Yuanfang Guan
The CausalBench challenge: A machine learning contest for gene network inference from single-cell perturbation data
Aug 29, 2023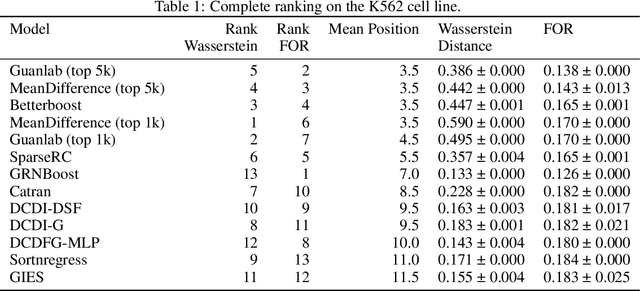
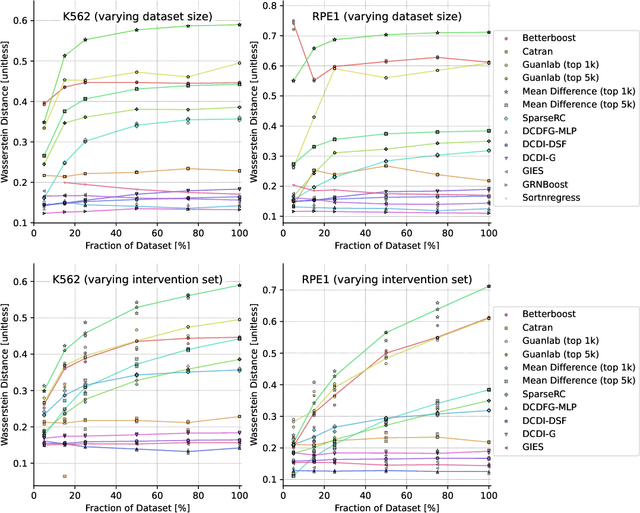
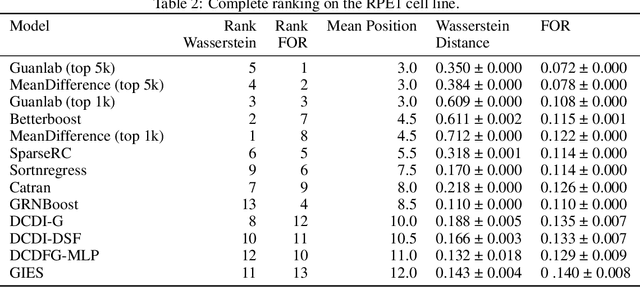
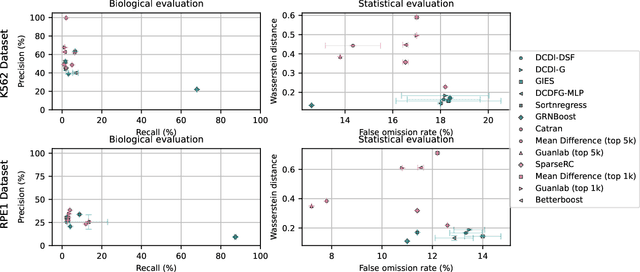
Abstract:In drug discovery, mapping interactions between genes within cellular systems is a crucial early step. This helps formulate hypotheses regarding molecular mechanisms that could potentially be targeted by future medicines. The CausalBench Challenge was an initiative to invite the machine learning community to advance the state of the art in constructing gene-gene interaction networks. These networks, derived from large-scale, real-world datasets of single cells under various perturbations, are crucial for understanding the causal mechanisms underlying disease biology. Using the framework provided by the CausalBench benchmark, participants were tasked with enhancing the capacity of the state of the art methods to leverage large-scale genetic perturbation data. This report provides an analysis and summary of the methods submitted during the challenge to give a partial image of the state of the art at the time of the challenge. The winning solutions significantly improved performance compared to previous baselines, establishing a new state of the art for this critical task in biology and medicine.
Deep learning prediction of patient response time course from early data via neural-pharmacokinetic/pharmacodynamic modeling
Oct 22, 2020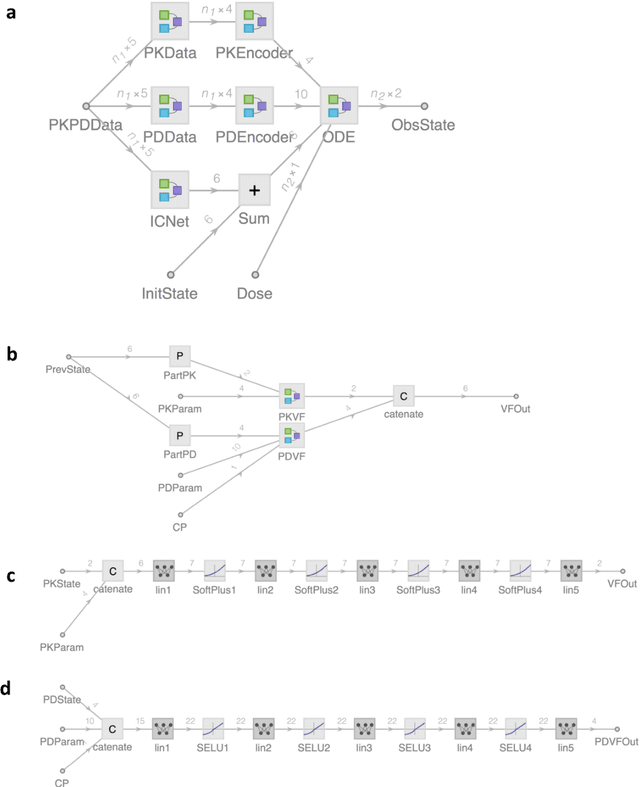
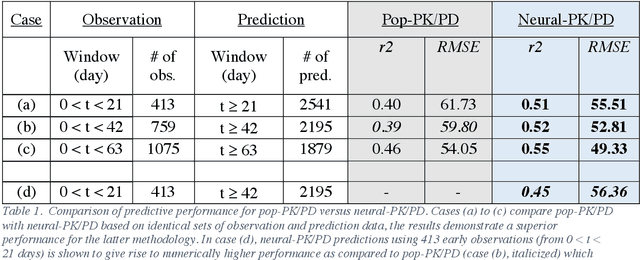
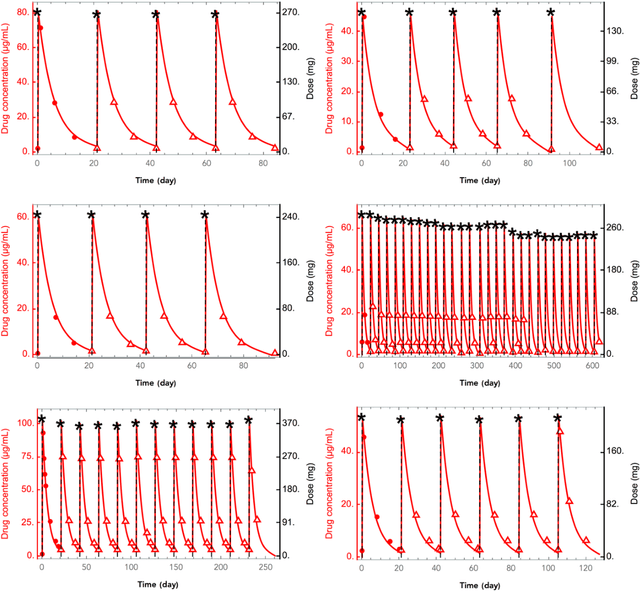
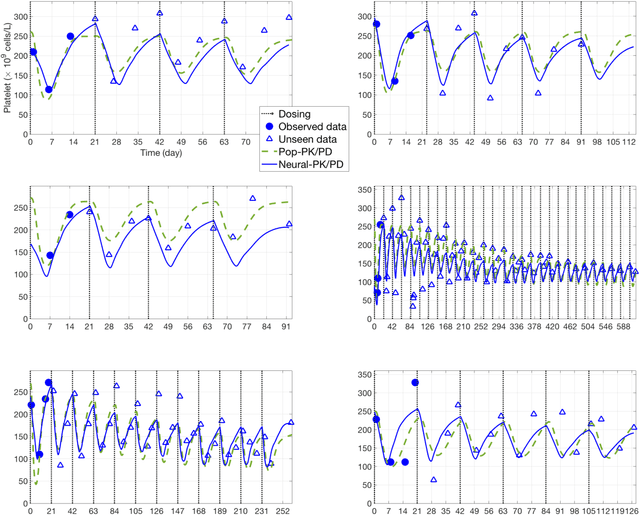
Abstract:The longitudinal analysis of patient response time course following doses of therapeutics is currently performed using Pharmacokinetic/Pharmacodynamic (PK/PD) methodologies, which requires significant human experience and expertise in the modeling of dynamical systems. By utilizing recent advancements in deep learning, we show that the governing differential equations can be learnt directly from longitudinal patient data. In particular, we propose a novel neural-PK/PD framework that combines key pharmacological principles with neural ordinary differential equations. We applied it to an analysis of drug concentration and platelet response from a clinical dataset consisting of over 600 patients. We show that the neural-PK/PD model improves upon a state-of-the-art model with respect to metrics for temporal prediction. Furthermore, by incorporating key PK/PD concepts into its architecture, the model can generalize and enable the simulations of patient responses to untested dosing regimens. These results demonstrate the potential of neural-PK/PD for automated predictive analytics of patient response time course.
Lung cancer screening with low-dose CT scans using a deep learning approach
Jun 01, 2019
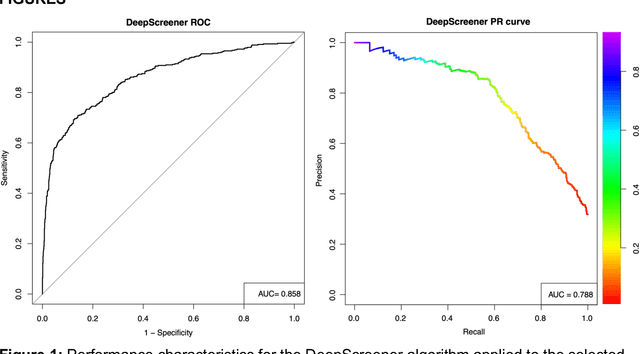
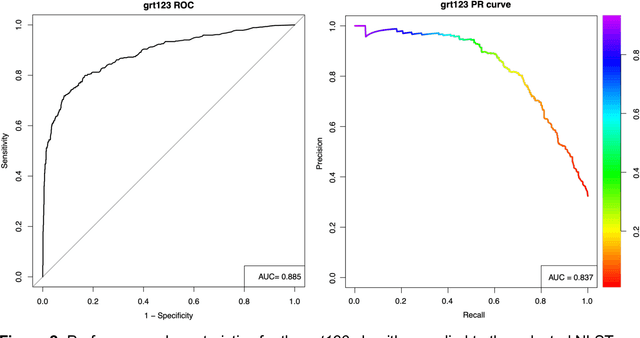
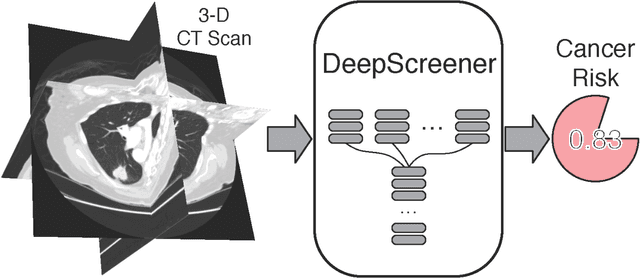
Abstract:Lung cancer is the leading cause of cancer deaths. Early detection through low-dose computed tomography (CT) screening has been shown to significantly reduce mortality but suffers from a high false positive rate that leads to unnecessary diagnostic procedures. Quantitative image analysis coupled to deep learning techniques has the potential to reduce this false positive rate. We conducted a computational analysis of 1449 low-dose CT studies drawn from the National Lung Screening Trial (NLST) cohort. We applied to this cohort our newly developed algorithm, DeepScreener, which is based on a novel deep learning approach. The algorithm, after the training process using about 3000 CT studies, does not require lung nodule annotations to conduct cancer prediction. The algorithm uses consecutive slices and multi-task features to determine whether a nodule is likely to be cancer, and a spatial pyramid to detect nodules at different scales. We find that the algorithm can predict a patient's cancer status from a volumetric lung CT image with high accuracy (78.2%, with area under the Receiver Operating Characteristic curve (AUC) of 0.858). Our preliminary framework ranked 16th of 1972 teams (top 1%) in the Data Science Bowl 2017 (DSB2017) competition, based on the challenge datasets. We report here the application of DeepScreener on an independent NLST test set. This study indicates that the deep learning approach has the potential to significantly reduce the false positive rate in lung cancer screening with low-dose CT scans.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge