Alexandre H. Thiéry
The 3D Structural Phenotype of the Glaucomatous Optic Nerve Head and its Relationship with The Severity of Visual Field Damage
Jan 07, 2023



Abstract:$\bf{Purpose}$: To describe the 3D structural changes in both connective and neural tissues of the optic nerve head (ONH) that occur concurrently at different stages of glaucoma using traditional and AI-driven approaches. $\bf{Methods}$: We included 213 normal, 204 mild glaucoma (mean deviation [MD] $\ge$ -6.00 dB), 118 moderate glaucoma (MD of -6.01 to -12.00 dB), and 118 advanced glaucoma patients (MD < -12.00 dB). All subjects had their ONHs imaged in 3D with Spectralis optical coherence tomography. To describe the 3D structural phenotype of glaucoma as a function of severity, we used two different approaches: (1) We extracted human-defined 3D structural parameters of the ONH including retinal nerve fiber layer (RNFL) thickness, lamina cribrosa (LC) shape and depth at different stages of glaucoma; (2) we also employed a geometric deep learning method (i.e. PointNet) to identify the most important 3D structural features that differentiate ONHs from different glaucoma severity groups without any human input. $\bf{Results}$: We observed that the majority of ONH structural changes occurred in the early glaucoma stage, followed by a plateau effect in the later stages. Using PointNet, we also found that 3D ONH structural changes were present in both neural and connective tissues. In both approaches, we observed that structural changes were more prominent in the superior and inferior quadrant of the ONH, particularly in the RNFL, the prelamina, and the LC. As the severity of glaucoma increased, these changes became more diffuse (i.e. widespread), particularly in the LC. $\bf{Conclusions}$: In this study, we were able to uncover complex 3D structural changes of the ONH in both neural and connective tissues as a function of glaucoma severity. We hope to provide new insights into the complex pathophysiology of glaucoma that might help clinicians in their daily clinical care.
Geometric Deep Learning to Identify the Critical 3D Structural Features of the Optic Nerve Head for Glaucoma Diagnosis
Apr 20, 2022

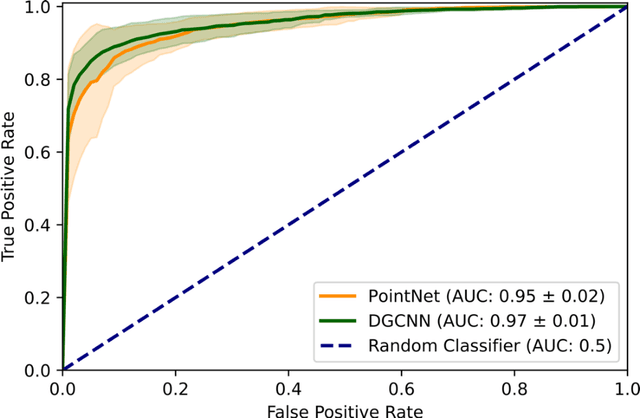

Abstract:Purpose: The optic nerve head (ONH) undergoes complex and deep 3D morphological changes during the development and progression of glaucoma. Optical coherence tomography (OCT) is the current gold standard to visualize and quantify these changes, however the resulting 3D deep-tissue information has not yet been fully exploited for the diagnosis and prognosis of glaucoma. To this end, we aimed: (1) To compare the performance of two relatively recent geometric deep learning techniques in diagnosing glaucoma from a single OCT scan of the ONH; and (2) To identify the 3D structural features of the ONH that are critical for the diagnosis of glaucoma. Methods: In this study, we included a total of 2,247 non-glaucoma and 2,259 glaucoma scans from 1,725 subjects. All subjects had their ONHs imaged in 3D with Spectralis OCT. All OCT scans were automatically segmented using deep learning to identify major neural and connective tissues. Each ONH was then represented as a 3D point cloud. We used PointNet and dynamic graph convolutional neural network (DGCNN) to diagnose glaucoma from such 3D ONH point clouds and to identify the critical 3D structural features of the ONH for glaucoma diagnosis. Results: Both the DGCNN (AUC: 0.97$\pm$0.01) and PointNet (AUC: 0.95$\pm$0.02) were able to accurately detect glaucoma from 3D ONH point clouds. The critical points formed an hourglass pattern with most of them located in the inferior and superior quadrant of the ONH. Discussion: The diagnostic accuracy of both geometric deep learning approaches was excellent. Moreover, we were able to identify the critical 3D structural features of the ONH for glaucoma diagnosis that tremendously improved the transparency and interpretability of our method. Consequently, our approach may have strong potential to be used in clinical applications for the diagnosis and prognosis of a wide range of ophthalmic disorders.
3D Structural Analysis of the Optic Nerve Head to Robustly Discriminate Between Papilledema and Optic Disc Drusen
Dec 18, 2021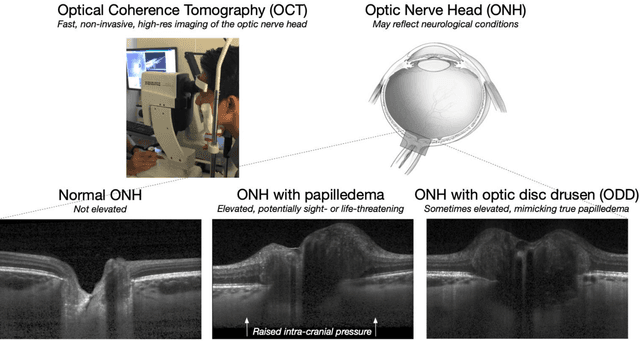
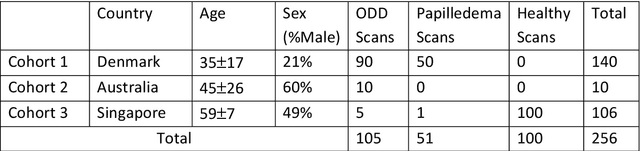
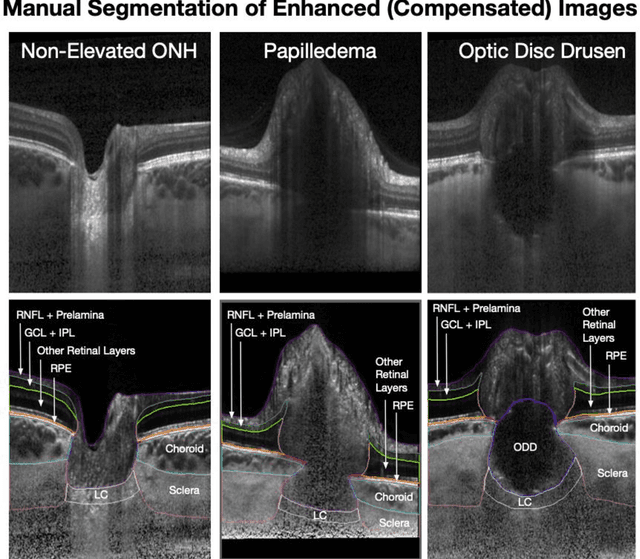
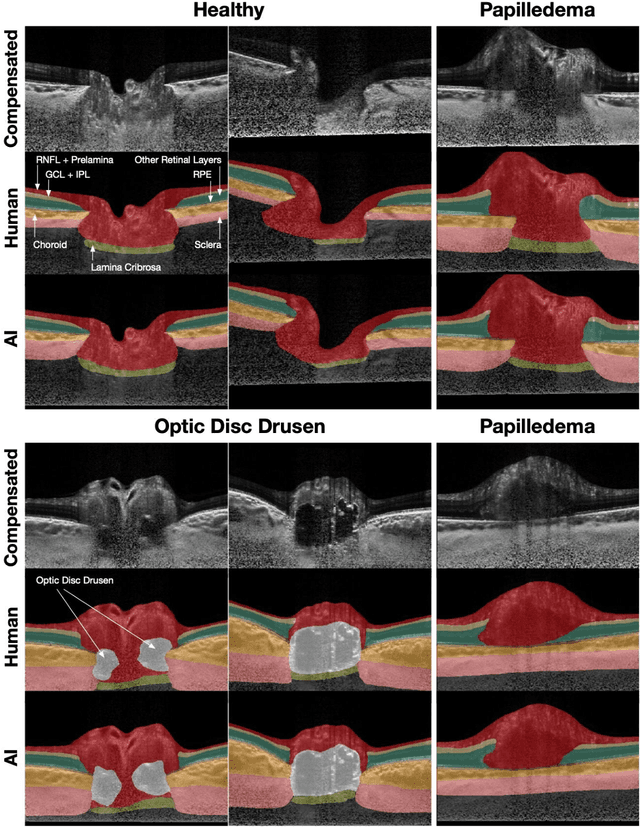
Abstract:Purpose: (1) To develop a deep learning algorithm to identify major tissue structures of the optic nerve head (ONH) in 3D optical coherence tomography (OCT) scans; (2) to exploit such information to robustly differentiate among healthy, optic disc drusen (ODD), and papilledema ONHs. It was a cross-sectional comparative study with confirmed ODD (105 eyes), papilledema due to high intracranial pressure (51 eyes), and healthy controls (100 eyes). 3D scans of the ONHs were acquired using OCT, then processed to improve deep-tissue visibility. At first, a deep learning algorithm was developed using 984 B-scans (from 130 eyes) in order to identify: major neural/connective tissues, and ODD regions. The performance of our algorithm was assessed using the Dice coefficient (DC). In a 2nd step, a classification algorithm (random forest) was designed using 150 OCT volumes to perform 3-class classifications (1: ODD, 2: papilledema, 3: healthy) strictly from their drusen and prelamina swelling scores (derived from the segmentations). To assess performance, we reported the area under the receiver operating characteristic curves (AUCs) for each class. Our segmentation algorithm was able to isolate neural and connective tissues, and ODD regions whenever present. This was confirmed by an average DC of 0.93$\pm$0.03 on the test set, corresponding to good performance. Classification was achieved with high AUCs, i.e. 0.99$\pm$0.01 for the detection of ODD, 0.99 $\pm$ 0.01 for the detection of papilledema, and 0.98$\pm$0.02 for the detection of healthy ONHs. Our AI approach accurately discriminated ODD from papilledema, using a single OCT scan. Our classification performance was excellent, with the caveat that validation in a much larger population is warranted. Our approach may have the potential to establish OCT as the mainstay of diagnostic imaging in neuro-ophthalmology.
Describing the Structural Phenotype of the Glaucomatous Optic Nerve Head Using Artificial Intelligence
Dec 17, 2020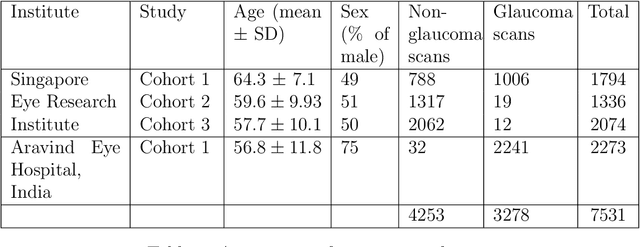
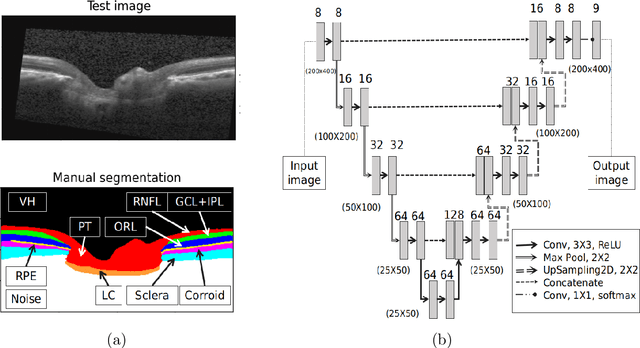
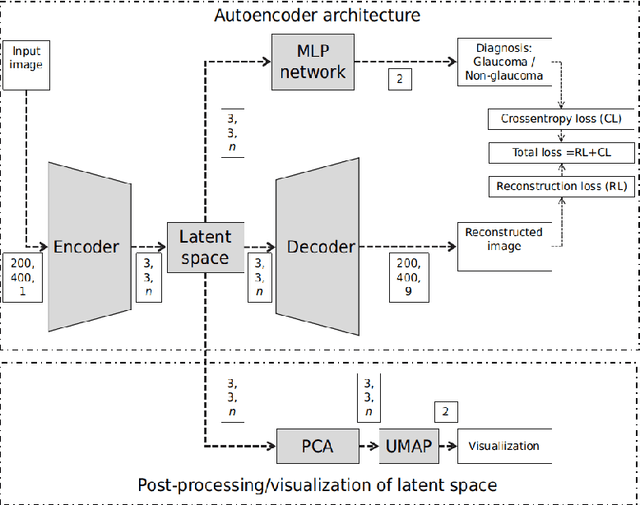

Abstract:The optic nerve head (ONH) typically experiences complex neural- and connective-tissue structural changes with the development and progression of glaucoma, and monitoring these changes could be critical for improved diagnosis and prognosis in the glaucoma clinic. The gold-standard technique to assess structural changes of the ONH clinically is optical coherence tomography (OCT). However, OCT is limited to the measurement of a few hand-engineered parameters, such as the thickness of the retinal nerve fiber layer (RNFL), and has not yet been qualified as a stand-alone device for glaucoma diagnosis and prognosis applications. We argue this is because the vast amount of information available in a 3D OCT scan of the ONH has not been fully exploited. In this study we propose a deep learning approach that can: \textbf{(1)} fully exploit information from an OCT scan of the ONH; \textbf{(2)} describe the structural phenotype of the glaucomatous ONH; and that can \textbf{(3)} be used as a robust glaucoma diagnosis tool. Specifically, the structural features identified by our algorithm were found to be related to clinical observations of glaucoma. The diagnostic accuracy from these structural features was $92.0 \pm 2.3 \%$ with a sensitivity of $90.0 \pm 2.4 \% $ (at $95 \%$ specificity). By changing their magnitudes in steps, we were able to reveal how the morphology of the ONH changes as one transitions from a `non-glaucoma' to a `glaucoma' condition. We believe our work may have strong clinical implication for our understanding of glaucoma pathogenesis, and could be improved in the future to also predict future loss of vision.
OCT-GAN: Single Step Shadow and Noise Removal from Optical Coherence Tomography Images of the Human Optic Nerve Head
Oct 06, 2020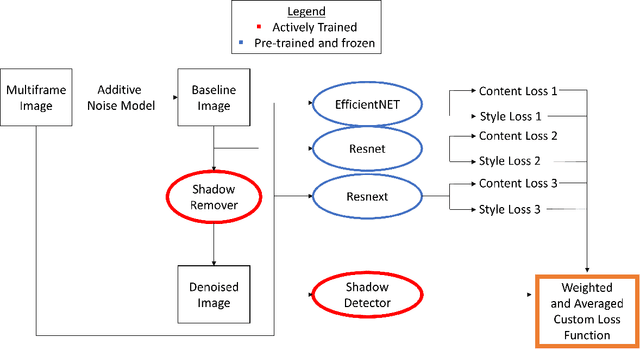
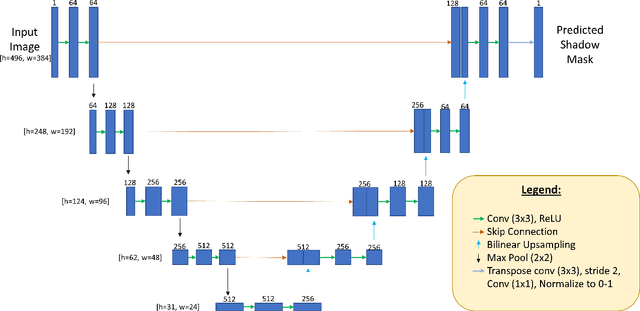
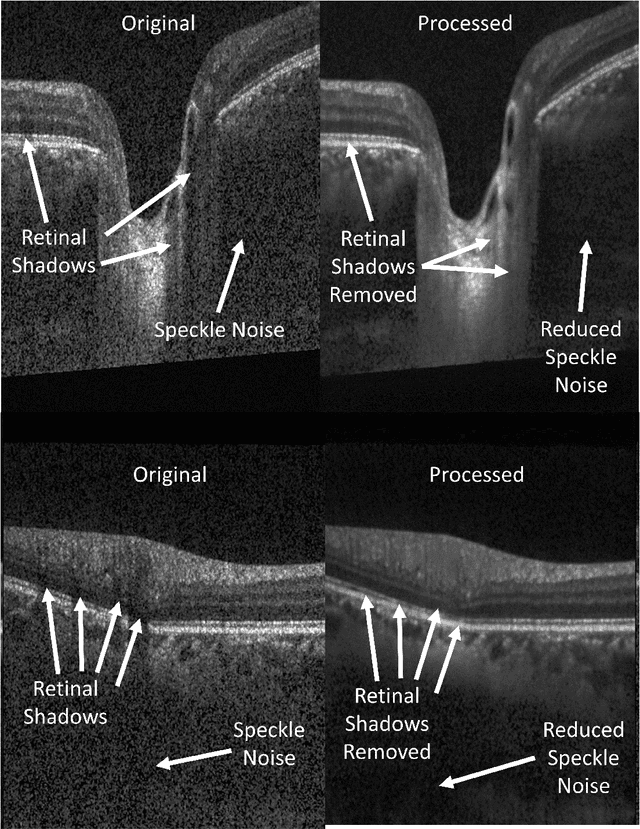

Abstract:Speckle noise and retinal shadows within OCT B-scans occlude important edges, fine textures and deep tissues, preventing accurate and robust diagnosis by algorithms and clinicians. We developed a single process that successfully removed both noise and retinal shadows from unseen single-frame B-scans within 10.4ms. Mean average gradient magnitude (AGM) for the proposed algorithm was 57.2% higher than current state-of-the-art, while mean peak signal to noise ratio (PSNR), contrast to noise ratio (CNR), and structural similarity index metric (SSIM) increased by 11.1%, 154% and 187% respectively compared to single-frame B-scans. Mean intralayer contrast (ILC) improvement for the retinal nerve fiber layer (RNFL), photoreceptor layer (PR) and retinal pigment epithelium (RPE) layers decreased from 0.362 \pm 0.133 to 0.142 \pm 0.102, 0.449 \pm 0.116 to 0.0904 \pm 0.0769, 0.381 \pm 0.100 to 0.0590 \pm 0.0451 respectively. The proposed algorithm reduces the necessity for long image acquisition times, minimizes expensive hardware requirements and reduces motion artifacts in OCT images.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge