David Schinz
Department of Diagnostic and Interventional Neuroradiology, School of Medicine, Klinikum rechts der Isar, Technical University of Munich, Germany
Counterfactual Explanations for Medical Image Classification and Regression using Diffusion Autoencoder
Aug 02, 2024



Abstract:Counterfactual explanations (CEs) aim to enhance the interpretability of machine learning models by illustrating how alterations in input features would affect the resulting predictions. Common CE approaches require an additional model and are typically constrained to binary counterfactuals. In contrast, we propose a novel method that operates directly on the latent space of a generative model, specifically a Diffusion Autoencoder (DAE). This approach offers inherent interpretability by enabling the generation of CEs and the continuous visualization of the model's internal representation across decision boundaries. Our method leverages the DAE's ability to encode images into a semantically rich latent space in an unsupervised manner, eliminating the need for labeled data or separate feature extraction models. We show that these latent representations are helpful for medical condition classification and the ordinal regression of severity pathologies, such as vertebral compression fractures (VCF) and diabetic retinopathy (DR). Beyond binary CEs, our method supports the visualization of ordinal CEs using a linear model, providing deeper insights into the model's decision-making process and enhancing interpretability. Experiments across various medical imaging datasets demonstrate the method's advantages in interpretability and versatility. The linear manifold of the DAE's latent space allows for meaningful interpolation and manipulation, making it a powerful tool for exploring medical image properties. Our code is available at https://github.com/matanat/dae_counterfactual.
Enhancing Interpretability of Vertebrae Fracture Grading using Human-interpretable Prototypes
Apr 03, 2024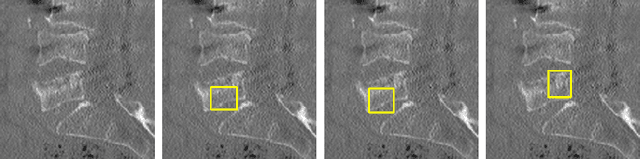

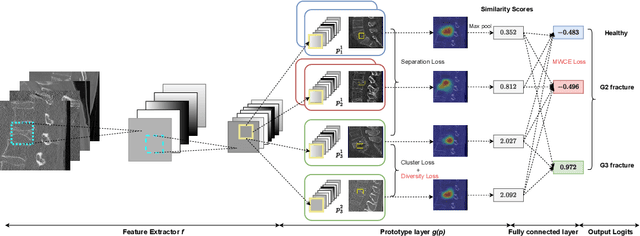

Abstract:Vertebral fracture grading classifies the severity of vertebral fractures, which is a challenging task in medical imaging and has recently attracted Deep Learning (DL) models. Only a few works attempted to make such models human-interpretable despite the need for transparency and trustworthiness in critical use cases like DL-assisted medical diagnosis. Moreover, such models either rely on post-hoc methods or additional annotations. In this work, we propose a novel interpretable-by-design method, ProtoVerse, to find relevant sub-parts of vertebral fractures (prototypes) that reliably explain the model's decision in a human-understandable way. Specifically, we introduce a novel diversity-promoting loss to mitigate prototype repetitions in small datasets with intricate semantics. We have experimented with the VerSe'19 dataset and outperformed the existing prototype-based method. Further, our model provides superior interpretability against the post-hoc method. Importantly, expert radiologists validated the visual interpretability of our results, showing clinical applicability.
Semantic Latent Space Regression of Diffusion Autoencoders for Vertebral Fracture Grading
Mar 21, 2023



Abstract:Vertebral fractures are a consequence of osteoporosis, with significant health implications for affected patients. Unfortunately, grading their severity using CT exams is hard and subjective, motivating automated grading methods. However, current approaches are hindered by imbalance and scarcity of data and a lack of interpretability. To address these challenges, this paper proposes a novel approach that leverages unlabelled data to train a generative Diffusion Autoencoder (DAE) model as an unsupervised feature extractor. We model fracture grading as a continuous regression, which is more reflective of the smooth progression of fractures. Specifically, we use a binary, supervised fracture classifier to construct a hyperplane in the DAE's latent space. We then regress the severity of the fracture as a function of the distance to this hyperplane, calibrating the results to the Genant scale. Importantly, the generative nature of our method allows us to visualize different grades of a given vertebra, providing interpretability and insight into the features that contribute to automated grading.
Interpretable Vertebral Fracture Diagnosis
Mar 30, 2022
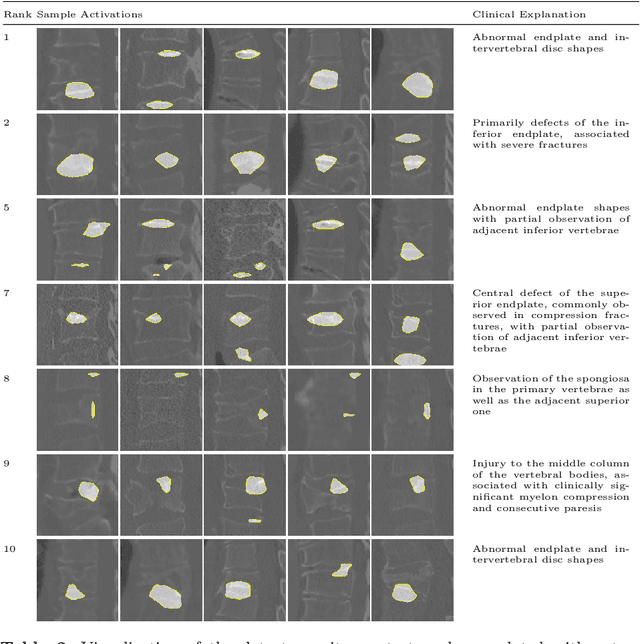
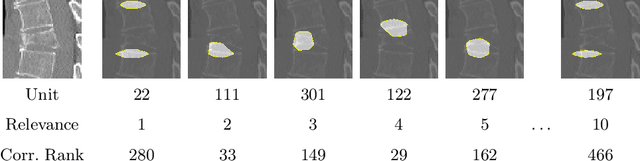
Abstract:Do black-box neural network models learn clinically relevant features for fracture diagnosis? The answer not only establishes reliability quenches scientific curiosity but also leads to explainable and verbose findings that can assist the radiologists in the final and increase trust. This work identifies the concepts networks use for vertebral fracture diagnosis in CT images. This is achieved by associating concepts to neurons highly correlated with a specific diagnosis in the dataset. The concepts are either associated with neurons by radiologists pre-hoc or are visualized during a specific prediction and left for the user's interpretation. We evaluate which concepts lead to correct diagnosis and which concepts lead to false positives. The proposed frameworks and analysis pave the way for reliable and explainable vertebral fracture diagnosis.
A Computed Tomography Vertebral Segmentation Dataset with Anatomical Variations and Multi-Vendor Scanner Data
Mar 10, 2021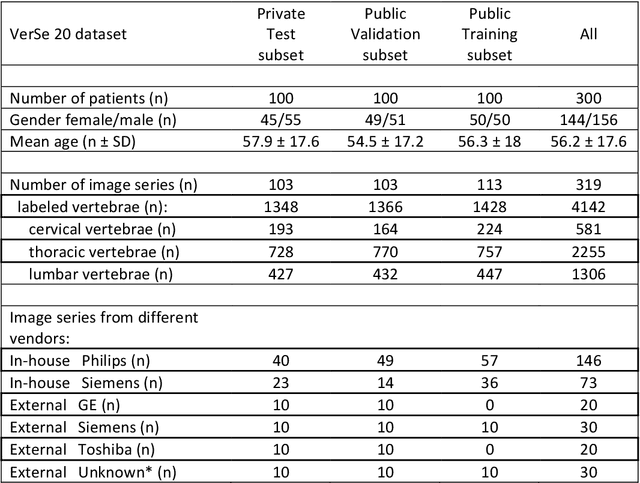
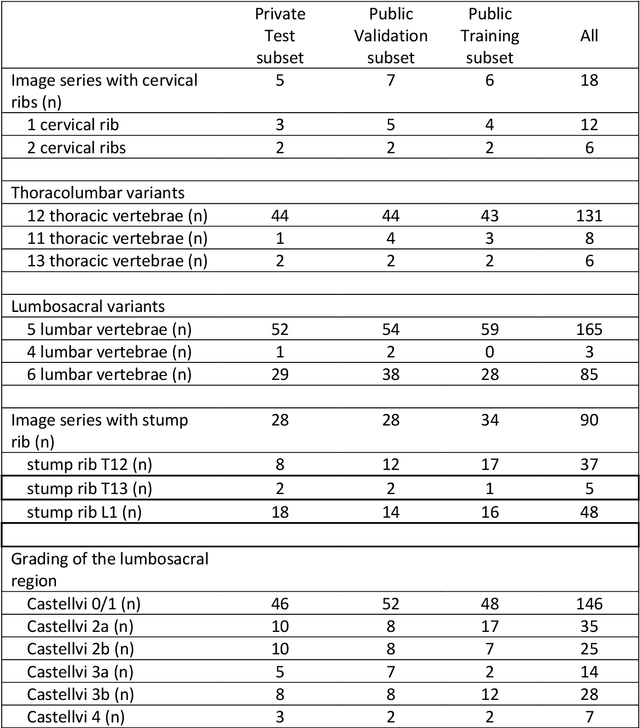
Abstract:With the advent of deep learning algorithms, fully automated radiological image analysis is within reach. In spine imaging, several atlas- and shape-based as well as deep learning segmentation algorithms have been proposed, allowing for subsequent automated analysis of morphology and pathology. The first Large Scale Vertebrae Segmentation Challenge (VerSe 2019) showed that these perform well on normal anatomy, but fail in variants not frequently present in the training dataset. Building on that experience, we report on the largely increased VerSe 2020 dataset and results from the second iteration of the VerSe challenge (MICCAI 2020, Lima, Peru). VerSe 2020 comprises annotated spine computed tomography (CT) images from 300 subjects with 4142 fully visualized and annotated vertebrae, collected across multiple centres from four different scanner manufacturers, enriched with cases that exhibit anatomical variants such as enumeration abnormalities (n=77) and transitional vertebrae (n=161). Metadata includes vertebral labelling information, voxel-level segmentation masks obtained with a human-machine hybrid algorithm and anatomical ratings, to enable the development and benchmarking of robust and accurate segmentation algorithms.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge