Catalina Raymond
Federated Learning Enables Big Data for Rare Cancer Boundary Detection
Apr 25, 2022Abstract:Although machine learning (ML) has shown promise in numerous domains, there are concerns about generalizability to out-of-sample data. This is currently addressed by centrally sharing ample, and importantly diverse, data from multiple sites. However, such centralization is challenging to scale (or even not feasible) due to various limitations. Federated ML (FL) provides an alternative to train accurate and generalizable ML models, by only sharing numerical model updates. Here we present findings from the largest FL study to-date, involving data from 71 healthcare institutions across 6 continents, to generate an automatic tumor boundary detector for the rare disease of glioblastoma, utilizing the largest dataset of such patients ever used in the literature (25,256 MRI scans from 6,314 patients). We demonstrate a 33% improvement over a publicly trained model to delineate the surgically targetable tumor, and 23% improvement over the tumor's entire extent. We anticipate our study to: 1) enable more studies in healthcare informed by large and diverse data, ensuring meaningful results for rare diseases and underrepresented populations, 2) facilitate further quantitative analyses for glioblastoma via performance optimization of our consensus model for eventual public release, and 3) demonstrate the effectiveness of FL at such scale and task complexity as a paradigm shift for multi-site collaborations, alleviating the need for data sharing.
Synthesizing MR Image Contrast Enhancement Using 3D High-resolution ConvNets
Apr 13, 2021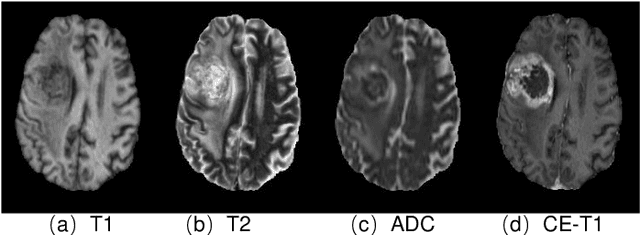
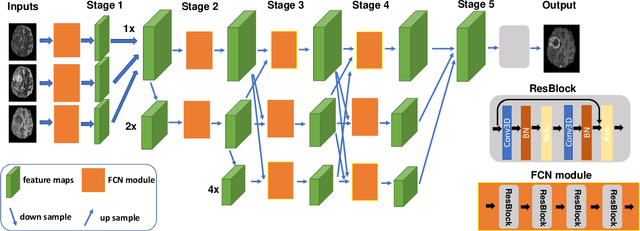
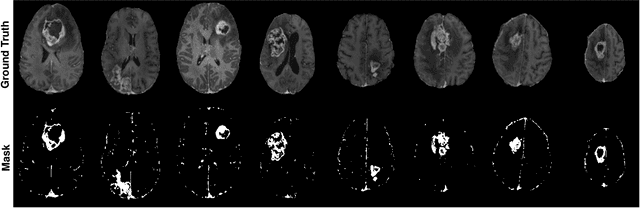
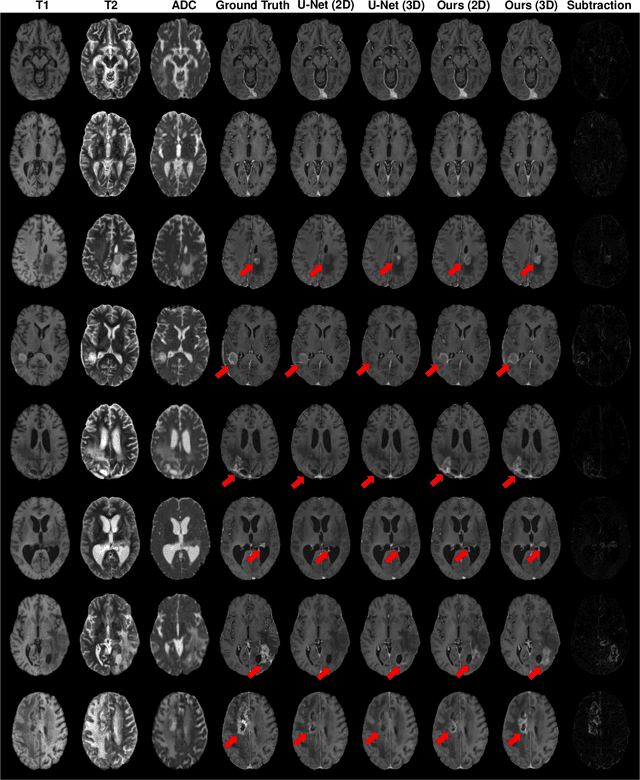
Abstract:Gadolinium-based contrast agents (GBCAs) have been widely used to better visualize disease in brain magnetic resonance imaging (MRI). However, gadolinium deposition within the brain and body has raised safety concerns about the use of GBCAs. Therefore, the development of novel approaches that can decrease or even eliminate GBCA exposure while providing similar contrast information would be of significant use clinically. For brain tumor patients, standard-of-care includes repeated MRI with gadolinium-based contrast for disease monitoring, increasing the risk of gadolinium deposition. In this work, we present a deep learning based approach for contrast-enhanced T1 synthesis on brain tumor patients. A 3D high-resolution fully convolutional network (FCN), which maintains high resolution information through processing and aggregates multi-scale information in parallel, is designed to map pre-contrast MRI sequences to contrast-enhanced MRI sequences. Specifically, three pre-contrast MRI sequences, T1, T2 and apparent diffusion coefficient map (ADC), are utilized as inputs and the post-contrast T1 sequences are utilized as target output. To alleviate the data imbalance problem between normal tissues and the tumor regions, we introduce a local loss to improve the contribution of the tumor regions, which leads to better enhancement results on tumors. Extensive quantitative and visual assessments are performed, with our proposed model achieving a PSNR of 28.24dB in the brain and 21.2dB in tumor regions. Our results suggests the potential of substituting GBCAs with synthetic contrast images generated via deep learning.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge