Isaac Galatzer-Levy
Passive Measurement of Autonomic Arousal in Real-World Settings
Apr 30, 2025Abstract:The autonomic nervous system (ANS) is activated during stress, which can have negative effects on cardiovascular health, sleep, the immune system, and mental health. While there are ways to quantify ANS activity in laboratories, there is a paucity of methods that have been validated in real-world contexts. We present the Fitbit Body Response Algorithm, an approach to continuous remote measurement of ANS activation through widely available remote wrist-based sensors. The design was validated via two experiments, a Trier Social Stress Test (n = 45) and ecological momentary assessments (EMA) of perceived stress (n=87), providing both controlled and ecologically valid test data. Model performance predicting perceived stress when using all available sensor modalities was consistent with expectations (accuracy=0.85) and outperformed models with access to only a subset of the signals. We discuss and address challenges to sensing that arise in real world settings that do not present in conventional lab environments.
Substance over Style: Evaluating Proactive Conversational Coaching Agents
Mar 25, 2025



Abstract:While NLP research has made strides in conversational tasks, many approaches focus on single-turn responses with well-defined objectives or evaluation criteria. In contrast, coaching presents unique challenges with initially undefined goals that evolve through multi-turn interactions, subjective evaluation criteria, mixed-initiative dialogue. In this work, we describe and implement five multi-turn coaching agents that exhibit distinct conversational styles, and evaluate them through a user study, collecting first-person feedback on 155 conversations. We find that users highly value core functionality, and that stylistic components in absence of core components are viewed negatively. By comparing user feedback with third-person evaluations from health experts and an LM, we reveal significant misalignment across evaluation approaches. Our findings provide insights into design and evaluation of conversational coaching agents and contribute toward improving human-centered NLP applications.
Sleepless Nights, Sugary Days: Creating Synthetic Users with Health Conditions for Realistic Coaching Agent Interactions
Feb 18, 2025Abstract:We present an end-to-end framework for generating synthetic users for evaluating interactive agents designed to encourage positive behavior changes, such as in health and lifestyle coaching. The synthetic users are grounded in health and lifestyle conditions, specifically sleep and diabetes management in this study, to ensure realistic interactions with the health coaching agent. Synthetic users are created in two stages: first, structured data are generated grounded in real-world health and lifestyle factors in addition to basic demographics and behavioral attributes; second, full profiles of the synthetic users are developed conditioned on the structured data. Interactions between synthetic users and the coaching agent are simulated using generative agent-based models such as Concordia, or directly by prompting a language model. Using two independently-developed agents for sleep and diabetes coaching as case studies, the validity of this framework is demonstrated by analyzing the coaching agent's understanding of the synthetic users' needs and challenges. Finally, through multiple blinded evaluations of user-coach interactions by human experts, we demonstrate that our synthetic users with health and behavioral attributes more accurately portray real human users with the same attributes, compared to generic synthetic users not grounded in such attributes. The proposed framework lays the foundation for efficient development of conversational agents through extensive, realistic, and grounded simulated interactions.
Large Language Models are Few-Shot Health Learners
May 24, 2023
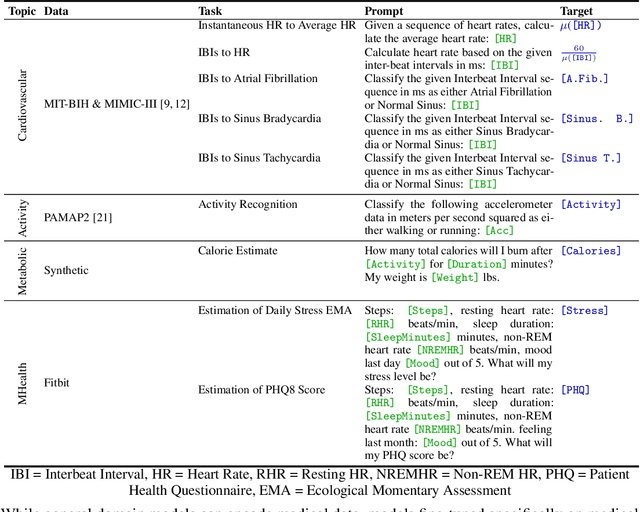

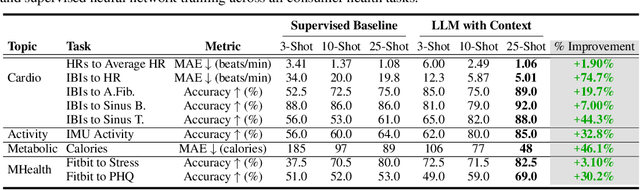
Abstract:Large language models (LLMs) can capture rich representations of concepts that are useful for real-world tasks. However, language alone is limited. While existing LLMs excel at text-based inferences, health applications require that models be grounded in numerical data (e.g., vital signs, laboratory values in clinical domains; steps, movement in the wellness domain) that is not easily or readily expressed as text in existing training corpus. We demonstrate that with only few-shot tuning, a large language model is capable of grounding various physiological and behavioral time-series data and making meaningful inferences on numerous health tasks for both clinical and wellness contexts. Using data from wearable and medical sensor recordings, we evaluate these capabilities on the tasks of cardiac signal analysis, physical activity recognition, metabolic calculation (e.g., calories burned), and estimation of stress reports and mental health screeners.
Prediction of clinical tremor severity using Rank Consistent Ordinal Regression
May 03, 2021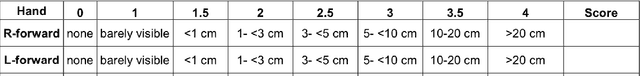
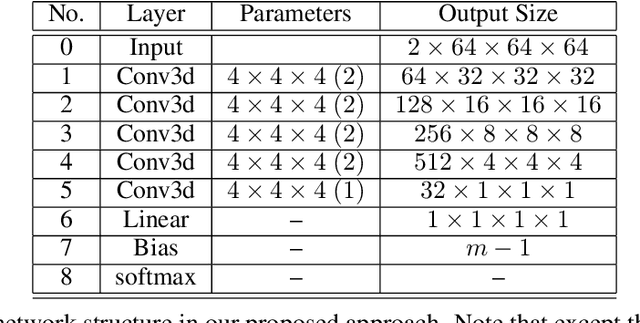
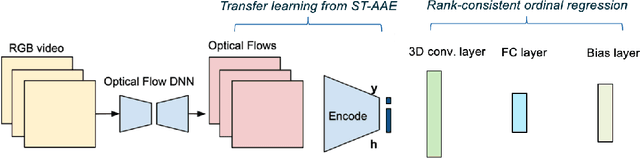

Abstract:Tremor is a key diagnostic feature of Parkinson's Disease (PD), Essential Tremor (ET), and other central nervous system (CNS) disorders. Clinicians or trained raters assess tremor severity with TETRAS scores by observing patients. Lacking quantitative measures, inter- or intra- observer variabilities are almost inevitable as the distinction between adjacent tremor scores is subtle. Moreover, clinician assessments also require patient visits, which limits the frequency of disease progress evaluation. Therefore it is beneficial to develop an automated assessment that can be performed remotely and repeatably at patients' convenience for continuous monitoring. In this work, we proposed to train a deep neural network (DNN) with rank-consistent ordinal regression using 276 clinical videos from 36 essential tremor patients. The videos are coupled with clinician assessed TETRAS scores, which are used as ground truth labels to train the DNN. To tackle the challenge of limited training data, optical flows are used to eliminate irrelevant background and statistic objects from RGB frames. In addition to optical flows, transfer learning is also applied to leverage pre-trained network weights from a related task of tremor frequency estimate. The approach was evaluated by splitting the clinical videos into training (67%) and testing sets (0.33%). The mean absolute error on TETRAS score of the testing results is 0.45, indicating that most of the errors were from the mismatch of adjacent labels, which is expected and acceptable. The model predications also agree well with clinical ratings. This model is further applied to smart phone videos collected from a PD patient who has an implanted device to turn "On" or "Off" tremor. The model outputs were consistent with the patient tremor states. The results demonstrate that our trained model can be used as a means to assess and track tremor severity.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge