIFAR-10-derived toy dataset. Results show improvement on all the datasets.
ProtoASNet: Dynamic Prototypes for Inherently Interpretable and Uncertainty-Aware Aortic Stenosis Classification in Echocardiography
Jul 26, 2023


Abstract:Aortic stenosis (AS) is a common heart valve disease that requires accurate and timely diagnosis for appropriate treatment. Most current automatic AS severity detection methods rely on black-box models with a low level of trustworthiness, which hinders clinical adoption. To address this issue, we propose ProtoASNet, a prototypical network that directly detects AS from B-mode echocardiography videos, while making interpretable predictions based on the similarity between the input and learned spatio-temporal prototypes. This approach provides supporting evidence that is clinically relevant, as the prototypes typically highlight markers such as calcification and restricted movement of aortic valve leaflets. Moreover, ProtoASNet utilizes abstention loss to estimate aleatoric uncertainty by defining a set of prototypes that capture ambiguity and insufficient information in the observed data. This provides a reliable system that can detect and explain when it may fail. We evaluate ProtoASNet on a private dataset and the publicly available TMED-2 dataset, where it outperforms existing state-of-the-art methods with an accuracy of 80.0% and 79.7%, respectively. Furthermore, ProtoASNet provides interpretability and an uncertainty measure for each prediction, which can improve transparency and facilitate the interactive usage of deep networks to aid clinical decision-making. Our source code is available at: https://github.com/hooman007/ProtoASNet.
EchoGLAD: Hierarchical Graph Neural Networks for Left Ventricle Landmark Detection on Echocardiograms
Jul 23, 2023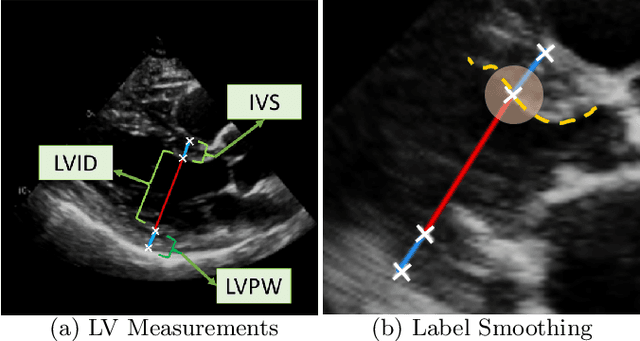
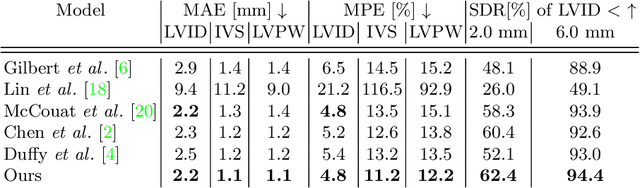
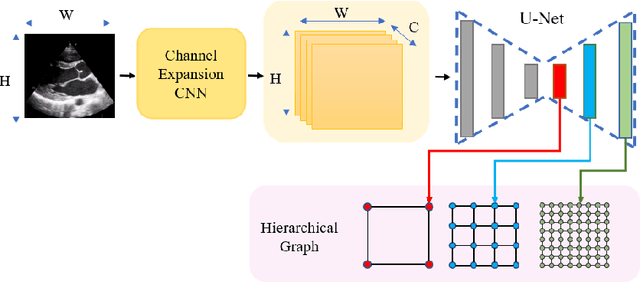
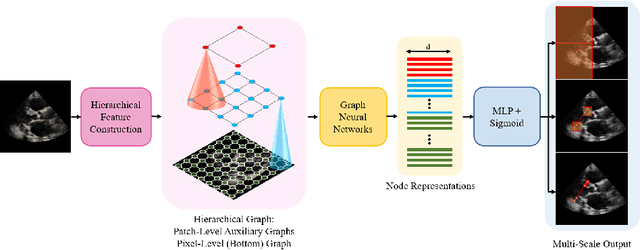
Abstract:The functional assessment of the left ventricle chamber of the heart requires detecting four landmark locations and measuring the internal dimension of the left ventricle and the approximate mass of the surrounding muscle. The key challenge of automating this task with machine learning is the sparsity of clinical labels, i.e., only a few landmark pixels in a high-dimensional image are annotated, leading many prior works to heavily rely on isotropic label smoothing. However, such a label smoothing strategy ignores the anatomical information of the image and induces some bias. To address this challenge, we introduce an echocardiogram-based, hierarchical graph neural network (GNN) for left ventricle landmark detection (EchoGLAD). Our main contributions are: 1) a hierarchical graph representation learning framework for multi-resolution landmark detection via GNNs; 2) induced hierarchical supervision at different levels of granularity using a multi-level loss. We evaluate our model on a public and a private dataset under the in-distribution (ID) and out-of-distribution (OOD) settings. For the ID setting, we achieve the state-of-the-art mean absolute errors (MAEs) of 1.46 mm and 1.86 mm on the two datasets. Our model also shows better OOD generalization than prior works with a testing MAE of 4.3 mm.
On Modelling Label Uncertainty in Deep Neural Networks: Automatic Estimation of Intra-observer Variability in 2D Echocardiography Quality Assessment
Nov 02, 2019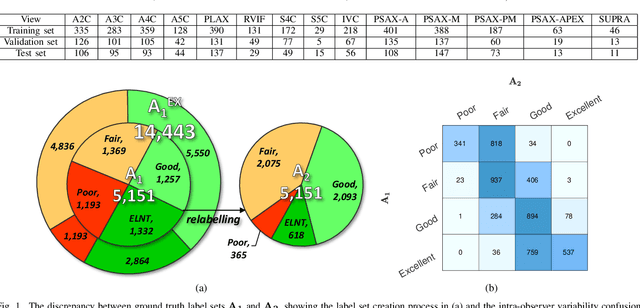
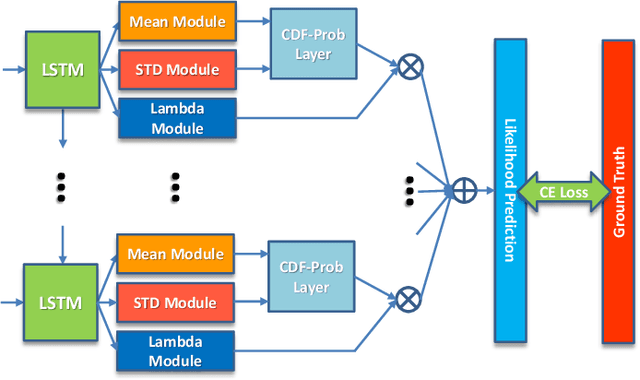
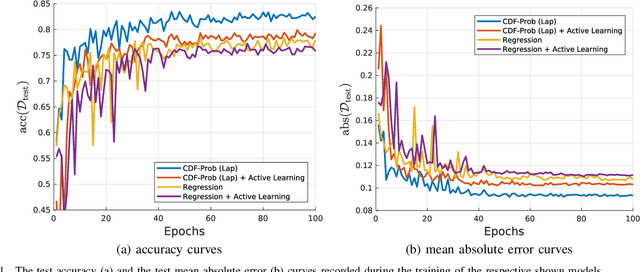
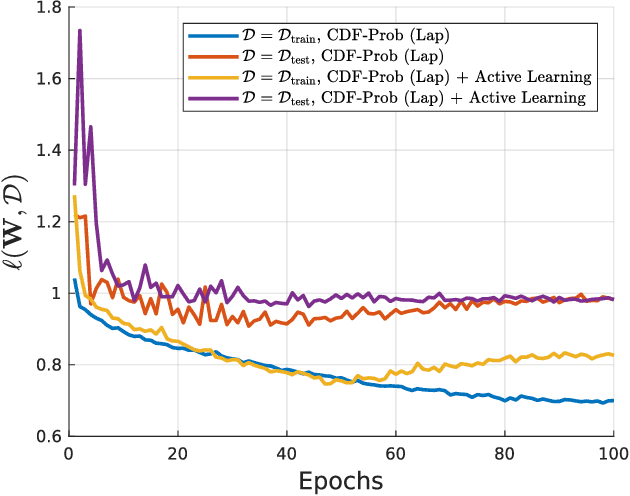
Abstract:Uncertainty of labels in clinical data resulting from intra-observer variability can have direct impact on the reliability of assessments made by deep neural networks. In this paper, we propose a method for modelling such uncertainty in the context of 2D echocardiography (echo), which is a routine procedure for detecting cardiovascular disease at point-of-care. Echo imaging quality and acquisition time is highly dependent on the operator's experience level. Recent developments have shown the possibility of automating echo image quality quantification by mapping an expert's assessment of quality to the echo image via deep learning techniques. Nevertheless, the observer variability in the expert's assessment can impact the quality quantification accuracy. Here, we aim to model the intra-observer variability in echo quality assessment as an aleatoric uncertainty modelling regression problem with the introduction of a novel method that handles the regression problem with categorical labels. A key feature of our design is that only a single forward pass is sufficient to estimate the level of uncertainty for the network output. Compared to the $0.11 \pm 0.09$ absolute error (in a scale from 0 to 1) archived by the conventional regression method, the proposed method brings the error down to $0.09 \pm 0.08$, where the improvement is statistically significant and equivalents to $5.7\%$ test accuracy improvement. The simplicity of the proposed approach means that it could be generalized to other applications of deep learning in medical imaging, where there is often uncertainty in clinical labels.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge