Amar Kumar
Pixels Under Pressure: Exploring Fine-Tuning Paradigms for Foundation Models in High-Resolution Medical Imaging
Aug 19, 2025



Abstract:Advancements in diffusion-based foundation models have improved text-to-image generation, yet most efforts have been limited to low-resolution settings. As high-resolution image synthesis becomes increasingly essential for various applications, particularly in medical imaging domains, fine-tuning emerges as a crucial mechanism for adapting these powerful pre-trained models to task-specific requirements and data distributions. In this work, we present a systematic study, examining the impact of various fine-tuning techniques on image generation quality when scaling to high resolution 512x512 pixels. We benchmark a diverse set of fine-tuning methods, including full fine-tuning strategies and parameter-efficient fine-tuning (PEFT). We dissect how different fine-tuning methods influence key quality metrics, including Fr\'echet Inception Distance (FID), Vendi score, and prompt-image alignment. We also evaluate the utility of generated images in a downstream classification task under data-scarce conditions, demonstrating that specific fine-tuning strategies improve both generation fidelity and downstream performance when synthetic images are used for classifier training and evaluation on real images. Our code is accessible through the project website - https://tehraninasab.github.io/PixelUPressure/.
AURA: A Multi-Modal Medical Agent for Understanding, Reasoning & Annotation
Jul 22, 2025



Abstract:Recent advancements in Large Language Models (LLMs) have catalyzed a paradigm shift from static prediction systems to agentic AI agents capable of reasoning, interacting with tools, and adapting to complex tasks. While LLM-based agentic systems have shown promise across many domains, their application to medical imaging remains in its infancy. In this work, we introduce AURA, the first visual linguistic explainability agent designed specifically for comprehensive analysis, explanation, and evaluation of medical images. By enabling dynamic interactions, contextual explanations, and hypothesis testing, AURA represents a significant advancement toward more transparent, adaptable, and clinically aligned AI systems. We highlight the promise of agentic AI in transforming medical image analysis from static predictions to interactive decision support. Leveraging Qwen-32B, an LLM-based architecture, AURA integrates a modular toolbox comprising: (i) a segmentation suite with phase grounding, pathology segmentation, and anatomy segmentation to localize clinically meaningful regions; (ii) a counterfactual image-generation module that supports reasoning through image-level explanations; and (iii) a set of evaluation tools including pixel-wise difference-map analysis, classification, and advanced state-of-the-art components to assess diagnostic relevance and visual interpretability.
Language-Guided Trajectory Traversal in Disentangled Stable Diffusion Latent Space for Factorized Medical Image Generation
Mar 30, 2025



Abstract:Text-to-image diffusion models have demonstrated a remarkable ability to generate photorealistic images from natural language prompts. These high-resolution, language-guided synthesized images are essential for the explainability of disease or exploring causal relationships. However, their potential for disentangling and controlling latent factors of variation in specialized domains like medical imaging remains under-explored. In this work, we present the first investigation of the power of pre-trained vision-language foundation models, once fine-tuned on medical image datasets, to perform latent disentanglement for factorized medical image generation and interpolation. Through extensive experiments on chest X-ray and skin datasets, we illustrate that fine-tuned, language-guided Stable Diffusion inherently learns to factorize key attributes for image generation, such as the patient's anatomical structures or disease diagnostic features. We devise a framework to identify, isolate, and manipulate key attributes through latent space trajectory traversal of generative models, facilitating precise control over medical image synthesis.
RL4Med-DDPO: Reinforcement Learning for Controlled Guidance Towards Diverse Medical Image Generation using Vision-Language Foundation Models
Mar 20, 2025



Abstract:Vision-Language Foundation Models (VLFM) have shown a tremendous increase in performance in terms of generating high-resolution, photorealistic natural images. While VLFMs show a rich understanding of semantic content across modalities, they often struggle with fine-grained alignment tasks that require precise correspondence between image regions and textual descriptions a limitation in medical imaging, where accurate localization and detection of clinical features are essential for diagnosis and analysis. To address this issue, we propose a multi-stage architecture where a pre-trained VLFM provides a cursory semantic understanding, while a reinforcement learning (RL) algorithm refines the alignment through an iterative process that optimizes for understanding semantic context. The reward signal is designed to align the semantic information of the text with synthesized images. We demonstrate the effectiveness of our method on a medical imaging skin dataset where the generated images exhibit improved generation quality and alignment with prompt over the fine-tuned Stable Diffusion. We also show that the synthesized samples could be used to improve disease classifier performance for underrepresented subgroups through augmentation.
DeCoDEx: Confounder Detector Guidance for Improved Diffusion-based Counterfactual Explanations
May 15, 2024

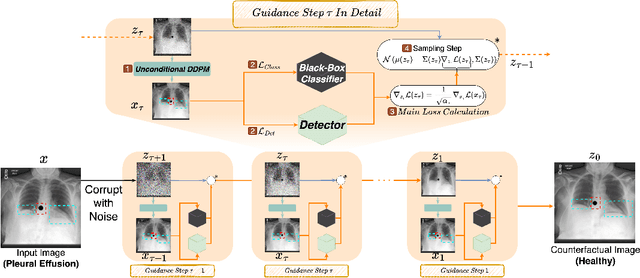

Abstract:Deep learning classifiers are prone to latching onto dominant confounders present in a dataset rather than on the causal markers associated with the target class, leading to poor generalization and biased predictions. Although explainability via counterfactual image generation has been successful at exposing the problem, bias mitigation strategies that permit accurate explainability in the presence of dominant and diverse artifacts remain unsolved. In this work, we propose the DeCoDEx framework and show how an external, pre-trained binary artifact detector can be leveraged during inference to guide a diffusion-based counterfactual image generator towards accurate explainability. Experiments on the CheXpert dataset, using both synthetic artifacts and real visual artifacts (support devices), show that the proposed method successfully synthesizes the counterfactual images that change the causal pathology markers associated with Pleural Effusion while preserving or ignoring the visual artifacts. Augmentation of ERM and Group-DRO classifiers with the DeCoDEx generated images substantially improves the results across underrepresented groups that are out of distribution for each class. The code is made publicly available at https://github.com/NimaFathi/DeCoDEx.
Debiasing Counterfactuals In the Presence of Spurious Correlations
Aug 21, 2023



Abstract:Deep learning models can perform well in complex medical imaging classification tasks, even when basing their conclusions on spurious correlations (i.e. confounders), should they be prevalent in the training dataset, rather than on the causal image markers of interest. This would thereby limit their ability to generalize across the population. Explainability based on counterfactual image generation can be used to expose the confounders but does not provide a strategy to mitigate the bias. In this work, we introduce the first end-to-end training framework that integrates both (i) popular debiasing classifiers (e.g. distributionally robust optimization (DRO)) to avoid latching onto the spurious correlations and (ii) counterfactual image generation to unveil generalizable imaging markers of relevance to the task. Additionally, we propose a novel metric, Spurious Correlation Latching Score (SCLS), to quantify the extent of the classifier reliance on the spurious correlation as exposed by the counterfactual images. Through comprehensive experiments on two public datasets (with the simulated and real visual artifacts), we demonstrate that the debiasing method: (i) learns generalizable markers across the population, and (ii) successfully ignores spurious correlations and focuses on the underlying disease pathology.
Counterfactual Image Synthesis for Discovery of Personalized Predictive Image Markers
Aug 03, 2022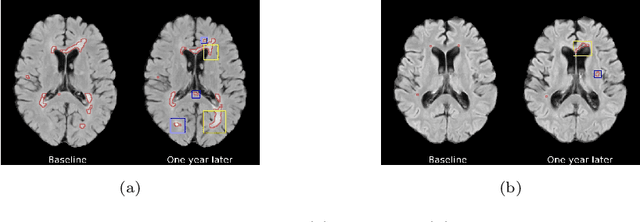

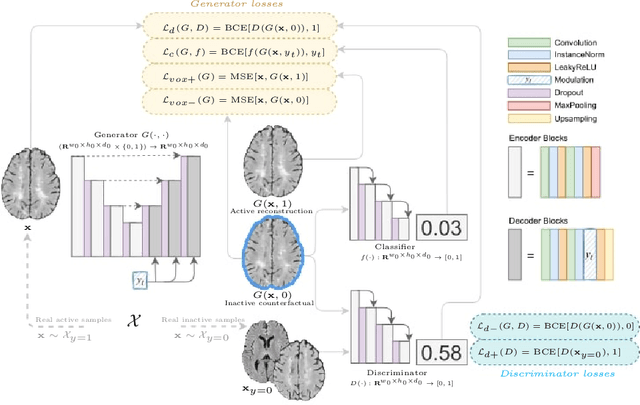

Abstract:The discovery of patient-specific imaging markers that are predictive of future disease outcomes can help us better understand individual-level heterogeneity of disease evolution. In fact, deep learning models that can provide data-driven personalized markers are much more likely to be adopted in medical practice. In this work, we demonstrate that data-driven biomarker discovery can be achieved through a counterfactual synthesis process. We show how a deep conditional generative model can be used to perturb local imaging features in baseline images that are pertinent to subject-specific future disease evolution and result in a counterfactual image that is expected to have a different future outcome. Candidate biomarkers, therefore, result from examining the set of features that are perturbed in this process. Through several experiments on a large-scale, multi-scanner, multi-center multiple sclerosis (MS) clinical trial magnetic resonance imaging (MRI) dataset of relapsing-remitting (RRMS) patients, we demonstrate that our model produces counterfactuals with changes in imaging features that reflect established clinical markers predictive of future MRI lesional activity at the population level. Additional qualitative results illustrate that our model has the potential to discover novel and subject-specific predictive markers of future activity.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge