Liyuan Sun
SkinGPT-X: A Self-Evolving Collaborative Multi-Agent System for Transparent and Trustworthy Dermatological Diagnosis
Mar 27, 2026Abstract:While recent advancements in Large Language Models have significantly advanced dermatological diagnosis, monolithic LLMs frequently struggle with fine-grained, large-scale multi-class diagnostic tasks and rare skin disease diagnosis owing to training data sparsity, while also lacking the interpretability and traceability essential for clinical reasoning. Although multi-agent systems can offer more transparent and explainable diagnostics, existing frameworks are primarily concentrated on Visual Question Answering and conversational tasks, and their heavy reliance on static knowledge bases restricts adaptability in complex real-world clinical settings. Here, we present SkinGPT-X, a multimodal collaborative multi-agent system for dermatological diagnosis integrated with a self-evolving dermatological memory mechanism. By simulating the diagnostic workflow of dermatologists and enabling continuous memory evolution, SkinGPT-X delivers transparent and trustworthy diagnostics for the management of complex and rare dermatological cases. To validate the robustness of SkinGPT-X, we design a three-tier comparative experiment. First, we benchmark SkinGPT-X against four state-of-the-art LLMs across four public datasets, demonstrating its state-of-the-art performance with a +9.6% accuracy improvement on DDI31 and +13% weighted F1 gain on Dermnet over the state-of-the-art model. Second, we construct a large-scale multi-class dataset covering 498 distinct dermatological categories to evaluate its fine-grained classification capabilities. Finally, we curate the rare skin disease dataset, the first benchmark to address the scarcity of clinical rare skin diseases which contains 564 clinical samples with eight rare dermatological diseases. On this dataset, SkinGPT-X achieves a +9.8% accuracy improvement, a +7.1% weighted F1 improvement, a +10% Cohen's Kappa improvement.
SkinCAP: A Multi-modal Dermatology Dataset Annotated with Rich Medical Captions
May 28, 2024



Abstract:With the widespread application of artificial intelligence (AI), particularly deep learning (DL) and vision-based large language models (VLLMs), in skin disease diagnosis, the need for interpretability becomes crucial. However, existing dermatology datasets are limited in their inclusion of concept-level meta-labels, and none offer rich medical descriptions in natural language. This deficiency impedes the advancement of LLM-based methods in dermatological diagnosis. To address this gap and provide a meticulously annotated dermatology dataset with comprehensive natural language descriptions, we introduce SkinCAP: a multi-modal dermatology dataset annotated with rich medical captions. SkinCAP comprises 4,000 images sourced from the Fitzpatrick 17k skin disease dataset and the Diverse Dermatology Images dataset, annotated by board-certified dermatologists to provide extensive medical descriptions and captions. Notably, SkinCAP represents the world's first such dataset and is publicly available at https://huggingface.co/datasets/joshuachou/SkinCAP.
Collaborative Teacher-Student Learning via Multiple Knowledge Transfer
Jan 27, 2021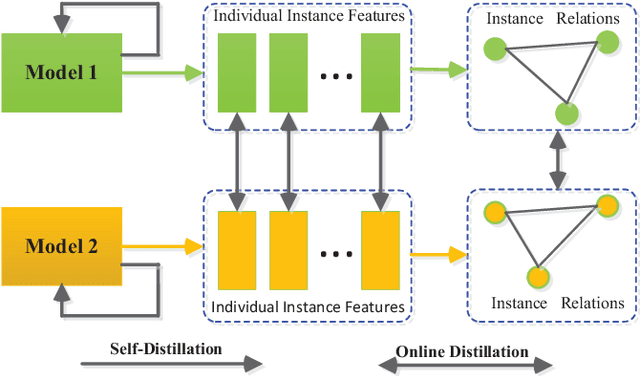

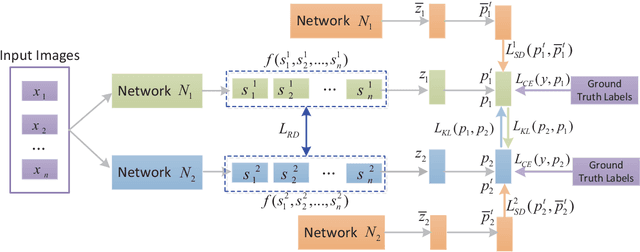
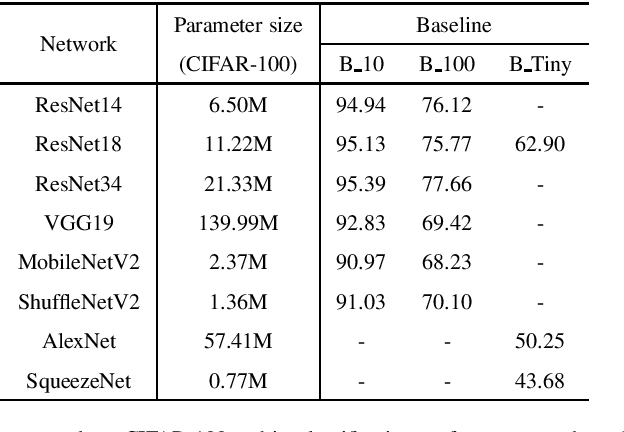
Abstract:Knowledge distillation (KD), as an efficient and effective model compression technique, has been receiving considerable attention in deep learning. The key to its success is to transfer knowledge from a large teacher network to a small student one. However, most of the existing knowledge distillation methods consider only one type of knowledge learned from either instance features or instance relations via a specific distillation strategy in teacher-student learning. There are few works that explore the idea of transferring different types of knowledge with different distillation strategies in a unified framework. Moreover, the frequently used offline distillation suffers from a limited learning capacity due to the fixed teacher-student architecture. In this paper we propose a collaborative teacher-student learning via multiple knowledge transfer (CTSL-MKT) that prompts both self-learning and collaborative learning. It allows multiple students learn knowledge from both individual instances and instance relations in a collaborative way. While learning from themselves with self-distillation, they can also guide each other via online distillation. The experiments and ablation studies on four image datasets demonstrate that the proposed CTSL-MKT significantly outperforms the state-of-the-art KD methods.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge