Shadab Ahamed
Preconditioned Score and Flow Matching
Mar 02, 2026Abstract:Flow matching and score-based diffusion train vector fields under intermediate distributions $p_t$, whose geometry can strongly affect their optimization. We show that the covariance $Σ_t$ of $p_t$ governs optimization bias: when $Σ_t$ is ill-conditioned, and gradient-based training rapidly fits high-variance directions while systematically under-optimizing low-variance modes, leading to learning that plateaus at suboptimal weights. We formalize this effect in analytically tractable settings and propose reversible, label-conditional \emph{preconditioning} maps that reshape the geometry of $p_t$ by improving the conditioning of $Σ_t$ without altering the underlying generative model. Rather than accelerating early convergence, preconditioning primarily mitigates optimization stagnation by enabling continued progress along previously suppressed directions. Across MNIST latent flow matching, and additional high-resolution datasets, we empirically track conditioning diagnostics and distributional metrics and show that preconditioning consistently yields better-trained models by avoiding suboptimal plateaus.
Comprehensive Evaluation of Quantitative Measurements from Automated Deep Segmentations of PSMA PET/CT Images
Apr 22, 2025Abstract:This study performs a comprehensive evaluation of quantitative measurements as extracted from automated deep-learning-based segmentation methods, beyond traditional Dice Similarity Coefficient assessments, focusing on six quantitative metrics, namely SUVmax, SUVmean, total lesion activity (TLA), tumor volume (TMTV), lesion count, and lesion spread. We analyzed 380 prostate-specific membrane antigen (PSMA) targeted [18F]DCFPyL PET/CT scans of patients with biochemical recurrence of prostate cancer, training deep neural networks, U-Net, Attention U-Net and SegResNet with four loss functions: Dice Loss, Dice Cross Entropy, Dice Focal Loss, and our proposed L1 weighted Dice Focal Loss (L1DFL). Evaluations indicated that Attention U-Net paired with L1DFL achieved the strongest correlation with the ground truth (concordance correlation = 0.90-0.99 for SUVmax and TLA), whereas models employing the Dice Loss and the other two compound losses, particularly with SegResNet, underperformed. Equivalence testing (TOST, alpha = 0.05, Delta = 20%) confirmed high performance for SUV metrics, lesion count and TLA, with L1DFL yielding the best performance. By contrast, tumor volume and lesion spread exhibited greater variability. Bland-Altman, Coverage Probability, and Total Deviation Index analyses further highlighted that our proposed L1DFL minimizes variability in quantification of the ground truth clinical measures. The code is publicly available at: https://github.com/ObedDzik/pca\_segment.git.
Iterative Flow Matching -- Path Correction and Gradual Refinement for Enhanced Generative Modeling
Feb 26, 2025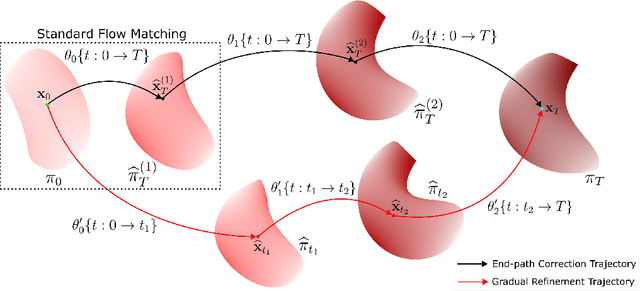
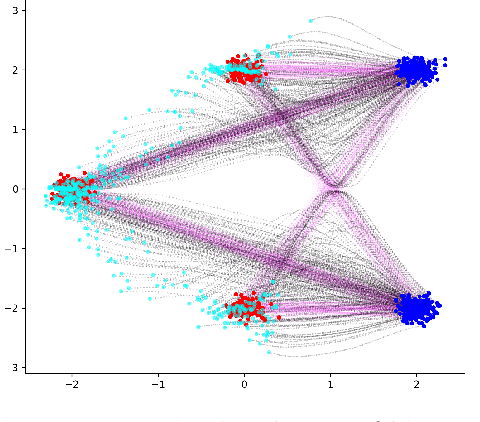
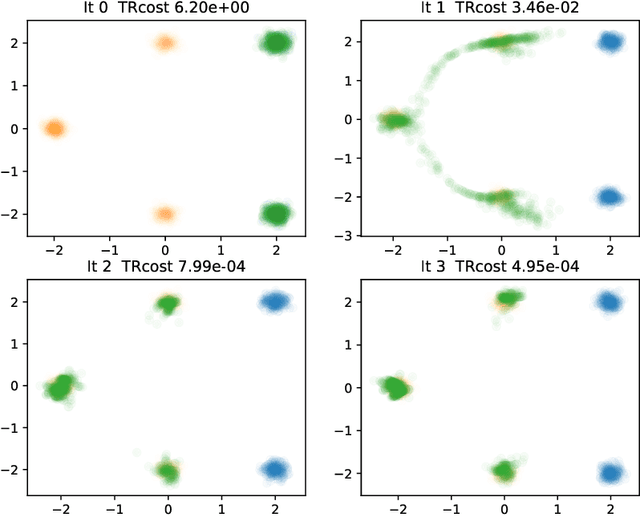
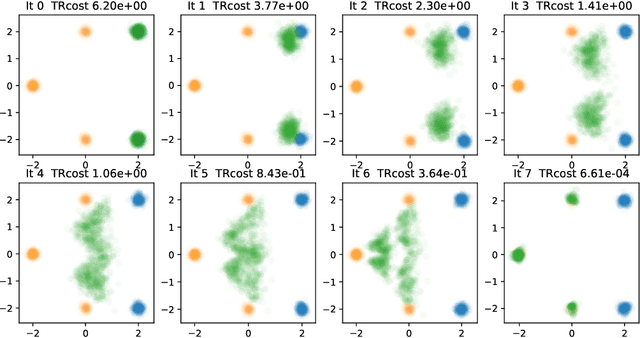
Abstract:Generative models for image generation are now commonly used for a wide variety of applications, ranging from guided image generation for entertainment to solving inverse problems. Nonetheless, training a generator is a non-trivial feat that requires fine-tuning and can lead to so-called hallucinations, that is, the generation of images that are unrealistic. In this work, we explore image generation using flow matching. We explain and demonstrate why flow matching can generate hallucinations, and propose an iterative process to improve the generation process. Our iterative process can be integrated into virtually $\textit{any}$ generative modeling technique, thereby enhancing the performance and robustness of image synthesis systems.
Inversion of Magnetic Data using Learned Dictionaries and Scale Space
Feb 08, 2025
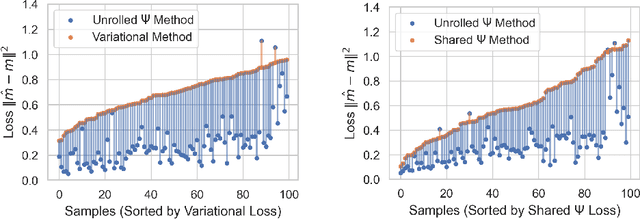

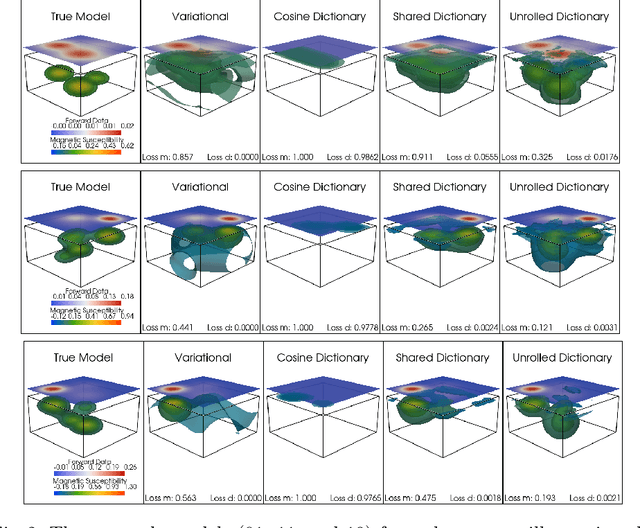
Abstract:Magnetic data inversion is an important tool in geophysics, used to infer subsurface magnetic susceptibility distributions from surface magnetic field measurements. This inverse problem is inherently ill-posed, characterized by non-unique solutions, depth ambiguity, and sensitivity to noise. Traditional inversion approaches rely on predefined regularization techniques to stabilize solutions, limiting their adaptability to complex or diverse geological scenarios. In this study, we propose an approach that integrates variable dictionary learning and scale-space methods to address these challenges. Our method employs learned dictionaries, allowing for adaptive representation of complex subsurface features that are difficult to capture with predefined bases. Additionally, we extend classical variational inversion by incorporating multi-scale representations through a scale-space framework, enabling the progressive introduction of structural detail while mitigating overfitting. We implement both fixed and dynamic dictionary learning techniques, with the latter introducing iteration-dependent dictionaries for enhanced flexibility. Using a synthetic dataset to simulate geological scenarios, we demonstrate significant improvements in reconstruction accuracy and robustness compared to conventional variational and dictionary-based methods. Our results highlight the potential of learned dictionaries, especially when coupled with scale-space dynamics, to improve model recovery and noise handling. These findings underscore the promise of our data-driven approach for advance magnetic data inversion and its applications in geophysical exploration, environmental assessment, and mineral prospecting.
Adaptive Voxel-Weighted Loss Using L1 Norms in Deep Neural Networks for Detection and Segmentation of Prostate Cancer Lesions in PET/CT Images
Feb 04, 2025



Abstract:This study proposes a new loss function for deep neural networks, L1-weighted Dice Focal Loss (L1DFL), that leverages L1 norms for adaptive weighting of voxels based on their classification difficulty, towards automated detection and segmentation of metastatic prostate cancer lesions in PET/CT scans. We obtained 380 PSMA [18-F] DCFPyL PET/CT scans of patients diagnosed with biochemical recurrence metastatic prostate cancer. We trained two 3D convolutional neural networks, Attention U-Net and SegResNet, and concatenated the PET and CT volumes channel-wise as input. The performance of our custom loss function was evaluated against the Dice and Dice Focal Loss functions. For clinical significance, we considered a detected region of interest (ROI) as a true positive if at least the voxel with the maximum standardized uptake value falls within the ROI. We assessed the models' performance based on the number of lesions in an image, tumour volume, activity, and extent of spread. The L1DFL outperformed the comparative loss functions by at least 13% on the test set. In addition, the F1 scores of the Dice Loss and the Dice Focal Loss were lower than that of L1DFL by at least 6% and 34%, respectively. The Dice Focal Loss yielded more false positives, whereas the Dice Loss was more sensitive to smaller volumes and struggled to segment larger lesions accurately. They also exhibited network-specific variations and yielded declines in segmentation accuracy with increased tumour spread. Our results demonstrate the potential of L1DFL to yield robust segmentation of metastatic prostate cancer lesions in PSMA PET/CT images. The results further highlight potential complexities arising from the variations in lesion characteristics that may influence automated prostate cancer tumour detection and segmentation. The code is publicly available at: https://github.com/ObedDzik/pca_segment.git.
DAWN-SI: Data-Aware and Noise-Informed Stochastic Interpolation for Solving Inverse Problems
Dec 06, 2024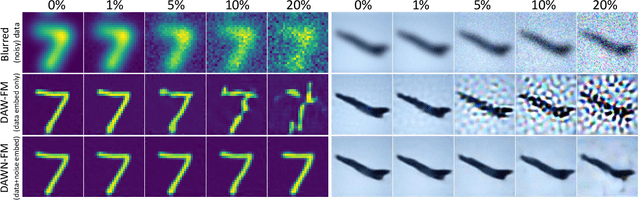
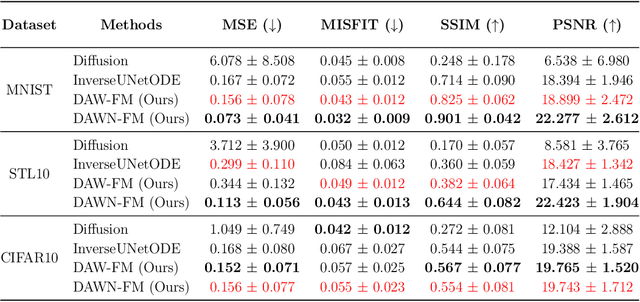
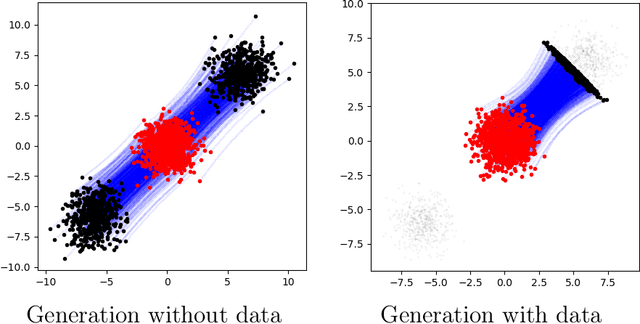
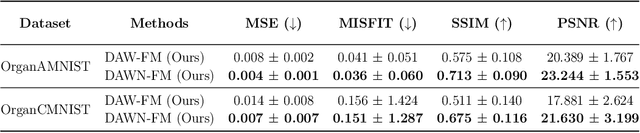
Abstract:Inverse problems, which involve estimating parameters from incomplete or noisy observations, arise in various fields such as medical imaging, geophysics, and signal processing. These problems are often ill-posed, requiring regularization techniques to stabilize the solution. In this work, we employ $\textit{Stochastic Interpolation}$ (SI), a generative framework that integrates both deterministic and stochastic processes to map a simple reference distribution, such as a Gaussian, to the target distribution. Our method $\textbf{DAWN-SI}$: $\textbf{D}$ata-$\textbf{AW}$are and $\textbf{N}$oise-informed $\textbf{S}$tochastic $\textbf{I}$nterpolation incorporates data and noise embedding, allowing the model to access representations about the measured data explicitly and also account for noise in the observations, making it particularly robust in scenarios where data is noisy or incomplete. By learning a time-dependent velocity field, SI not only provides accurate solutions but also enables uncertainty quantification by generating multiple plausible outcomes. Unlike pre-trained diffusion models, which may struggle in highly ill-posed settings, our approach is trained specifically for each inverse problem and adapts to varying noise levels. We validate the effectiveness and robustness of our method through extensive numerical experiments on tasks such as image deblurring and tomography.
AutoPET Challenge III: Testing the Robustness of Generalized Dice Focal Loss trained 3D Residual UNet for FDG and PSMA Lesion Segmentation from Whole-Body PET/CT Images
Sep 16, 2024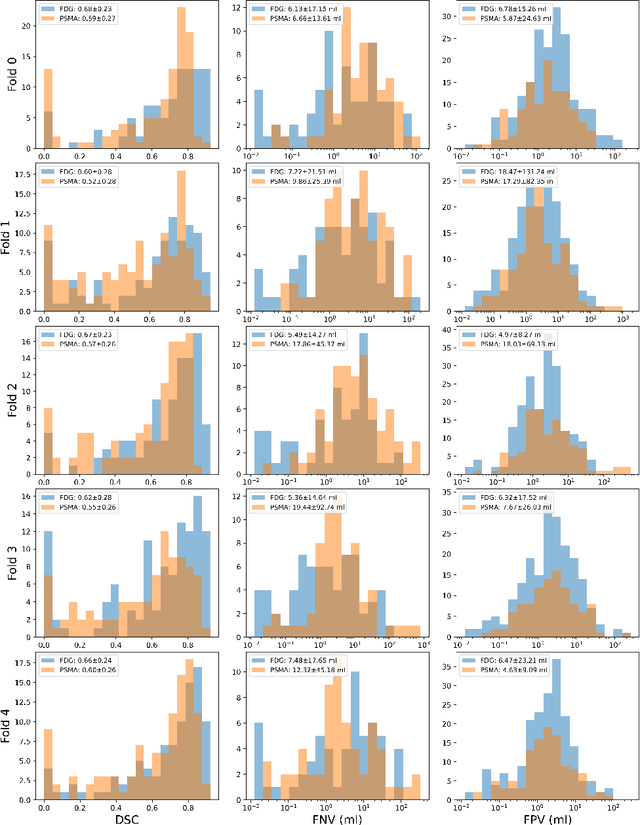
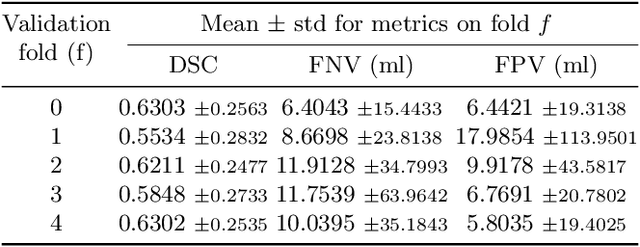

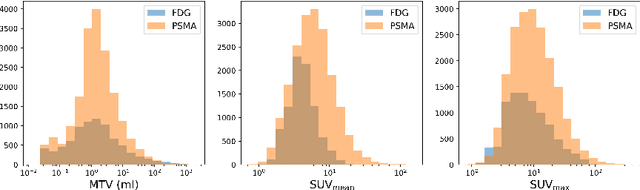
Abstract:Automated segmentation of cancerous lesions in PET/CT scans is a crucial first step in quantitative image analysis. However, training deep learning models for segmentation with high accuracy is particularly challenging due to the variations in lesion size, shape, and radiotracer uptake. These lesions can appear in different parts of the body, often near healthy organs that also exhibit considerable uptake, making the task even more complex. As a result, creating an effective segmentation model for routine PET/CT image analysis is challenging. In this study, we utilized a 3D Residual UNet model and employed the Generalized Dice Focal Loss function to train the model on the AutoPET Challenge 2024 dataset. We conducted a 5-fold cross-validation and used an average ensembling technique using the models from the five folds. In the preliminary test phase for Task-1, the average ensemble achieved a mean Dice Similarity Coefficient (DSC) of 0.6687, mean false negative volume (FNV) of 10.9522 ml and mean false positive volume (FPV) 2.9684 ml. More details about the algorithm can be found on our GitHub repository: https://github.com/ahxmeds/autosegnet2024.git. The training code has been shared via the repository: https://github.com/ahxmeds/autopet2024.git.
Thyroidiomics: An Automated Pipeline for Segmentation and Classification of Thyroid Pathologies from Scintigraphy Images
Jul 14, 2024



Abstract:The objective of this study was to develop an automated pipeline that enhances thyroid disease classification using thyroid scintigraphy images, aiming to decrease assessment time and increase diagnostic accuracy. Anterior thyroid scintigraphy images from 2,643 patients were collected and categorized into diffuse goiter (DG), multinodal goiter (MNG), and thyroiditis (TH) based on clinical reports, and then segmented by an expert. A ResUNet model was trained to perform auto-segmentation. Radiomic features were extracted from both physician (scenario 1) and ResUNet segmentations (scenario 2), followed by omitting highly correlated features using Spearman's correlation, and feature selection using Recursive Feature Elimination (RFE) with XGBoost as the core. All models were trained under leave-one-center-out cross-validation (LOCOCV) scheme, where nine instances of algorithms were iteratively trained and validated on data from eight centers and tested on the ninth for both scenarios separately. Segmentation performance was assessed using the Dice similarity coefficient (DSC), while classification performance was assessed using metrics, such as precision, recall, F1-score, accuracy, area under the Receiver Operating Characteristic (ROC AUC), and area under the precision-recall curve (PRC AUC). ResUNet achieved DSC values of 0.84$\pm$0.03, 0.71$\pm$0.06, and 0.86$\pm$0.02 for MNG, TH, and DG, respectively. Classification in scenario 1 achieved an accuracy of 0.76$\pm$0.04 and a ROC AUC of 0.92$\pm$0.02 while in scenario 2, classification yielded an accuracy of 0.74$\pm$0.05 and a ROC AUC of 0.90$\pm$0.02. The automated pipeline demonstrated comparable performance to physician segmentations on several classification metrics across different classes, effectively reducing assessment time while maintaining high diagnostic accuracy. Code available at: https://github.com/ahxmeds/thyroidiomics.git.
IgCONDA-PET: Implicitly-Guided Counterfactual Diffusion for Detecting Anomalies in PET Images
Apr 30, 2024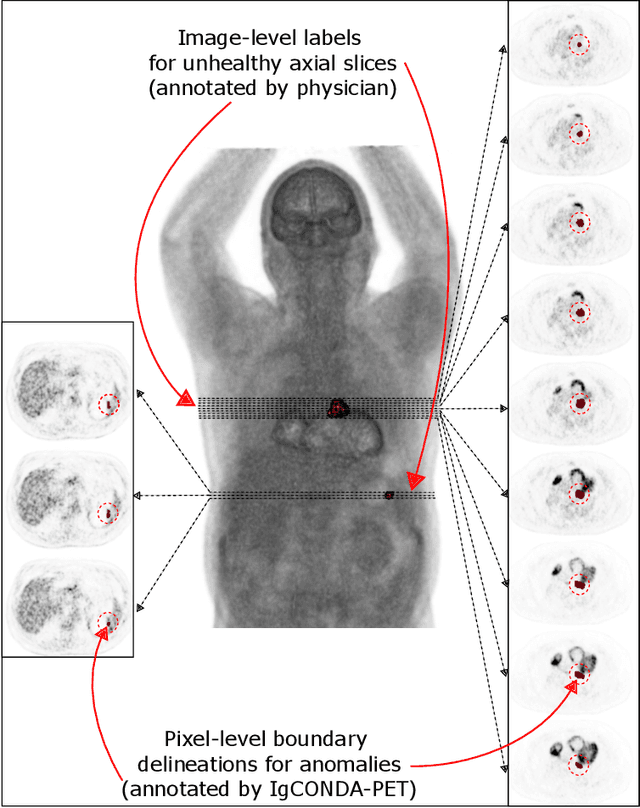
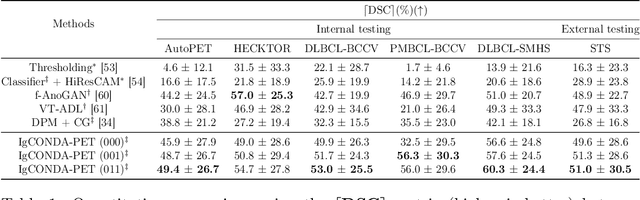
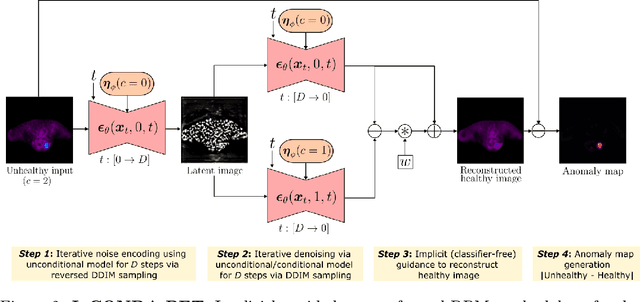
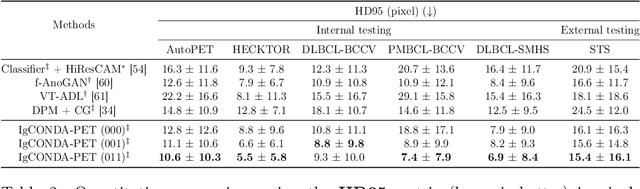
Abstract:Minimizing the need for pixel-level annotated data for training PET anomaly segmentation networks is crucial, particularly due to time and cost constraints related to expert annotations. Current un-/weakly-supervised anomaly detection methods rely on autoencoder or generative adversarial networks trained only on healthy data, although these are more challenging to train. In this work, we present a weakly supervised and Implicitly guided COuNterfactual diffusion model for Detecting Anomalies in PET images, branded as IgCONDA-PET. The training is conditioned on image class labels (healthy vs. unhealthy) along with implicit guidance to generate counterfactuals for an unhealthy image with anomalies. The counterfactual generation process synthesizes the healthy counterpart for a given unhealthy image, and the difference between the two facilitates the identification of anomaly locations. The code is available at: https://github.com/igcondapet/IgCONDA-PET.git
A cascaded deep network for automated tumor detection and segmentation in clinical PET imaging of diffuse large B-cell lymphoma
Mar 11, 2024Abstract:Accurate detection and segmentation of diffuse large B-cell lymphoma (DLBCL) from PET images has important implications for estimation of total metabolic tumor volume, radiomics analysis, surgical intervention and radiotherapy. Manual segmentation of tumors in whole-body PET images is time-consuming, labor-intensive and operator-dependent. In this work, we develop and validate a fast and efficient three-step cascaded deep learning model for automated detection and segmentation of DLBCL tumors from PET images. As compared to a single end-to-end network for segmentation of tumors in whole-body PET images, our three-step model is more effective (improves 3D Dice score from 58.9% to 78.1%) since each of its specialized modules, namely the slice classifier, the tumor detector and the tumor segmentor, can be trained independently to a high degree of skill to carry out a specific task, rather than a single network with suboptimal performance on overall segmentation.
* 8 pages, 3 figures, 3 tables
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge