Barbaros S. Erdal
for the Alzheimer's Disease Neuroimaging Initiative
MONAI: An open-source framework for deep learning in healthcare
Nov 04, 2022



Abstract:Artificial Intelligence (AI) is having a tremendous impact across most areas of science. Applications of AI in healthcare have the potential to improve our ability to detect, diagnose, prognose, and intervene on human disease. For AI models to be used clinically, they need to be made safe, reproducible and robust, and the underlying software framework must be aware of the particularities (e.g. geometry, physiology, physics) of medical data being processed. This work introduces MONAI, a freely available, community-supported, and consortium-led PyTorch-based framework for deep learning in healthcare. MONAI extends PyTorch to support medical data, with a particular focus on imaging, and provide purpose-specific AI model architectures, transformations and utilities that streamline the development and deployment of medical AI models. MONAI follows best practices for software-development, providing an easy-to-use, robust, well-documented, and well-tested software framework. MONAI preserves the simple, additive, and compositional approach of its underlying PyTorch libraries. MONAI is being used by and receiving contributions from research, clinical and industrial teams from around the world, who are pursuing applications spanning nearly every aspect of healthcare.
Cascading Neural Network Methodology for Artificial Intelligence-Assisted Radiographic Detection and Classification of Lead-Less Implanted Electronic Devices within the Chest
Aug 25, 2021



Abstract:Background & Purpose: Chest X-Ray (CXR) use in pre-MRI safety screening for Lead-Less Implanted Electronic Devices (LLIEDs), easily overlooked or misidentified on a frontal view (often only acquired), is common. Although most LLIED types are "MRI conditional": 1. Some are stringently conditional; 2. Different conditional types have specific patient- or device- management requirements; and 3. Particular types are "MRI unsafe". This work focused on developing CXR interpretation-assisting Artificial Intelligence (AI) methodology with: 1. 100% detection for LLIED presence/location; and 2. High classification in LLIED typing. Materials & Methods: Data-mining (03/1993-02/2021) produced an AI Model Development Population (1,100 patients/4,871 images) creating 4,924 LLIED Region-Of-Interests (ROIs) (with image-quality grading) used in Training, Validation, and Testing. For developing the cascading neural network (detection via Faster R-CNN and classification via Inception V3), "ground-truth" CXR annotation (ROI labeling per LLIED), as well as inference display (as Generated Bounding Boxes (GBBs)), relied on a GPU-based graphical user interface. Results: To achieve 100% LLIED detection, probability threshold reduction to 0.00002 was required by Model 1, resulting in increasing GBBs per LLIED-related ROI. Targeting LLIED-type classification following detection of all LLIEDs, Model 2 multi-classified to reach high-performance while decreasing falsely positive GBBs. Despite 24% suboptimal ROI image quality, classification was correct in 98.9% and AUCs for the 9 LLIED-types were 1.00 for 8 and 0.92 for 1. For all misclassification cases: 1. None involved stringently conditional or unsafe LLIEDs; and 2. Most were attributable to suboptimal images. Conclusion: This project successfully developed a LLIED-related AI methodology supporting: 1. 100% detection; and 2. Typically 100% type classification.
Artificial Intelligence to Assist in Exclusion of Coronary Atherosclerosis during CCTA Evaluation of Chest-Pain in the Emergency Department: Preparing an Application for Real-World Use
Aug 10, 2020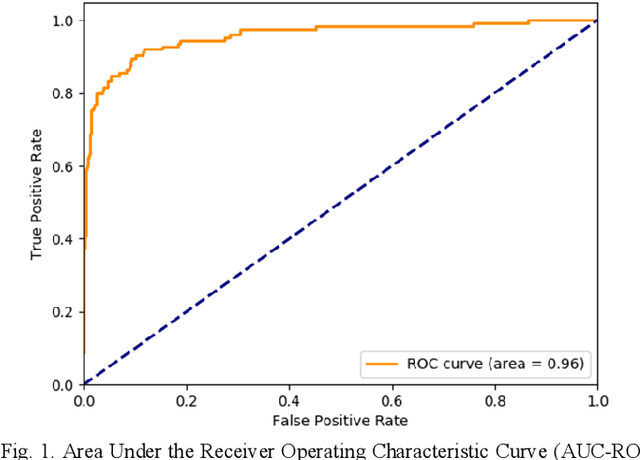
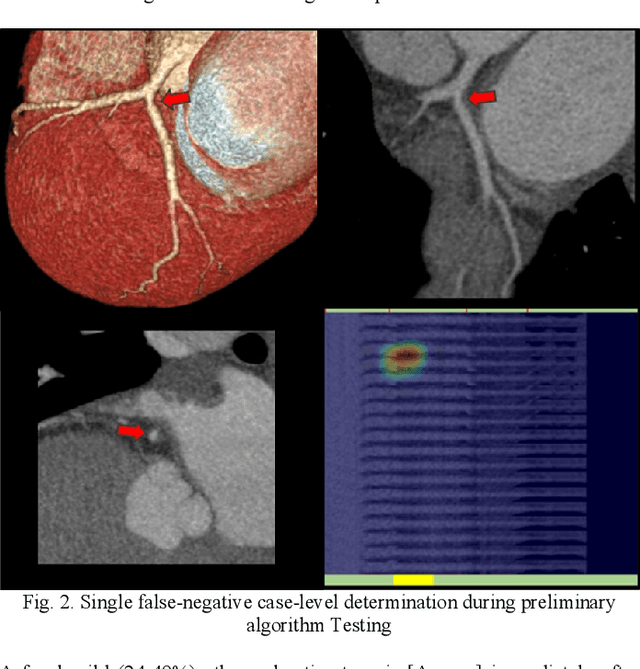
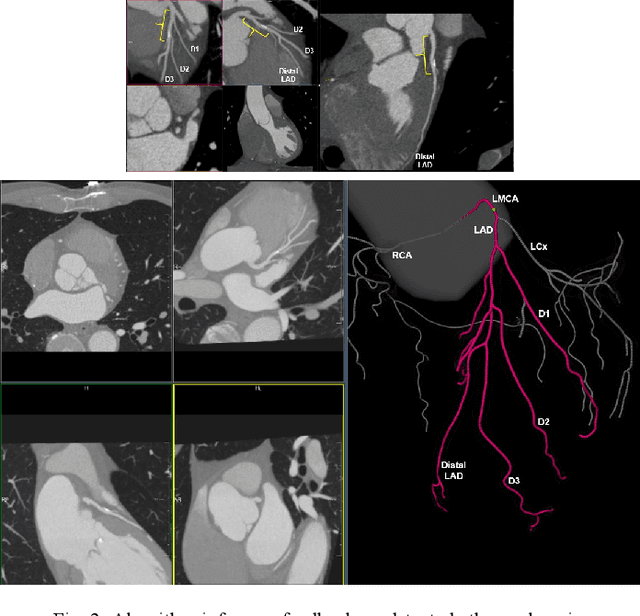
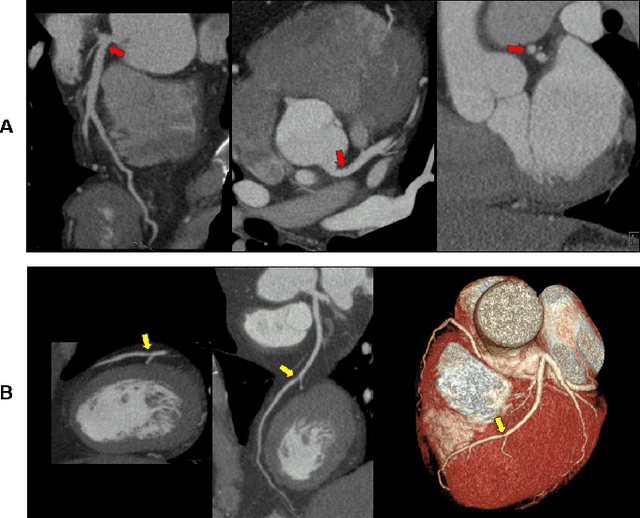
Abstract:Coronary Computed Tomography Angiography (CCTA) evaluation of chest-pain patients in an Emergency Department (ED) is considered appropriate. While a negative CCTA interpretation supports direct patient discharge from an ED, labor-intensive analyses are required, with accuracy in jeopardy from distractions. We describe the development of an Artificial Intelligence (AI) algorithm and workflow for assisting interpreting physicians in CCTA screening for the absence of coronary atherosclerosis. The two-phase approach consisted of (1) Phase 1 - focused on the development and preliminary testing of an algorithm for vessel-centerline extraction classification in a balanced study population (n = 500 with 50% disease prevalence) derived by retrospective random case selection; and (2) Phase 2 - concerned with simulated-clinical Trialing of the developed algorithm on a per-case basis in a more real-world study population (n = 100 with 28% disease prevalence) from an ED chest-pain series. This allowed pre-deployment evaluation of the AI-based CCTA screening application which provides a vessel-by-vessel graphic display of algorithm inference results integrated into a clinically capable viewer. Algorithm performance evaluation used Area Under the Receiver-Operating-Characteristic Curve (AUC-ROC); confusion matrices reflected ground-truth vs AI determinations. The vessel-based algorithm demonstrated strong performance with AUC-ROC = 0.96. In both Phase 1 and Phase 2, independent of disease prevalence differences, negative predictive values at the case level were very high at 95%. The rate of completion of the algorithm workflow process (96% with inference results in 55-80 seconds) in Phase 2 depended on adequate image quality. There is potential for this AI application to assist in CCTA interpretation to help extricate atherosclerosis from chest-pain presentations.
Predicting Rate of Cognitive Decline at Baseline Using a Deep Neural Network with Multidata Analysis
Feb 24, 2020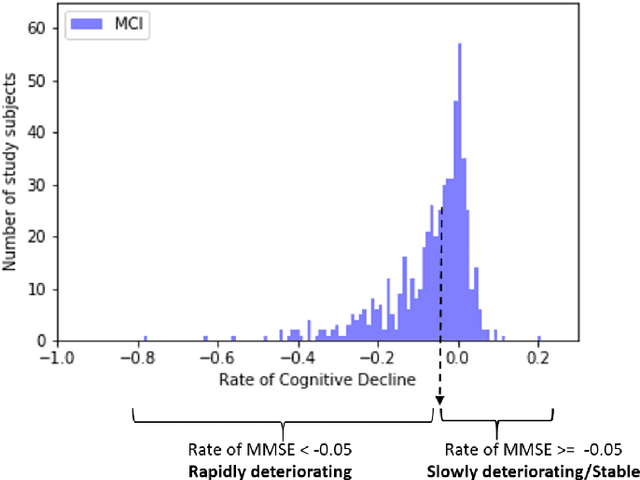

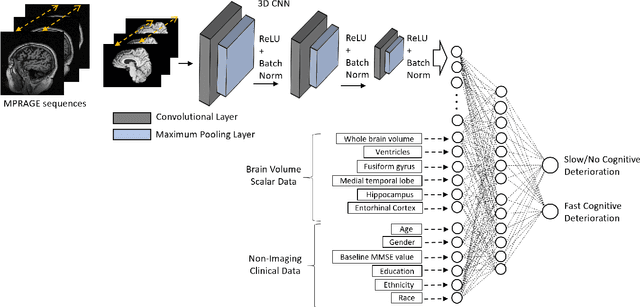

Abstract:This study investigates whether a machine-learning-based system can predict the rate of cognitive-decline in mildly cognitively impaired (MCI) patients by processing only the clinical and imaging data collected at the initial visit. We build a predictive model based on a supervised hybrid neural network utilizing a 3-Dimensional Convolutional Neural Network to perform volume analysis of Magnetic Resonance Imaging (MRI) and integration of non-imaging clinical data at the fully connected layer of the architecture. The analysis is performed on the Alzheimer's Disease Neuroimaging Initiative (ADNI) dataset. Experimental results confirm that there is a correlation between cognitive decline and the data obtained at the first visit. The system achieved an area under the receiver operator curve (AUC) of 66.6% for cognitive decline class prediction.
Coronary Artery Classification and Weakly Supervised Abnormality Localization on Coronary CT Angiography with 3-Dimensional Convolutional Neural Networks
Nov 26, 2019
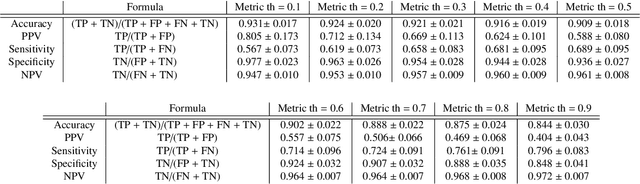
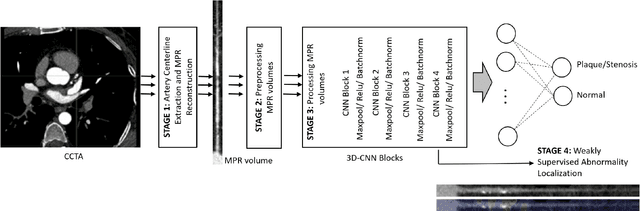
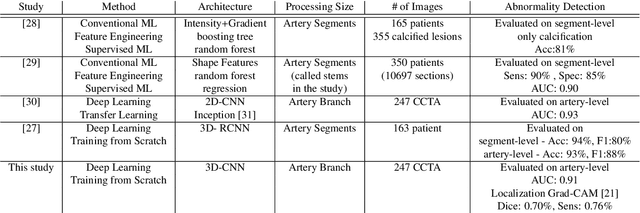
Abstract:We propose a fully automated algorithm based on a deep-learning framework enabling screening of a Coronary Computed Tomography Angiography (CCTA) examination for confident detection of the presence or complete absence of atherosclerotic plaque of the coronary arteries. The system starts with extracting the coronary arteries and their branches from CCTA datasets and representing them with multi-planar reformatted volumes; pre-processing and augmentation techniques are then applied to increase the robustness and generalization ability of the system. A 3-Dimensional Convolutional Neural Network (3D-CNN) is utilized to model pathological changes (e.g., calcification) in coronary arteries/branches. The system then learns the discriminatory features between vessels with and without atherosclerosis. The discriminative features at the final convolutional layer are visualized with a saliency map approach to localize the visual clues related to atherosclerosis. We have evaluated the system on a reference dataset representing 247 patients with atherosclerosis and 246 patients free of atherosclerosis. With 5-fold cross-validation, an accuracy = 90.9%, with Positive Predictive Value = 58.8%, Sensitivity = 68.9%, Specificity of 93.6%, and Negative Predictive Value = 96.1% are achieved at the artery/branch level with a threshold of 0.5. The average area under the curve = 0.91. The system indicates a high negative predictive value, which may be potentially useful for assisting physicians in identifying patients with no coronary atherosclerosis that need no further diagnostic evaluation.
Are Quantitative Features of Lung Nodules Reproducible at Different CT Acquisition and Reconstruction Parameters?
Aug 14, 2019
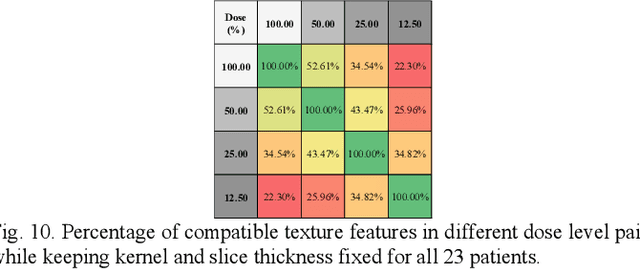
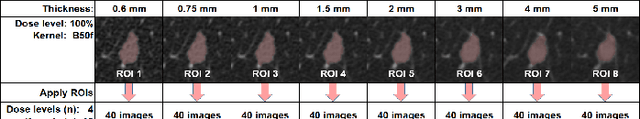
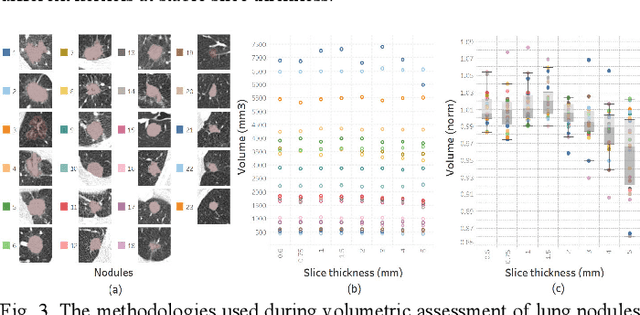
Abstract:Consistency and duplicability in Computed Tomography (CT) output is essential to quantitative imaging for lung cancer detection and monitoring. This study of CT-detected lung nodules investigated the reproducibility of volume-, density-, and texture-based features (outcome variables) over routine ranges of radiation-dose, reconstruction kernel, and slice thickness. CT raw data of 23 nodules were reconstructed using 320 acquisition/reconstruction conditions (combinations of 4 doses, 10 kernels, and 8 thicknesses). Scans at 12.5%, 25%, and 50% of protocol dose were simulated; reduced-dose and full-dose data were reconstructed using conventional filtered back-projection and iterative-reconstruction kernels at a range of thicknesses (0.6-5.0 mm). Full-dose/B50f kernel reconstructions underwent expert segmentation for reference Region-Of-Interest (ROI) and nodule volume per thickness; each ROI was applied to 40 corresponding images (combinations of 4 doses and 10 kernels). Typical texture analysis metrics (including 5 histogram features, 13 Gray Level Co-occurrence Matrix, 5 Run Length Matrix, 2 Neighboring Gray-Level Dependence Matrix, and 2 Neighborhood Gray-Tone Difference Matrix) were computed per ROI. Reconstruction conditions resulting in no significant change in volume, density, or texture metrics were identified as "compatible pairs" for a given outcome variable. Our results indicate that as thickness increases, volumetric reproducibility decreases, while reproducibility of histogram- and texture-based features across different acquisition and reconstruction parameters improves. In order to achieve concomitant reproducibility of volumetric and radiomic results across studies, balanced standardization of the imaging acquisition parameters is required.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge