Veeru Kasivisvanathan
Maximizing T2-Only Prostate Cancer Localization from Expected Diffusion Weighted Imaging
Apr 01, 2026Abstract:Multiparametric MRI is increasingly recommended as a first-line noninvasive approach to detect and localize prostate cancer, requiring at minimum diffusion-weighted (DWI) and T2-weighted (T2w) MR sequences. Early machine learning attempts using only T2w images have shown promising diagnostic performance in segmenting radiologist-annotated lesions. Such uni-modal T2-only approaches deliver substantial clinical benefits by reducing costs and expertise required to acquire other sequences. This work investigates an arguably more challenging application using only T2w at inference, but to localize individual cancers based on independent histopathology labels. We formulate DWI images as a latent modality (readily available during training) to classify cancer presence at local Barzell zones, given only T2w images as input. In the resulting expectation-maximization algorithm, a latent modality generator (implemented using a flow matching-based generative model) approximates the latent DWI image posterior distribution in the E-steps, while in M-steps a cancer localizer is simultaneously optimized with the generative model to maximize the expected likelihood of cancer presence. The proposed approach provides a novel theoretical framework for learning from a privileged DWI modality, yielding superior cancer localization performance compared to approaches that lack training DWI images or existing frameworks for privileged learning and incomplete modalities. The proposed T2-only methods perform competitively or better than baseline methods using multiple input sequences (e.g., improving the patient-level F1 score by 14.4\% and zone-level QWK by 5.3\% over the T2w+DWI baseline). We present quantitative evaluations using internal and external datasets from 4,133 prostate cancer patients with histopathology-verified labels.
ProFound: A moderate-sized vision foundation model for multi-task prostate imaging
Mar 04, 2026Abstract:Many diagnostic and therapeutic clinical tasks for prostate cancer increasingly rely on multi-parametric MRI. Automating these tasks is challenging because they necessitate expert interpretations, which are difficult to scale to capitalise on modern deep learning. Although modern automated systems achieve expert-level performance in isolated tasks, their general clinical utility remains limited by the requirement of large task-specific labelled datasets. In this paper, we present ProFound, a domain-specialised vision foundation model for volumetric prostate mpMRI. ProFound is pre-trained using several variants of self-supervised approaches on a diverse, multi-institutional collection of 5,000 patients, with a total of over 22,000 unique 3D MRI volumes (over 1,800,000 2D image slices). We conducted a systematic evaluation of ProFound across a broad spectrum of $11$ downstream clinical tasks on over 3,000 independent patients, including prostate cancer detection, Gleason grading, lesion localisation, gland volume estimation, zonal and surrounding structure segmentation. Experimental results demonstrate that finetuned ProFound consistently outperforms or remains competitive with state-of-the-art specialised models and existing medical vision foundation models trained/finetuned on the same data.
Understanding the Transfer Limits of Vision Foundation Models
Jan 22, 2026Abstract:Foundation models leverage large-scale pretraining to capture extensive knowledge, demonstrating generalization in a wide range of language tasks. By comparison, vision foundation models (VFMs) often exhibit uneven improvements across downstream tasks, despite substantial computational investment. We postulate that this limitation arises from a mismatch between pretraining objectives and the demands of downstream vision-and-imaging tasks. Pretraining strategies like masked image reconstruction or contrastive learning shape representations for tasks such as recovery of generic visual patterns or global semantic structures, which may not align with the task-specific requirements of downstream applications including segmentation, classification, or image synthesis. To investigate this in a concrete real-world clinical area, we assess two VFMs, a reconstruction-focused MAE-based model (ProFound) and a contrastive-learning-based model (ProViCNet), on five prostate multiparametric MR imaging tasks, examining how such task alignment influences transfer performance, i.e., from pretraining to fine-tuning. Our findings indicate that better alignment between pretraining and downstream tasks, measured by simple divergence metrics such as maximum-mean-discrepancy (MMD) between the same features before and after fine-tuning, correlates with greater performance improvements and faster convergence, emphasizing the importance of designing and analyzing pretraining objectives with downstream applicability in mind.
Promptable cancer segmentation using minimal expert-curated data
May 23, 2025Abstract:Automated segmentation of cancer on medical images can aid targeted diagnostic and therapeutic procedures. However, its adoption is limited by the high cost of expert annotations required for training and inter-observer variability in datasets. While weakly-supervised methods mitigate some challenges, using binary histology labels for training as opposed to requiring full segmentation, they require large paired datasets of histology and images, which are difficult to curate. Similarly, promptable segmentation aims to allow segmentation with no re-training for new tasks at inference, however, existing models perform poorly on pathological regions, again necessitating large datasets for training. In this work we propose a novel approach for promptable segmentation requiring only 24 fully-segmented images, supplemented by 8 weakly-labelled images, for training. Curating this minimal data to a high standard is relatively feasible and thus issues with the cost and variability of obtaining labels can be mitigated. By leveraging two classifiers, one weakly-supervised and one fully-supervised, our method refines segmentation through a guided search process initiated by a single-point prompt. Our approach outperforms existing promptable segmentation methods, and performs comparably with fully-supervised methods, for the task of prostate cancer segmentation, while using substantially less annotated data (up to 100X less). This enables promptable segmentation with very minimal labelled data, such that the labels can be curated to a very high standard.
T2-Only Prostate Cancer Prediction by Meta-Learning from Bi-Parametric MR Imaging
Nov 11, 2024


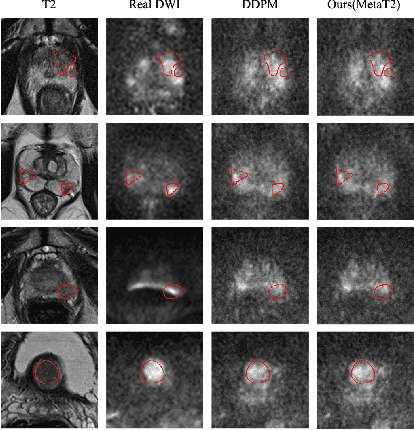
Abstract:Current imaging-based prostate cancer diagnosis requires both MR T2-weighted (T2w) and diffusion-weighted imaging (DWI) sequences, with additional sequences for potentially greater accuracy improvement. However, measuring diffusion patterns in DWI sequences can be time-consuming, prone to artifacts and sensitive to imaging parameters. While machine learning (ML) models have demonstrated radiologist-level accuracy in detecting prostate cancer from these two sequences, this study investigates the potential of ML-enabled methods using only the T2w sequence as input during inference time. We first discuss the technical feasibility of such a T2-only approach, and then propose a novel ML formulation, where DWI sequences - readily available for training purposes - are only used to train a meta-learning model, which subsequently only uses T2w sequences at inference. Using multiple datasets from more than 3,000 prostate cancer patients, we report superior or comparable performance in localising radiologist-identified prostate cancer using our proposed T2-only models, compared with alternative models using T2-only or both sequences as input. Real patient cases are presented and discussed to demonstrate, for the first time, the exclusively true-positive cases from models with different input sequences.
AI-assisted prostate cancer detection and localisation on biparametric MR by classifying radiologist-positives
Oct 30, 2024



Abstract:Prostate cancer diagnosis through MR imaging have currently relied on radiologists' interpretation, whilst modern AI-based methods have been developed to detect clinically significant cancers independent of radiologists. In this study, we propose to develop deep learning models that improve the overall cancer diagnostic accuracy, by classifying radiologist-identified patients or lesions (i.e. radiologist-positives), as opposed to the existing models that are trained to discriminate over all patients. We develop a single voxel-level classification model, with a simple percentage threshold to determine positive cases, at levels of lesions, Barzell-zones and patients. Based on the presented experiments from two clinical data sets, consisting of histopathology-labelled MR images from more than 800 and 500 patients in the respective UCLA and UCL PROMIS studies, we show that the proposed strategy can improve the diagnostic accuracy, by augmenting the radiologist reading of the MR imaging. Among varying definition of clinical significance, the proposed strategy, for example, achieved a specificity of 44.1% (with AI assistance) from 36.3% (by radiologists alone), at a controlled sensitivity of 80.0% on the publicly available UCLA data set. This provides measurable clinical values in a range of applications such as reducing unnecessary biopsies, lowering cost in cancer screening and quantifying risk in therapies.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge