Ahmed I. Ghanem
Modality-AGnostic Image Cascade (MAGIC) for Multi-Modality Cardiac Substructure Segmentation
Jun 12, 2025Abstract:Cardiac substructures are essential in thoracic radiation therapy planning to minimize risk of radiation-induced heart disease. Deep learning (DL) offers efficient methods to reduce contouring burden but lacks generalizability across different modalities and overlapping structures. This work introduces and validates a Modality-AGnostic Image Cascade (MAGIC) for comprehensive and multi-modal cardiac substructure segmentation. MAGIC is implemented through replicated encoding and decoding branches of an nnU-Net-based, U-shaped backbone conserving the function of a single model. Twenty cardiac substructures (heart, chambers, great vessels (GVs), valves, coronary arteries (CAs), and conduction nodes) from simulation CT (Sim-CT), low-field MR-Linac, and cardiac CT angiography (CCTA) modalities were manually delineated and used to train (n=76), validate (n=15), and test (n=30) MAGIC. Twelve comparison models (four segmentation subgroups across three modalities) were equivalently trained. All methods were compared for training efficiency and against reference contours using the Dice Similarity Coefficient (DSC) and two-tailed Wilcoxon Signed-Rank test (threshold, p<0.05). Average DSC scores were 0.75(0.16) for Sim-CT, 0.68(0.21) for MR-Linac, and 0.80(0.16) for CCTA. MAGIC outperforms the comparison in 57% of cases, with limited statistical differences. MAGIC offers an effective and accurate segmentation solution that is lightweight and capable of segmenting multiple modalities and overlapping structures in a single model. MAGIC further enables clinical implementation by simplifying the computational requirements and offering unparalleled flexibility for clinical settings.
Hybrid Student-Teacher Large Language Model Refinement for Cancer Toxicity Symptom Extraction
Aug 08, 2024


Abstract:Large Language Models (LLMs) offer significant potential for clinical symptom extraction, but their deployment in healthcare settings is constrained by privacy concerns, computational limitations, and operational costs. This study investigates the optimization of compact LLMs for cancer toxicity symptom extraction using a novel iterative refinement approach. We employ a student-teacher architecture, utilizing Zephyr-7b-beta and Phi3-mini-128 as student models and GPT-4o as the teacher, to dynamically select between prompt refinement, Retrieval-Augmented Generation (RAG), and fine-tuning strategies. Our experiments on 294 clinical notes covering 12 post-radiotherapy toxicity symptoms demonstrate the effectiveness of this approach. The RAG method proved most efficient, improving average accuracy scores from 0.32 to 0.73 for Zephyr-7b-beta and from 0.40 to 0.87 for Phi3-mini-128 during refinement. In the test set, both models showed an approximate 0.20 increase in accuracy across symptoms. Notably, this improvement was achieved at a cost 45 times lower than GPT-4o for Zephyr and 79 times lower for Phi-3. These results highlight the potential of iterative refinement techniques in enhancing the capabilities of compact LLMs for clinical applications, offering a balance between performance, cost-effectiveness, and privacy preservation in healthcare settings.
An Introduction to Natural Language Processing Techniques and Framework for Clinical Implementation in Radiation Oncology
Nov 08, 2023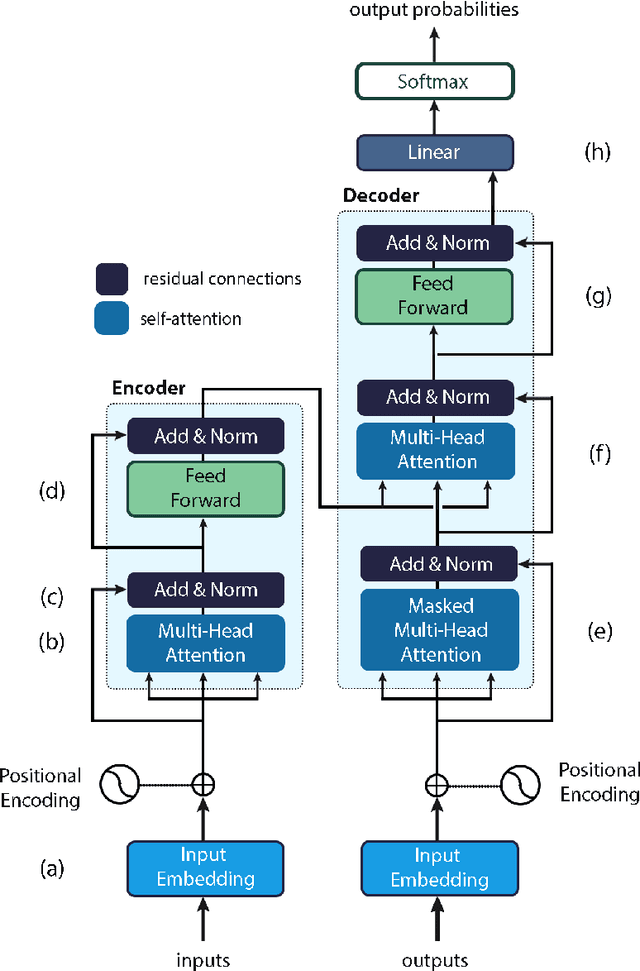

Abstract:Natural Language Processing (NLP) is a key technique for developing Medical Artificial Intelligence (AI) systems that leverage Electronic Health Record (EHR) data to build diagnostic and prognostic models. NLP enables the conversion of unstructured clinical text into structured data that can be fed into AI algorithms. The emergence of the transformer architecture and large language models (LLMs) has led to remarkable advances in NLP for various healthcare tasks, such as entity recognition, relation extraction, sentence similarity, text summarization, and question answering. In this article, we review the major technical innovations that underpin modern NLP models and present state-of-the-art NLP applications that employ LLMs in radiation oncology research. However, these LLMs are prone to many errors such as hallucinations, biases, and ethical violations, which necessitate rigorous evaluation and validation before clinical deployment. As such, we propose a comprehensive framework for assessing the NLP models based on their purpose and clinical fit, technical performance, bias and trust, legal and ethical implications, and quality assurance, prior to implementation in clinical radiation oncology. Our article aims to provide guidance and insights for researchers and clinicians who are interested in developing and using NLP models in clinical radiation oncology.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge