Egor Panfilov
Active Sensing of Knee Osteoarthritis Progression with Reinforcement Learning
Aug 05, 2024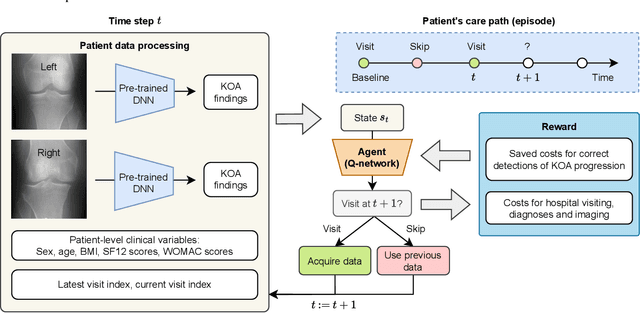
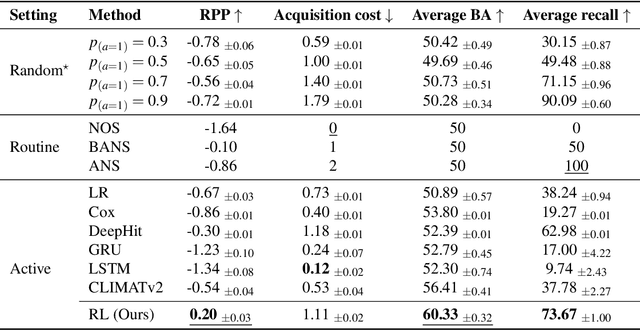
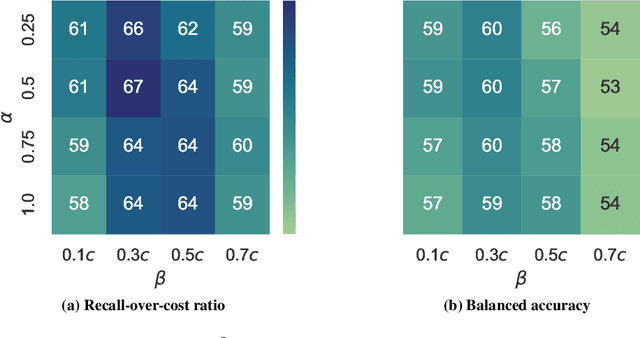
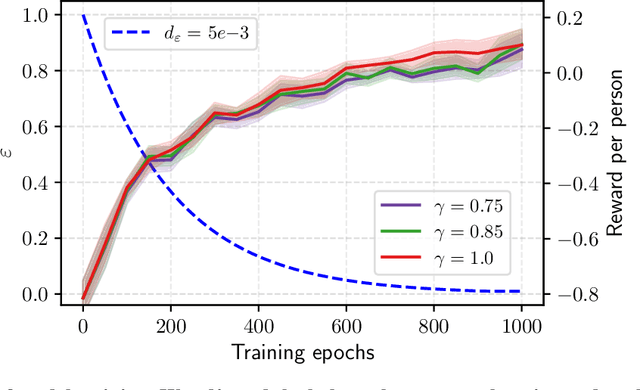
Abstract:Osteoarthritis (OA) is the most common musculoskeletal disease, which has no cure. Knee OA (KOA) is one of the highest causes of disability worldwide, and it costs billions of United States dollars to the global community. Prediction of KOA progression has been of high interest to the community for years, as it can advance treatment development through more efficient clinical trials and improve patient outcomes through more efficient healthcare utilization. Existing approaches for predicting KOA, however, are predominantly static, i.e. consider data from a single time point to predict progression many years into the future, and knee level, i.e. consider progression in a single joint only. Due to these and related reasons, these methods fail to deliver the level of predictive performance, which is sufficient to result in cost savings and better patient outcomes. Collecting extensive data from all patients on a regular basis could address the issue, but it is limited by the high cost at a population level. In this work, we propose to go beyond static prediction models in OA, and bring a novel Active Sensing (AS) approach, designed to dynamically follow up patients with the objective of maximizing the number of informative data acquisitions, while minimizing their total cost over a period of time. Our approach is based on Reinforcement Learning (RL), and it leverages a novel reward function designed specifically for AS of disease progression in more than one part of a human body. Our method is end-to-end, relies on multi-modal Deep Learning, and requires no human input at inference time. Throughout an exhaustive experimental evaluation, we show that using RL can provide a higher monetary benefit when compared to state-of-the-art baselines.
End-To-End Prediction of Knee Osteoarthritis Progression With Multi-Modal Transformers
Jul 03, 2023



Abstract:Knee Osteoarthritis (KOA) is a highly prevalent chronic musculoskeletal condition with no currently available treatment. The manifestation of KOA is heterogeneous and prediction of its progression is challenging. Current literature suggests that the use of multi-modal data and advanced modeling methods, such as the ones based on Deep Learning, has promise in tackling this challenge. To date, however, the evidence on the efficacy of this approach is limited. In this study, we leveraged recent advances in Deep Learning and, using a Transformer approach, developed a unified framework for the multi-modal fusion of knee imaging data. Subsequently, we analyzed its performance across a range of scenarios by investigating multiple progression horizons -- from short-term to long-term. We report our findings using a large cohort (n=2421-3967) derived from the Osteoarthritis Initiative dataset. We show that structural knee MRI allows identifying radiographic KOA progressors on par with multi-modal fusion approaches, achieving an area under the ROC curve (ROC AUC) of 0.70-0.76 and Average Precision (AP) of 0.15-0.54 in 2-8 year horizons. Progression within 1 year was better predicted with a multi-modal method using X-ray, structural, and compositional MR images -- ROC AUC of 0.76(0.04), AP of 0.13(0.04) -- or via clinical data. Our follow-up analysis generally shows that prediction from the imaging data is more accurate for post-traumatic subjects, and we further investigate which subject subgroups may benefit the most. The present study provides novel insights into multi-modal imaging of KOA and brings a unified data-driven framework for studying its progression in an end-to-end manner, providing new tools for the design of more efficient clinical trials. The source code of our framework and the pre-trained models are made publicly available.
Predicting Knee Osteoarthritis Progression from Structural MRI using Deep Learning
Jan 26, 2022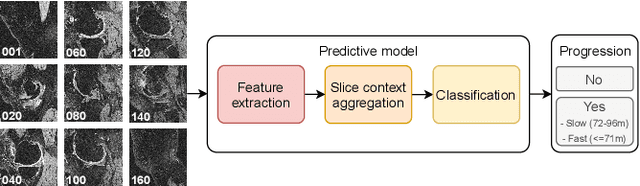
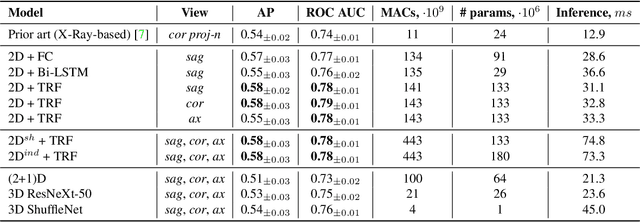

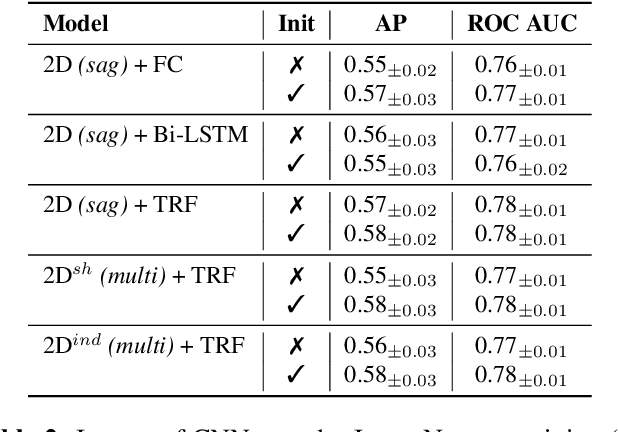
Abstract:Accurate prediction of knee osteoarthritis (KOA) progression from structural MRI has a potential to enhance disease understanding and support clinical trials. Prior art focused on manually designed imaging biomarkers, which may not fully exploit all disease-related information present in MRI scan. In contrast, our method learns relevant representations from raw data end-to-end using Deep Learning, and uses them for progression prediction. The method employs a 2D CNN to process the data slice-wise and aggregate the extracted features using a Transformer. Evaluated on a large cohort (n=4,866), the proposed method outperforms conventional 2D and 3D CNN-based models and achieves average precision of $0.58\pm0.03$ and ROC AUC of $0.78\pm0.01$. This paper sets a baseline on end-to-end KOA progression prediction from structural MRI. Our code is publicly available at https://github.com/MIPT-Oulu/OAProgressionMR.
Improving Robustness of Deep Learning Based Knee MRI Segmentation: Mixup and Adversarial Domain Adaptation
Aug 14, 2019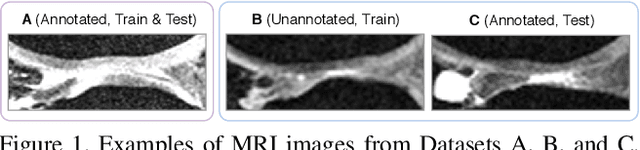
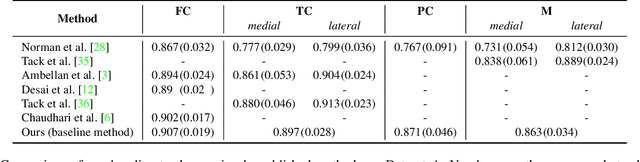
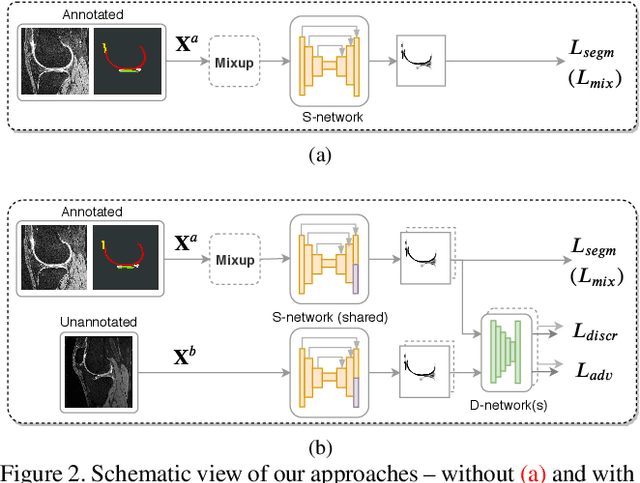
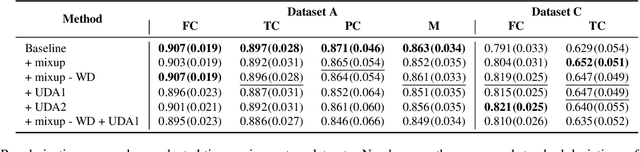
Abstract:Degeneration of articular cartilage (AC) is actively studied in knee osteoarthritis (OA) research via magnetic resonance imaging (MRI). Segmentation of AC tissues from MRI data is an essential step in quantification of their damage. Deep learning (DL) based methods have shown potential in this realm and are the current state-of-the-art, however, their robustness to heterogeneity of MRI acquisition settings remains an open problem. In this study, we investigated two modern regularization techniques -- mixup and adversarial unsupervised domain adaptation (UDA) -- to improve the robustness of DL-based knee cartilage segmentation to new MRI acquisition settings. Our validation setup included two datasets produced by different MRI scanners and using distinct data acquisition protocols. We assessed the robustness of automatic segmentation by comparing mixup and UDA approaches to a strong baseline method at different OA severity stages and, additionally, in relation to anatomical locations. Our results showed that for moderate changes in knee MRI data acquisition settings both approaches may provide notable improvements in the robustness, which are consistent for all stages of the disease and affect the clinically important areas of the knee joint. However, mixup may be considered as a recommended approach, since it is more computationally efficient and does not require additional data from the target acquisition setup.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge