Lorna Smith
for the ALFA study
Dual Deep Learning Approach for Non-invasive Renal Tumour Subtyping with VERDICT-MRI
Apr 09, 2025Abstract:This work aims to characterise renal tumour microstructure using diffusion MRI (dMRI); via the Vascular, Extracellular and Restricted Diffusion for Cytometry in Tumours (VERDICT)-MRI framework with self-supervised learning. Comprehensive datasets were acquired from 14 patients with 15 biopsy-confirmed renal tumours, with nine b-values in the range b=[0,2500]s/mm2. A three-compartment VERDICT model for renal tumours was fitted to the dMRI data using a self-supervised deep neural network, and ROIs were drawn by an experienced uroradiologist. An economical acquisition protocol for future studies with larger patient cohorts was optimised using a recursive feature selection approach. The VERDICT model described the diffusion data in renal tumours more accurately than IVIM or ADC. Combined with self-supervised deep learning, VERDICT identified significant differences in the intracellular volume fraction between cancerous and normal tissue, and in the vascular volume fraction between vascular and non-vascular. The feature selector yields a 4 b-value acquisition of b = [70,150,1000,2000], with a duration of 14 minutes.
Where is VALDO? VAscular Lesions Detection and segmentatiOn challenge at MICCAI 2021
Aug 15, 2022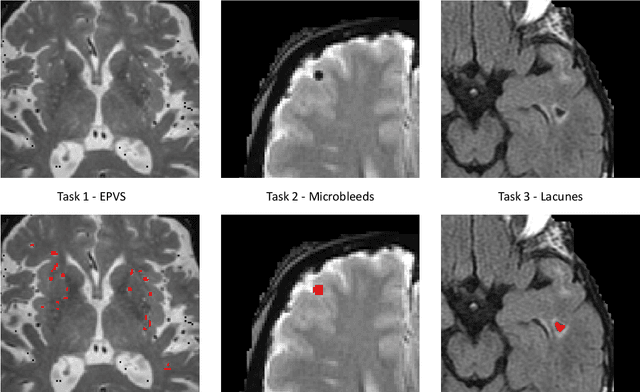
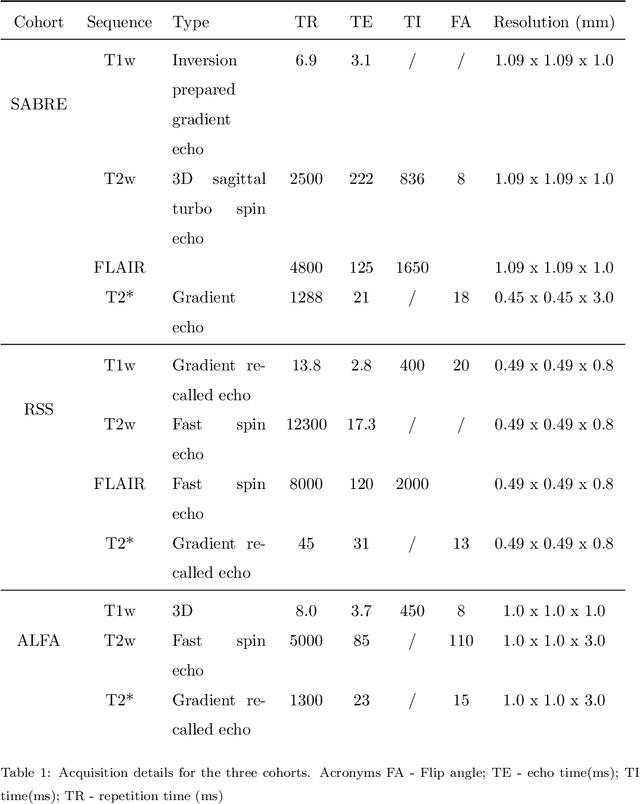
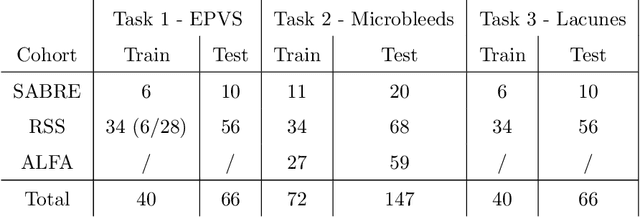
Abstract:Imaging markers of cerebral small vessel disease provide valuable information on brain health, but their manual assessment is time-consuming and hampered by substantial intra- and interrater variability. Automated rating may benefit biomedical research, as well as clinical assessment, but diagnostic reliability of existing algorithms is unknown. Here, we present the results of the \textit{VAscular Lesions DetectiOn and Segmentation} (\textit{Where is VALDO?}) challenge that was run as a satellite event at the international conference on Medical Image Computing and Computer Aided Intervention (MICCAI) 2021. This challenge aimed to promote the development of methods for automated detection and segmentation of small and sparse imaging markers of cerebral small vessel disease, namely enlarged perivascular spaces (EPVS) (Task 1), cerebral microbleeds (Task 2) and lacunes of presumed vascular origin (Task 3) while leveraging weak and noisy labels. Overall, 12 teams participated in the challenge proposing solutions for one or more tasks (4 for Task 1 - EPVS, 9 for Task 2 - Microbleeds and 6 for Task 3 - Lacunes). Multi-cohort data was used in both training and evaluation. Results showed a large variability in performance both across teams and across tasks, with promising results notably for Task 1 - EPVS and Task 2 - Microbleeds and not practically useful results yet for Task 3 - Lacunes. It also highlighted the performance inconsistency across cases that may deter use at an individual level, while still proving useful at a population level.
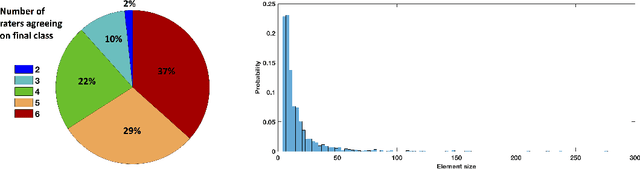
Let's agree to disagree: learning highly debatable multirater labelling
Sep 04, 2019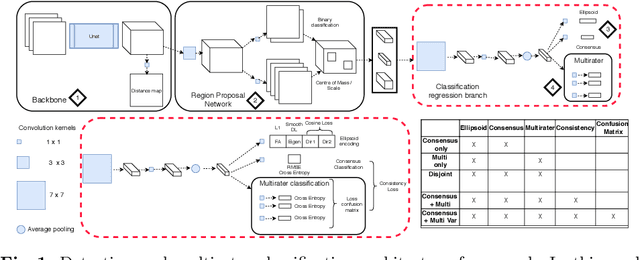
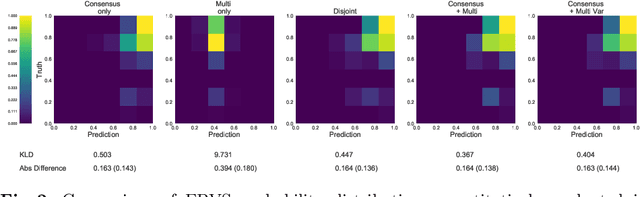
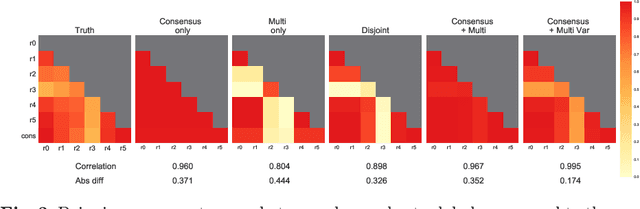
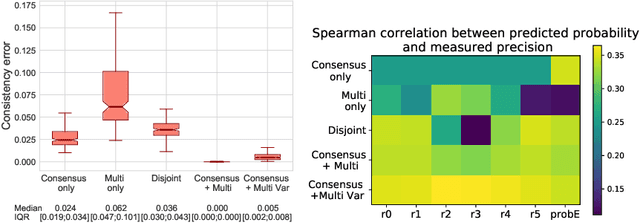
Abstract:Classification and differentiation of small pathological objects may greatly vary among human raters due to differences in training, expertise and their consistency over time. In a radiological setting, objects commonly have high within-class appearance variability whilst sharing certain characteristics across different classes, making their distinction even more difficult. As an example, markers of cerebral small vessel disease, such as enlarged perivascular spaces (EPVS) and lacunes, can be very varied in their appearance while exhibiting high inter-class similarity, making this task highly challenging for human raters. In this work, we investigate joint models of individual rater behaviour and multirater consensus in a deep learning setting, and apply it to a brain lesion object-detection task. Results show that jointly modelling both individual and consensus estimates leads to significant improvements in performance when compared to directly predicting consensus labels, while also allowing the characterization of human-rater consistency.
3D multirater RCNN for multimodal multiclass detection and characterisation of extremely small objects
Dec 21, 2018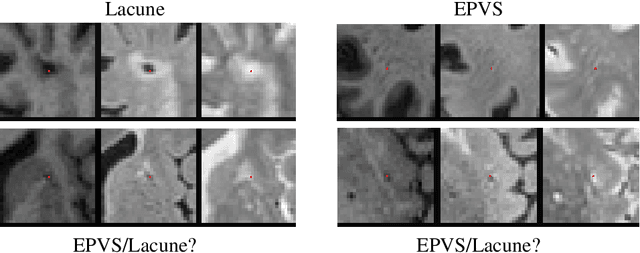
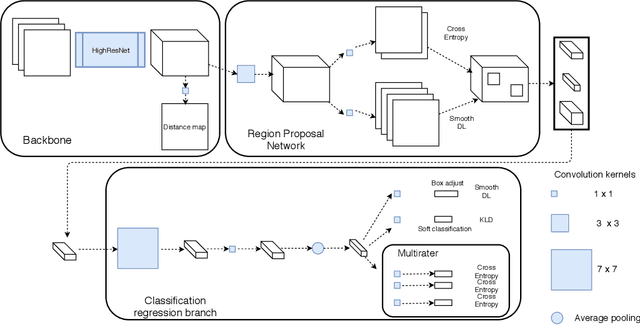
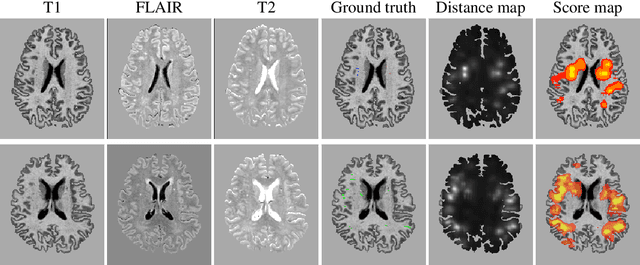
Abstract:Extremely small objects (ESO) have become observable on clinical routine magnetic resonance imaging acquisitions, thanks to a reduction in acquisition time at higher resolution. Despite their small size (usually $<$10 voxels per object for an image of more than $10^6$ voxels), these markers reflect tissue damage and need to be accounted for to investigate the complete phenotype of complex pathological pathways. In addition to their very small size, variability in shape and appearance leads to high labelling variability across human raters, resulting in a very noisy gold standard. Such objects are notably present in the context of cerebral small vessel disease where enlarged perivascular spaces and lacunes, commonly observed in the ageing population, are thought to be associated with acceleration of cognitive decline and risk of dementia onset. In this work, we redesign the RCNN model to scale to 3D data, and to jointly detect and characterise these important markers of age-related neurovascular changes. We also propose training strategies enforcing the detection of extremely small objects, ensuring a tractable and stable training process.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge