Stanislav Motyka
WALINET: A water and lipid identification convolutional Neural Network for nuisance signal removal in 1H MR Spectroscopic Imaging
Oct 01, 2024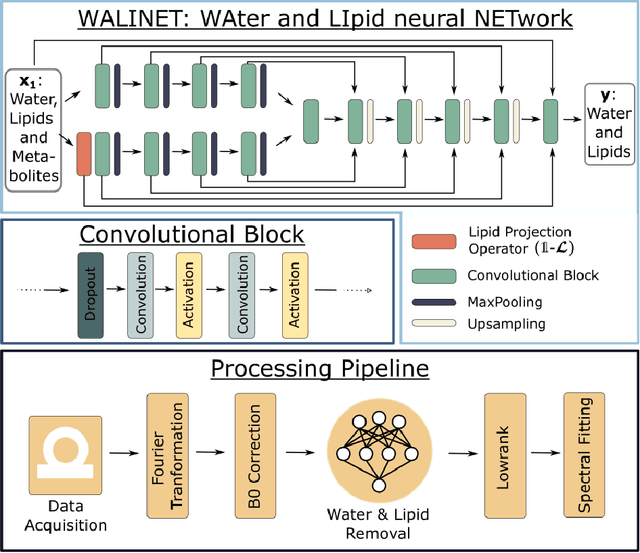

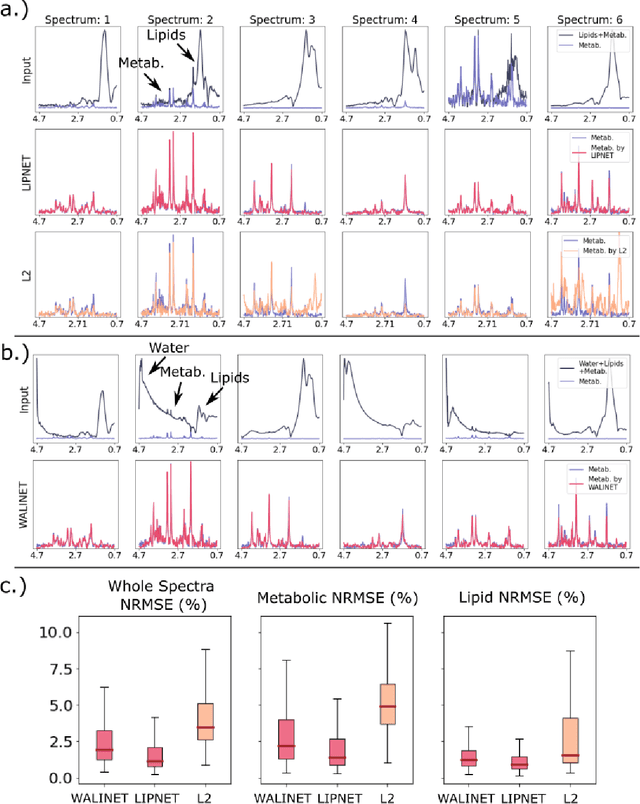
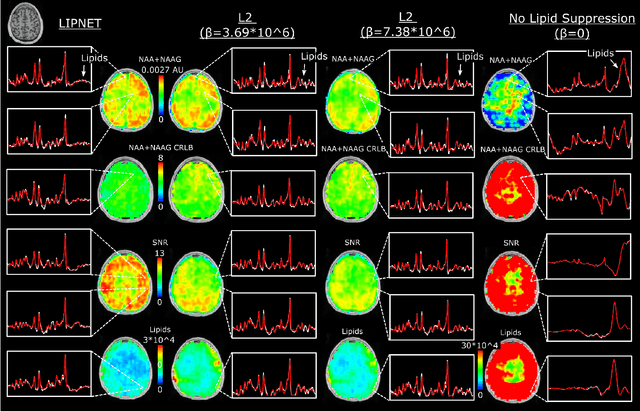
Abstract:Purpose. Proton Magnetic Resonance Spectroscopic Imaging (1H-MRSI) provides non-invasive spectral-spatial mapping of metabolism. However, long-standing problems in whole-brain 1H-MRSI are spectral overlap of metabolite peaks with large lipid signal from scalp, and overwhelming water signal that distorts spectra. Fast and effective methods are needed for high-resolution 1H-MRSI to accurately remove lipid and water signals while preserving the metabolite signal. The potential of supervised neural networks for this task remains unexplored, despite their success for other MRSI processing. Methods. We introduce a deep-learning method based on a modified Y-NET network for water and lipid removal in whole-brain 1H-MRSI. The WALINET (WAter and LIpid neural NETwork) was compared to conventional methods such as the state-of-the-art lipid L2 regularization and Hankel-Lanczos singular value decomposition (HLSVD) water suppression. Methods were evaluated on simulated and in-vivo whole-brain MRSI using NMRSE, SNR, CRLB, and FWHM metrics. Results. WALINET is significantly faster and needs 8s for high-resolution whole-brain MRSI, compared to 42 minutes for conventional HLSVD+L2. Quantitative analysis shows WALINET has better performance than HLSVD+L2: 1) more lipid removal with 41% lower NRMSE, 2) better metabolite signal preservation with 71% lower NRMSE in simulated data, 155% higher SNR and 50% lower CRLB in in-vivo data. Metabolic maps obtained by WALINET in healthy subjects and patients show better gray/white-matter contrast with more visible structural details. Conclusions. WALINET has superior performance for nuisance signal removal and metabolite quantification on whole-brain 1H-MRSI compared to conventional state-of-the-art techniques. This represents a new application of deep-learning for MRSI processing, with potential for automated high-throughput workflow.
Deep-ER: Deep Learning ECCENTRIC Reconstruction for fast high-resolution neurometabolic imaging
Sep 26, 2024
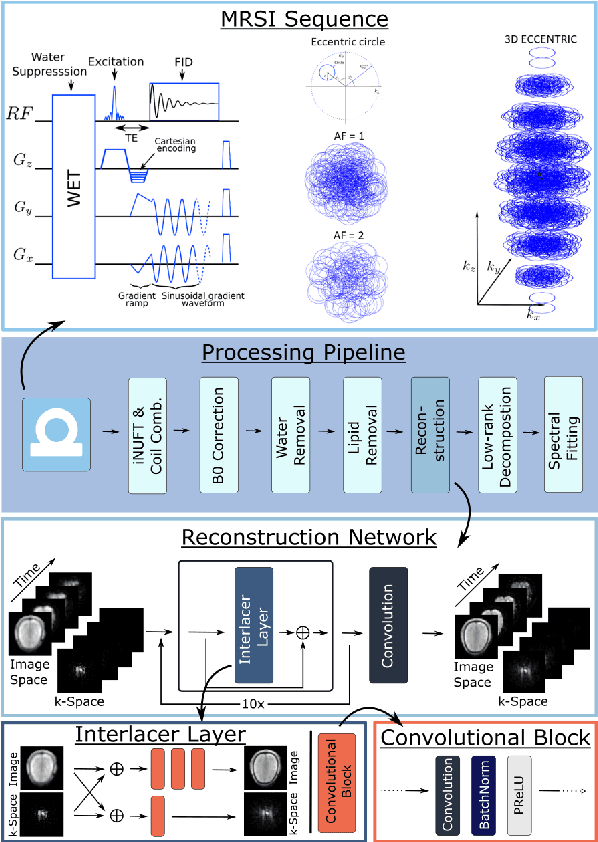
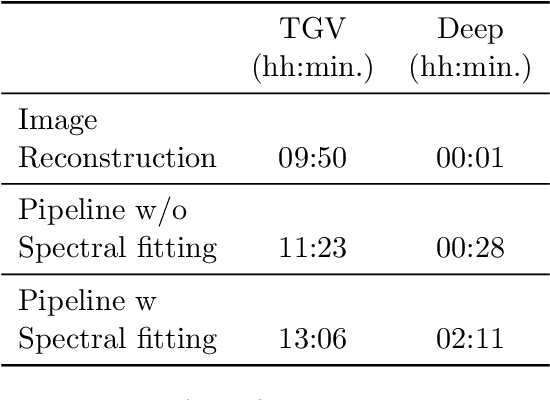

Abstract:Introduction: Altered neurometabolism is an important pathological mechanism in many neurological diseases and brain cancer, which can be mapped non-invasively by Magnetic Resonance Spectroscopic Imaging (MRSI). Advanced MRSI using non-cartesian compressed-sense acquisition enables fast high-resolution metabolic imaging but has lengthy reconstruction times that limits throughput and needs expert user interaction. Here, we present a robust and efficient Deep Learning reconstruction to obtain high-quality metabolic maps. Methods: Fast high-resolution whole-brain metabolic imaging was performed at 3.4 mm$^3$ isotropic resolution with acquisition times between 4:11-9:21 min:s using ECCENTRIC pulse sequence on a 7T MRI scanner. Data were acquired in a high-resolution phantom and 27 human participants, including 22 healthy volunteers and 5 glioma patients. A deep neural network using recurring interlaced convolutional layers with joint dual-space feature representation was developed for deep learning ECCENTRIC reconstruction (Deep-ER). 21 subjects were used for training and 6 subjects for testing. Deep-ER performance was compared to conventional iterative Total Generalized Variation reconstruction using image and spectral quality metrics. Results: Deep-ER demonstrated 600-fold faster reconstruction than conventional methods, providing improved spatial-spectral quality and metabolite quantification with 12%-45% (P<0.05) higher signal-to-noise and 8%-50% (P<0.05) smaller Cramer-Rao lower bounds. Metabolic images clearly visualize glioma tumor heterogeneity and boundary. Conclusion: Deep-ER provides efficient and robust reconstruction for sparse-sampled MRSI. The accelerated acquisition-reconstruction MRSI is compatible with high-throughput imaging workflow. It is expected that such improved performance will facilitate basic and clinical MRSI applications.
Predicting dynamic, motion-related changes in B0 field in the brain at a 7 T MRI using a subject-specific fine-tuned U-net
Apr 17, 2023Abstract:Subject movement during the magnetic resonance examination is inevitable and causes not only image artefacts but also deteriorates the homogeneity of the main magnetic field (B0), which is a prerequisite for high quality data. Thus, characterization of changes to B0, e.g. induced by patient movement, is important for MR applications that are prone to B0 inhomogeneities. We propose a deep learning based method to predict such changes within the brain from the change of the head position to facilitate retrospective or even real-time correction. A 3D U-net was trained on in vivo brain 7T MRI data. The input consisted of B0 maps and anatomical images at an initial position, and anatomical images at a different head position (obtained by applying a rigid-body transformation on the initial anatomical image). The output consisted of B0 maps at the new head positions. We further fine-tuned the network weights to each subject by measuring a limited number of head positions of the given subject, and trained the U-net with these data. Our approach was compared to established dynamic B0 field mapping via interleaved navigators, which suffer from limited spatial resolution and the need for undesirable sequence modifications. Qualitative and quantitative comparison showed similar performance between an interleaved navigator-equivalent method and proposed method. We therefore conclude that it is feasible to predict B0 maps from rigid subject movement and, when combined with external tracking hardware, this information could be used to improve the quality of magnetic resonance acquisitions without the use of navigators.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge