Binfeng Zou
Toward Robust Early Detection of Alzheimer's Disease via an Integrated Multimodal Learning Approach
Aug 29, 2024


Abstract:Alzheimer's Disease (AD) is a complex neurodegenerative disorder marked by memory loss, executive dysfunction, and personality changes. Early diagnosis is challenging due to subtle symptoms and varied presentations, often leading to misdiagnosis with traditional unimodal diagnostic methods due to their limited scope. This study introduces an advanced multimodal classification model that integrates clinical, cognitive, neuroimaging, and EEG data to enhance diagnostic accuracy. The model incorporates a feature tagger with a tabular data coding architecture and utilizes the TimesBlock module to capture intricate temporal patterns in Electroencephalograms (EEG) data. By employing Cross-modal Attention Aggregation module, the model effectively fuses Magnetic Resonance Imaging (MRI) spatial information with EEG temporal data, significantly improving the distinction between AD, Mild Cognitive Impairment, and Normal Cognition. Simultaneously, we have constructed the first AD classification dataset that includes three modalities: EEG, MRI, and tabular data. Our innovative approach aims to facilitate early diagnosis and intervention, potentially slowing the progression of AD. The source code and our private ADMC dataset are available at https://github.com/JustlfC03/MSTNet.
GFE-Mamba: Mamba-based AD Multi-modal Progression Assessment via Generative Feature Extraction from MCI
Jul 22, 2024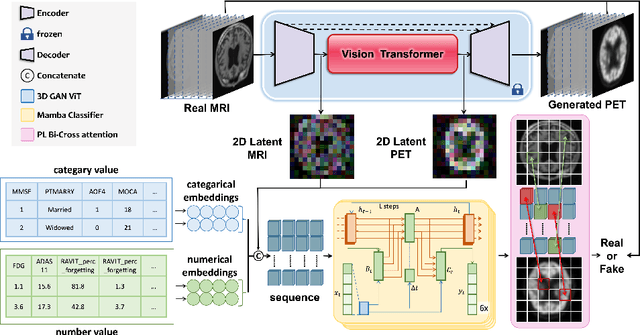



Abstract:Alzheimer's Disease (AD) is an irreversible neurodegenerative disorder that often progresses from Mild Cognitive Impairment (MCI), leading to memory loss and significantly impacting patients' lives. Clinical trials indicate that early targeted interventions for MCI patients can potentially slow or halt the development and progression of AD. Previous research has shown that accurate medical classification requires the inclusion of extensive multimodal data, such as assessment scales and various neuroimaging techniques like Magnetic Resonance Imaging (MRI) and Positron Emission Tomography (PET). However, consistently tracking the diagnosis of the same individual over time and simultaneously collecting multimodal data poses significant challenges. To address this issue, we introduce GFE-Mamba, a classifier based on Generative Feature Extraction (GFE). This classifier effectively integrates data from assessment scales, MRI, and PET, enabling deeper multimodal fusion. It efficiently extracts both long and short sequence information and incorporates additional information beyond the pixel space. This approach not only improves classification accuracy but also enhances the interpretability and stability of the model. We constructed datasets of over 3000 samples based on the Alzheimer's Disease Neuroimaging Initiative (ADNI) for a two-step training process. Our experimental results demonstrate that the GFE-Mamba model is effective in predicting the conversion from MCI to AD and outperforms several state-of-the-art methods. Our source code and ADNI dataset processing code are available at https://github.com/Tinysqua/GFE-Mamba.
SCKansformer: Fine-Grained Classification of Bone Marrow Cells via Kansformer Backbone and Hierarchical Attention Mechanisms
Jun 14, 2024



Abstract:The incidence and mortality rates of malignant tumors, such as acute leukemia, have risen significantly. Clinically, hospitals rely on cytological examination of peripheral blood and bone marrow smears to diagnose malignant tumors, with accurate blood cell counting being crucial. Existing automated methods face challenges such as low feature expression capability, poor interpretability, and redundant feature extraction when processing high-dimensional microimage data. We propose a novel fine-grained classification model, SCKansformer, for bone marrow blood cells, which addresses these challenges and enhances classification accuracy and efficiency. The model integrates the Kansformer Encoder, SCConv Encoder, and Global-Local Attention Encoder. The Kansformer Encoder replaces the traditional MLP layer with the KAN, improving nonlinear feature representation and interpretability. The SCConv Encoder, with its Spatial and Channel Reconstruction Units, enhances feature representation and reduces redundancy. The Global-Local Attention Encoder combines Multi-head Self-Attention with a Local Part module to capture both global and local features. We validated our model using the Bone Marrow Blood Cell Fine-Grained Classification Dataset (BMCD-FGCD), comprising over 10,000 samples and nearly 40 classifications, developed with a partner hospital. Comparative experiments on our private dataset, as well as the publicly available PBC and ALL-IDB datasets, demonstrate that SCKansformer outperforms both typical and advanced microcell classification methods across all datasets. Our source code and private BMCD-FGCD dataset are available at https://github.com/JustlfC03/SCKansformer.
SCUNet++: Swin-UNet and CNN Bottleneck Hybrid Architecture with Multi-Fusion Dense Skip Connection for Pulmonary Embolism CT Image Segmentation
Jan 03, 2024Abstract:Pulmonary embolism (PE) is a prevalent lung disease that can lead to right ventricular hypertrophy and failure in severe cases, ranking second in severity only to myocardial infarction and sudden death. Pulmonary artery CT angiography (CTPA) is a widely used diagnostic method for PE. However, PE detection presents challenges in clinical practice due to limitations in imaging technology. CTPA can produce noises similar to PE, making confirmation of its presence time-consuming and prone to overdiagnosis. Nevertheless, the traditional segmentation method of PE can not fully consider the hierarchical structure of features, local and global spatial features of PE CT images. In this paper, we propose an automatic PE segmentation method called SCUNet++ (Swin Conv UNet++). This method incorporates multiple fusion dense skip connections between the encoder and decoder, utilizing the Swin Transformer as the encoder. And fuses features of different scales in the decoder subnetwork to compensate for spatial information loss caused by the inevitable downsampling in Swin-UNet or other state-of-the-art methods, effectively solving the above problem. We provide a theoretical analysis of this method in detail and validate it on publicly available PE CT image datasets FUMPE and CAD-PE. The experimental results indicate that our proposed method achieved a Dice similarity coefficient (DSC) of 83.47% and a Hausdorff distance 95th percentile (HD95) of 3.83 on the FUMPE dataset, as well as a DSC of 83.42% and an HD95 of 5.10 on the CAD-PE dataset. These findings demonstrate that our method exhibits strong performance in PE segmentation tasks, potentially enhancing the accuracy of automatic segmentation of PE and providing a powerful diagnostic tool for clinical physicians. Our source code and new FUMPE dataset are available at https://github.com/JustlfC03/SCUNet-plusplus.
* 10 pages, 7 figures, accept WACV2024
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge