Sebastian Pölsterl
From Barlow Twins to Triplet Training: Differentiating Dementia with Limited Data
Apr 09, 2024Abstract:Differential diagnosis of dementia is challenging due to overlapping symptoms, with structural magnetic resonance imaging (MRI) being the primary method for diagnosis. Despite the clinical value of computer-aided differential diagnosis, research has been limited, mainly due to the absence of public datasets that contain diverse types of dementia. This leaves researchers with small in-house datasets that are insufficient for training deep neural networks (DNNs). Self-supervised learning shows promise for utilizing unlabeled MRI scans in training, but small batch sizes for volumetric brain scans make its application challenging. To address these issues, we propose Triplet Training for differential diagnosis with limited target data. It consists of three key stages: (i) self-supervised pre-training on unlabeled data with Barlow Twins, (ii) self-distillation on task-related data, and (iii) fine-tuning on the target dataset. Our approach significantly outperforms traditional training strategies, achieving a balanced accuracy of 75.6%. We further provide insights into the training process by visualizing changes in the latent space after each step. Finally, we validate the robustness of Triplet Training in terms of its individual components in a comprehensive ablation study. Our code is available at https://github.com/ai-med/TripletTraining.
Keep the Faith: Faithful Explanations in Convolutional Neural Networks for Case-Based Reasoning
Dec 19, 2023


Abstract:Explaining predictions of black-box neural networks is crucial when applied to decision-critical tasks. Thus, attribution maps are commonly used to identify important image regions, despite prior work showing that humans prefer explanations based on similar examples. To this end, ProtoPNet learns a set of class-representative feature vectors (prototypes) for case-based reasoning. During inference, similarities of latent features to prototypes are linearly classified to form predictions and attribution maps are provided to explain the similarity. In this work, we evaluate whether architectures for case-based reasoning fulfill established axioms required for faithful explanations using the example of ProtoPNet. We show that such architectures allow the extraction of faithful explanations. However, we prove that the attribution maps used to explain the similarities violate the axioms. We propose a new procedure to extract explanations for trained ProtoPNets, named ProtoPFaith. Conceptually, these explanations are Shapley values, calculated on the similarity scores of each prototype. They allow to faithfully answer which prototypes are present in an unseen image and quantify each pixel's contribution to that presence, thereby complying with all axioms. The theoretical violations of ProtoPNet manifest in our experiments on three datasets (CUB-200-2011, Stanford Dogs, RSNA) and five architectures (ConvNet, ResNet, ResNet50, WideResNet50, ResNeXt50). Our experiments show a qualitative difference between the explanations given by ProtoPNet and ProtoPFaith. Additionally, we quantify the explanations with the Area Over the Perturbation Curve, on which ProtoPFaith outperforms ProtoPNet on all experiments by a factor $>10^3$.
Don't PANIC: Prototypical Additive Neural Network for Interpretable Classification of Alzheimer's Disease
Mar 14, 2023Abstract:Alzheimer's disease (AD) has a complex and multifactorial etiology, which requires integrating information about neuroanatomy, genetics, and cerebrospinal fluid biomarkers for accurate diagnosis. Hence, recent deep learning approaches combined image and tabular information to improve diagnostic performance. However, the black-box nature of such neural networks is still a barrier for clinical applications, in which understanding the decision of a heterogeneous model is integral. We propose PANIC, a prototypical additive neural network for interpretable AD classification that integrates 3D image and tabular data. It is interpretable by design and, thus, avoids the need for post-hoc explanations that try to approximate the decision of a network. Our results demonstrate that PANIC achieves state-of-the-art performance in AD classification, while directly providing local and global explanations. Finally, we show that PANIC extracts biologically meaningful signatures of AD, and satisfies a set of desirable desiderata for trustworthy machine learning. Our implementation is available at https://github.com/ai-med/PANIC .
Joint Reconstruction and Parcellation of Cortical Surfaces
Sep 19, 2022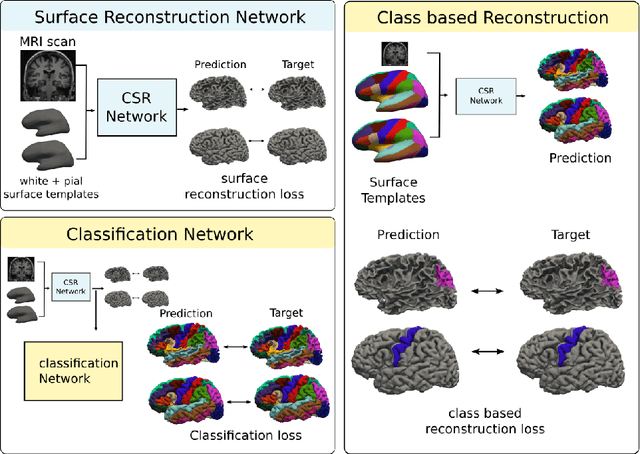
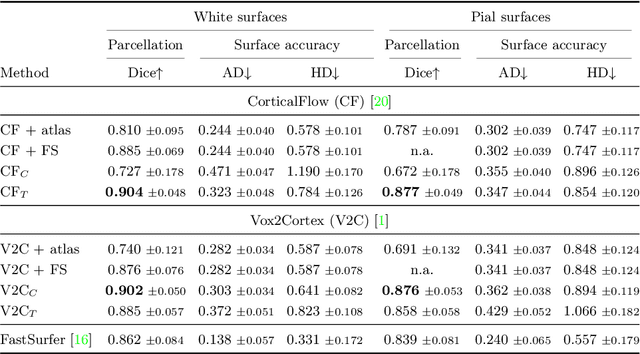
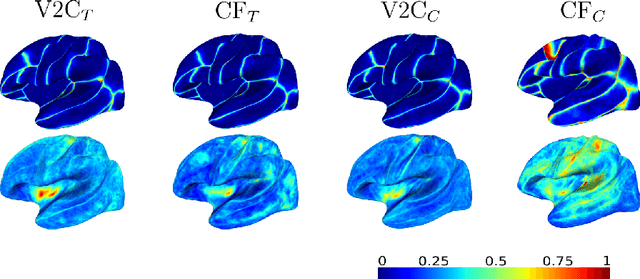
Abstract:The reconstruction of cerebral cortex surfaces from brain MRI scans is instrumental for the analysis of brain morphology and the detection of cortical thinning in neurodegenerative diseases like Alzheimer's disease (AD). Moreover, for a fine-grained analysis of atrophy patterns, the parcellation of the cortical surfaces into individual brain regions is required. For the former task, powerful deep learning approaches, which provide highly accurate brain surfaces of tissue boundaries from input MRI scans in seconds, have recently been proposed. However, these methods do not come with the ability to provide a parcellation of the reconstructed surfaces. Instead, separate brain-parcellation methods have been developed, which typically consider the cortical surfaces as given, often computed beforehand with FreeSurfer. In this work, we propose two options, one based on a graph classification branch and another based on a novel generic 3D reconstruction loss, to augment template-deformation algorithms such that the surface meshes directly come with an atlas-based brain parcellation. By combining both options with two of the latest cortical surface reconstruction algorithms, we attain highly accurate parcellations with a Dice score of 90.2 (graph classification branch) and 90.4 (novel reconstruction loss) together with state-of-the-art surfaces.
Is a PET all you need? A multi-modal study for Alzheimer's disease using 3D CNNs
Jul 05, 2022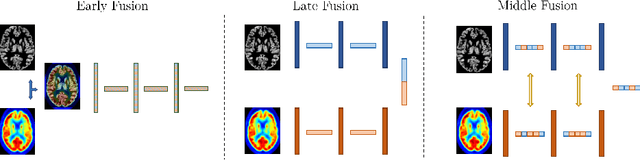
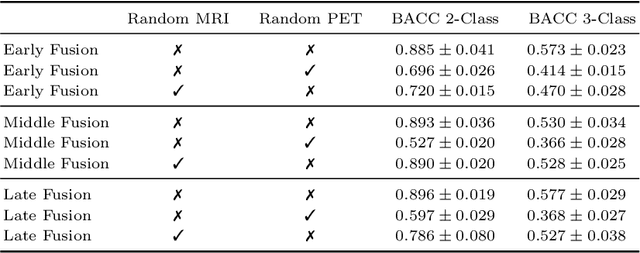

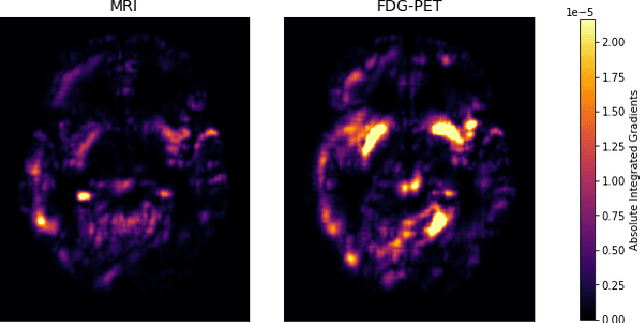
Abstract:Alzheimer's Disease (AD) is the most common form of dementia and often difficult to diagnose due to the multifactorial etiology of dementia. Recent works on neuroimaging-based computer-aided diagnosis with deep neural networks (DNNs) showed that fusing structural magnetic resonance images (sMRI) and fluorodeoxyglucose positron emission tomography (FDG-PET) leads to improved accuracy in a study population of healthy controls and subjects with AD. However, this result conflicts with the established clinical knowledge that FDG-PET better captures AD-specific pathologies than sMRI. Therefore, we propose a framework for the systematic evaluation of multi-modal DNNs and critically re-evaluate single- and multi-modal DNNs based on FDG-PET and sMRI for binary healthy vs. AD, and three-way healthy/mild cognitive impairment/AD classification. Our experiments demonstrate that a single-modality network using FDG-PET performs better than MRI (accuracy 0.91 vs 0.87) and does not show improvement when combined. This conforms with the established clinical knowledge on AD biomarkers, but raises questions about the true benefit of multi-modal DNNs. We argue that future work on multi-modal fusion should systematically assess the contribution of individual modalities following our proposed evaluation framework. Finally, we encourage the community to go beyond healthy vs. AD classification and focus on differential diagnosis of dementia, where fusing multi-modal image information conforms with a clinical need.
CASHformer: Cognition Aware SHape Transformer for Longitudinal Analysis
Jul 05, 2022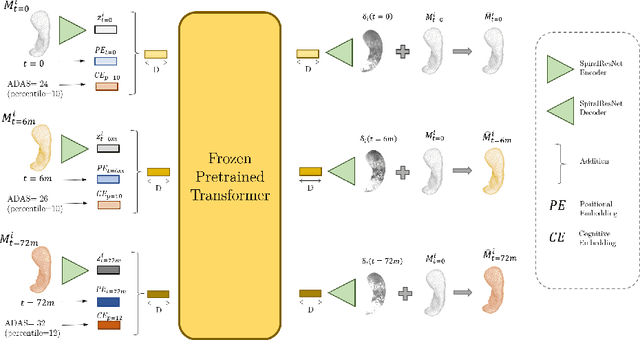
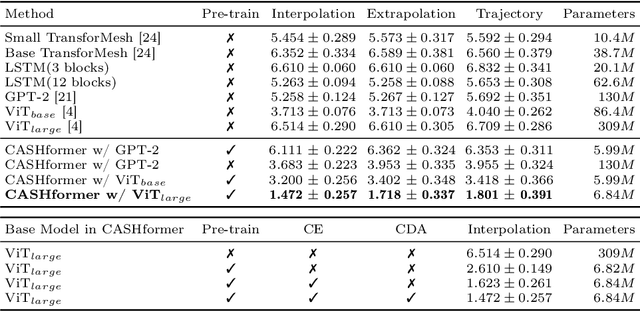
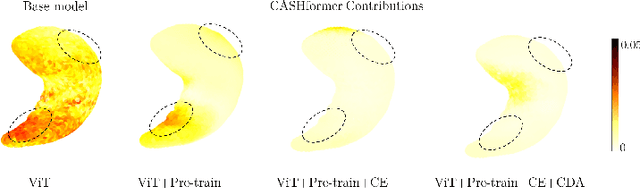
Abstract:Modeling temporal changes in subcortical structures is crucial for a better understanding of the progression of Alzheimer's disease (AD). Given their flexibility to adapt to heterogeneous sequence lengths, mesh-based transformer architectures have been proposed in the past for predicting hippocampus deformations across time. However, one of the main limitations of transformers is the large amount of trainable parameters, which makes the application on small datasets very challenging. In addition, current methods do not include relevant non-image information that can help to identify AD-related patterns in the progression. To this end, we introduce CASHformer, a transformer-based framework to model longitudinal shape trajectories in AD. CASHformer incorporates the idea of pre-trained transformers as universal compute engines that generalize across a wide range of tasks by freezing most layers during fine-tuning. This reduces the number of parameters by over 90% with respect to the original model and therefore enables the application of large models on small datasets without overfitting. In addition, CASHformer models cognitive decline to reveal AD atrophy patterns in the temporal sequence. Our results show that CASHformer reduces the reconstruction error by 73% compared to previously proposed methods. Moreover, the accuracy of detecting patients progressing to AD increases by 3% with imputing missing longitudinal shape data.
Vox2Cortex: Fast Explicit Reconstruction of Cortical Surfaces from 3D MRI Scans with Geometric Deep Neural Networks
Mar 18, 2022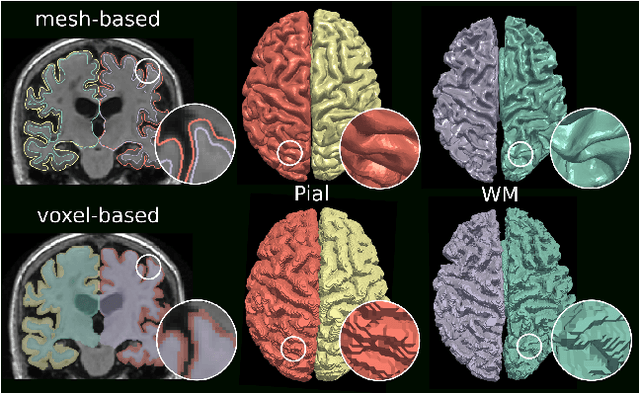
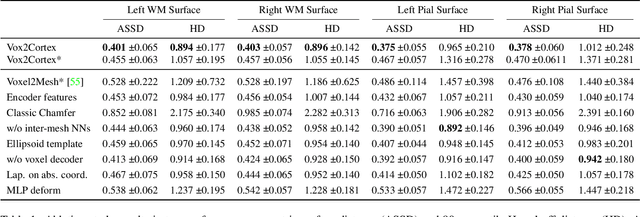

Abstract:The reconstruction of cortical surfaces from brain magnetic resonance imaging (MRI) scans is essential for quantitative analyses of cortical thickness and sulcal morphology. Although traditional and deep learning-based algorithmic pipelines exist for this purpose, they have two major drawbacks: lengthy runtimes of multiple hours (traditional) or intricate post-processing, such as mesh extraction and topology correction (deep learning-based). In this work, we address both of these issues and propose Vox2Cortex, a deep learning-based algorithm that directly yields topologically correct, three-dimensional meshes of the boundaries of the cortex. Vox2Cortex leverages convolutional and graph convolutional neural networks to deform an initial template to the densely folded geometry of the cortex represented by an input MRI scan. We show in extensive experiments on three brain MRI datasets that our meshes are as accurate as the ones reconstructed by state-of-the-art methods in the field, without the need for time- and resource-intensive post-processing. To accurately reconstruct the tightly folded cortex, we work with meshes containing about 168,000 vertices at test time, scaling deep explicit reconstruction methods to a new level.
TransforMesh: A Transformer Network for Longitudinal modeling of Anatomical Meshes
Sep 23, 2021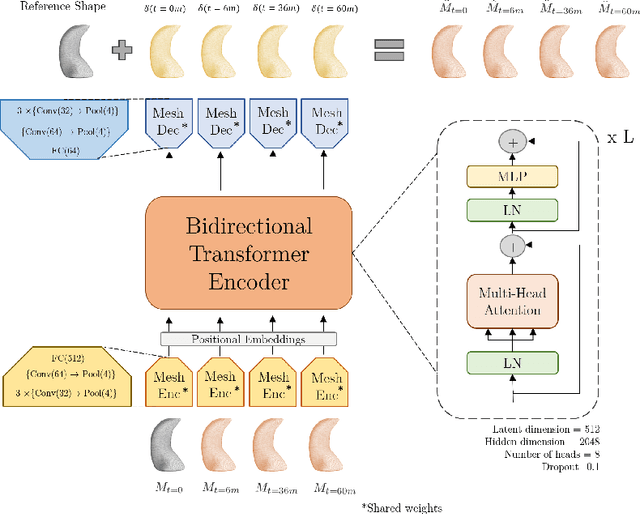
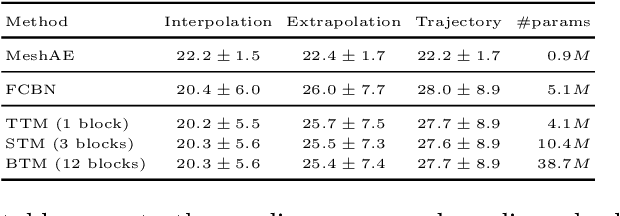
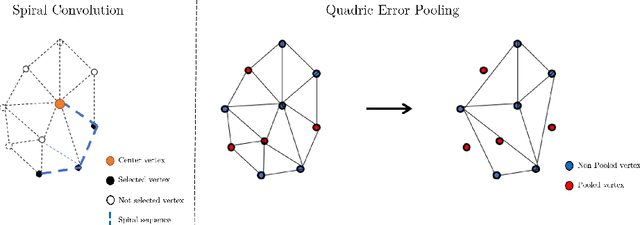
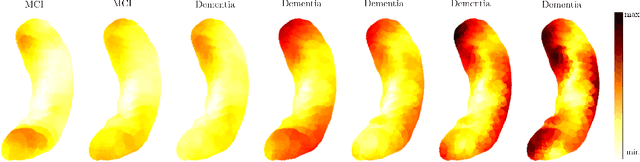
Abstract:The longitudinal modeling of neuroanatomical changes related to Alzheimer's disease (AD) is crucial for studying the progression of the disease. To this end, we introduce TransforMesh, a spatio-temporal network based on transformers that models longitudinal shape changes on 3D anatomical meshes. While transformer and mesh networks have recently shown impressive performances in natural language processing and computer vision, their application to medical image analysis has been very limited. To the best of our knowledge, this is the first work that combines transformer and mesh networks. Our results show that TransforMesh can model shape trajectories better than other baseline architectures that do not capture temporal dependencies. Moreover, we also explore the capabilities of TransforMesh in detecting structural anomalies of the hippocampus in patients developing AD.
Alzheimer's Disease Diagnosis via Deep Factorization Machine Models
Aug 12, 2021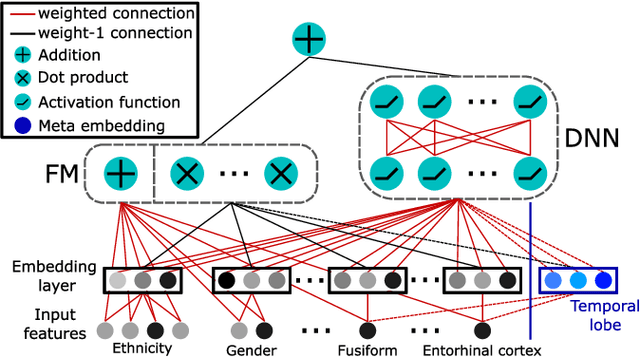
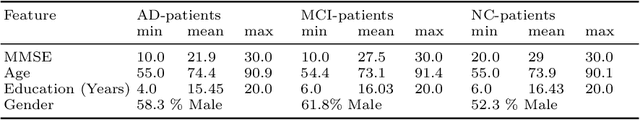


Abstract:The current state-of-the-art deep neural networks (DNNs) for Alzheimer's Disease diagnosis use different biomarker combinations to classify patients, but do not allow extracting knowledge about the interactions of biomarkers. However, to improve our understanding of the disease, it is paramount to extract such knowledge from the learned model. In this paper, we propose a Deep Factorization Machine model that combines the ability of DNNs to learn complex relationships and the ease of interpretability of a linear model. The proposed model has three parts: (i) an embedding layer to deal with sparse categorical data, (ii) a Factorization Machine to efficiently learn pairwise interactions, and (iii) a DNN to implicitly model higher order interactions. In our experiments on data from the Alzheimer's Disease Neuroimaging Initiative, we demonstrate that our proposed model classifies cognitive normal, mild cognitive impaired, and demented patients more accurately than competing models. In addition, we show that valuable knowledge about the interactions among biomarkers can be obtained.
Scalable, Axiomatic Explanations of Deep Alzheimer's Diagnosis from Heterogeneous Data
Jul 13, 2021
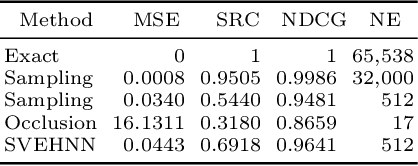
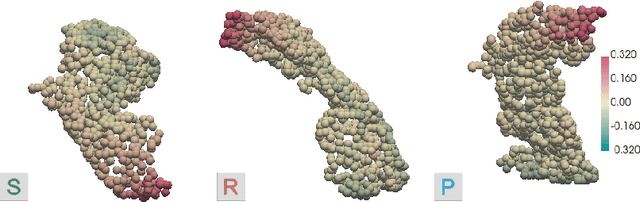

Abstract:Deep Neural Networks (DNNs) have an enormous potential to learn from complex biomedical data. In particular, DNNs have been used to seamlessly fuse heterogeneous information from neuroanatomy, genetics, biomarkers, and neuropsychological tests for highly accurate Alzheimer's disease diagnosis. On the other hand, their black-box nature is still a barrier for the adoption of such a system in the clinic, where interpretability is absolutely essential. We propose Shapley Value Explanation of Heterogeneous Neural Networks (SVEHNN) for explaining the Alzheimer's diagnosis made by a DNN from the 3D point cloud of the neuroanatomy and tabular biomarkers. Our explanations are based on the Shapley value, which is the unique method that satisfies all fundamental axioms for local explanations previously established in the literature. Thus, SVEHNN has many desirable characteristics that previous work on interpretability for medical decision making is lacking. To avoid the exponential time complexity of the Shapley value, we propose to transform a given DNN into a Lightweight Probabilistic Deep Network without re-training, thus achieving a complexity only quadratic in the number of features. In our experiments on synthetic and real data, we show that we can closely approximate the exact Shapley value with a dramatically reduced runtime and can reveal the hidden knowledge the network has learned from the data.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge