Benjamin Movsas
Automated stereotactic radiosurgery planning using a human-in-the-loop reasoning large language model agent
Dec 23, 2025


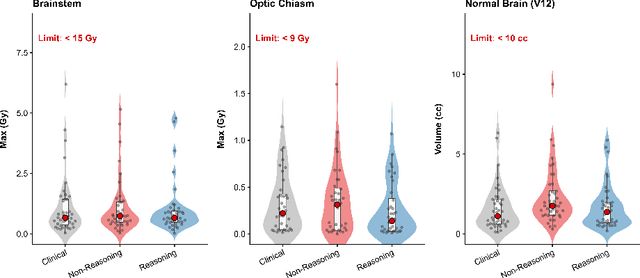
Abstract:Stereotactic radiosurgery (SRS) demands precise dose shaping around critical structures, yet black-box AI systems have limited clinical adoption due to opacity concerns. We tested whether chain-of-thought reasoning improves agentic planning in a retrospective cohort of 41 patients with brain metastases treated with 18 Gy single-fraction SRS. We developed SAGE (Secure Agent for Generative Dose Expertise), an LLM-based planning agent for automated SRS treatment planning. Two variants generated plans for each case: one using a non-reasoning model, one using a reasoning model. The reasoning variant showed comparable plan dosimetry relative to human planners on primary endpoints (PTV coverage, maximum dose, conformity index, gradient index; all p > 0.21) while reducing cochlear dose below human baselines (p = 0.022). When prompted to improve conformity, the reasoning model demonstrated systematic planning behaviors including prospective constraint verification (457 instances) and trade-off deliberation (609 instances), while the standard model exhibited none of these deliberative processes (0 and 7 instances, respectively). Content analysis revealed that constraint verification and causal explanation concentrated in the reasoning agent. The optimization traces serve as auditable logs, offering a path toward transparent automated planning.
Hybrid Student-Teacher Large Language Model Refinement for Cancer Toxicity Symptom Extraction
Aug 08, 2024


Abstract:Large Language Models (LLMs) offer significant potential for clinical symptom extraction, but their deployment in healthcare settings is constrained by privacy concerns, computational limitations, and operational costs. This study investigates the optimization of compact LLMs for cancer toxicity symptom extraction using a novel iterative refinement approach. We employ a student-teacher architecture, utilizing Zephyr-7b-beta and Phi3-mini-128 as student models and GPT-4o as the teacher, to dynamically select between prompt refinement, Retrieval-Augmented Generation (RAG), and fine-tuning strategies. Our experiments on 294 clinical notes covering 12 post-radiotherapy toxicity symptoms demonstrate the effectiveness of this approach. The RAG method proved most efficient, improving average accuracy scores from 0.32 to 0.73 for Zephyr-7b-beta and from 0.40 to 0.87 for Phi3-mini-128 during refinement. In the test set, both models showed an approximate 0.20 increase in accuracy across symptoms. Notably, this improvement was achieved at a cost 45 times lower than GPT-4o for Zephyr and 79 times lower for Phi-3. These results highlight the potential of iterative refinement techniques in enhancing the capabilities of compact LLMs for clinical applications, offering a balance between performance, cost-effectiveness, and privacy preservation in healthcare settings.
Iterative Prompt Refinement for Radiation Oncology Symptom Extraction Using Teacher-Student Large Language Models
Feb 06, 2024Abstract:This study introduces a novel teacher-student architecture utilizing Large Language Models (LLMs) to improve prostate cancer radiotherapy symptom extraction from clinical notes. Mixtral, the student model, initially extracts symptoms, followed by GPT-4, the teacher model, which refines prompts based on Mixtral's performance. This iterative process involved 294 single symptom clinical notes across 12 symptoms, with up to 16 rounds of refinement per epoch. Results showed significant improvements in extracting symptoms from both single and multi-symptom notes. For 59 single symptom notes, accuracy increased from 0.51 to 0.71, precision from 0.52 to 0.82, recall from 0.52 to 0.72, and F1 score from 0.49 to 0.73. In 375 multi-symptom notes, accuracy rose from 0.24 to 0.43, precision from 0.6 to 0.76, recall from 0.24 to 0.43, and F1 score from 0.20 to 0.44. These results demonstrate the effectiveness of advanced prompt engineering in LLMs for radiation oncology use.
An Introduction to Natural Language Processing Techniques and Framework for Clinical Implementation in Radiation Oncology
Nov 08, 2023

Abstract:Natural Language Processing (NLP) is a key technique for developing Medical Artificial Intelligence (AI) systems that leverage Electronic Health Record (EHR) data to build diagnostic and prognostic models. NLP enables the conversion of unstructured clinical text into structured data that can be fed into AI algorithms. The emergence of the transformer architecture and large language models (LLMs) has led to remarkable advances in NLP for various healthcare tasks, such as entity recognition, relation extraction, sentence similarity, text summarization, and question answering. In this article, we review the major technical innovations that underpin modern NLP models and present state-of-the-art NLP applications that employ LLMs in radiation oncology research. However, these LLMs are prone to many errors such as hallucinations, biases, and ethical violations, which necessitate rigorous evaluation and validation before clinical deployment. As such, we propose a comprehensive framework for assessing the NLP models based on their purpose and clinical fit, technical performance, bias and trust, legal and ethical implications, and quality assurance, prior to implementation in clinical radiation oncology. Our article aims to provide guidance and insights for researchers and clinicians who are interested in developing and using NLP models in clinical radiation oncology.
Prostate Cancer Malignancy Detection and localization from mpMRI using auto-Deep Learning: One Step Closer to Clinical Utilization
Jun 13, 2022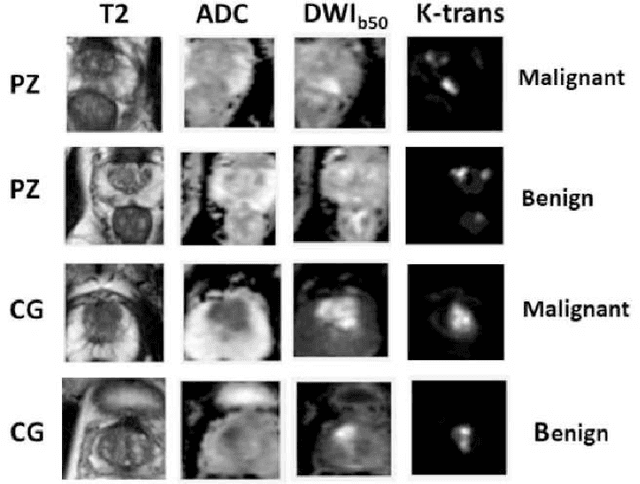

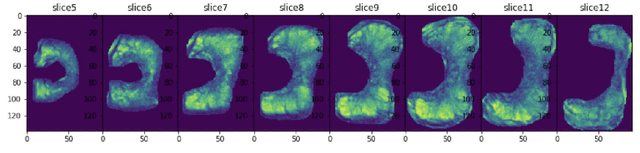

Abstract:Automatic diagnosis of malignant prostate cancer patients from mpMRI has been studied heavily in the past years. Model interpretation and domain drift have been the main road blocks for clinical utilization. As an extension from our previous work where we trained a customized convolutional neural network on a public cohort with 201 patients and the cropped 2D patches around the region of interest were used as the input, the cropped 2.5D slices of the prostate glands were used as the input, and the optimal model were searched in the model space using autoKeras. Something different was peripheral zone (PZ) and central gland (CG) were trained and tested separately, the PZ detector and CG detector were demonstrated effectively in highlighting the most suspicious slices out of a sequence, hopefully to greatly ease the workload for the physicians.
A Deep Dive into Understanding Tumor Foci Classification using Multiparametric MRI Based on Convolutional Neural Network
Apr 04, 2019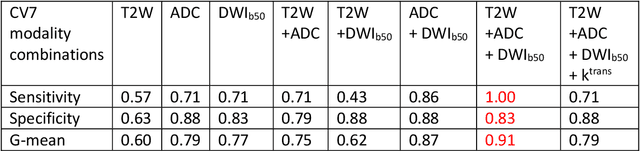
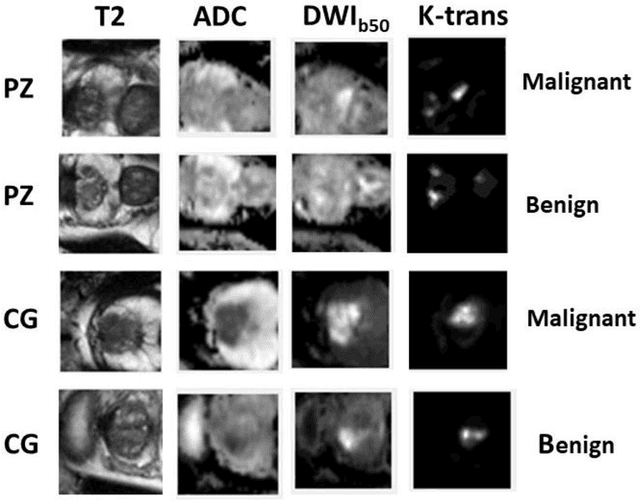
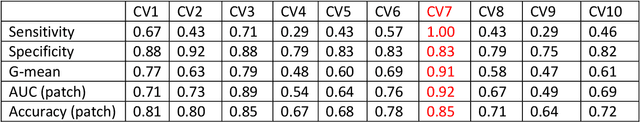
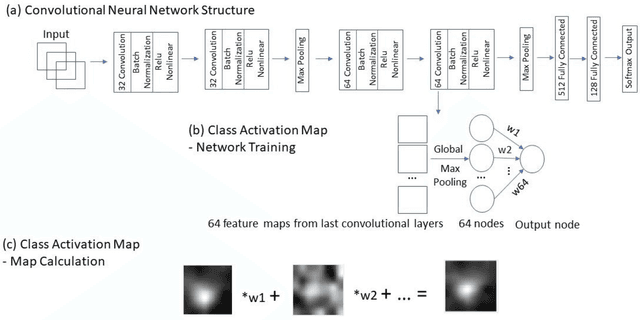
Abstract:Data scarcity has refrained deep learning models from making greater progress in prostate images analysis using multiparametric MRI. In this paper, an efficient convolutional neural network (CNN) was developed to classify lesion malignancy for prostate cancer patients, based on which model interpretation was systematically analyzed to bridge the gap between natural images and MR images, which is the first one of its kind in the literature. The problem of small sample size was addressed and successfully tackled by feeding the intermediate features into a traditional classification algorithm known as weighted extreme learning machine, with imbalanced distribution among output categories taken into consideration. Model trained on public data set was able to generalize to data from an independent institution to make accurate prediction. The generated saliency map was found to overlay well with the lesion and could benefit clinicians for diagnosing purpose.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge