John A. Detre
ICHOR: A Robust Representation Learning Approach for ASL CBF Maps with Self-Supervised Masked Autoencoders
Mar 05, 2026Abstract:Arterial spin labeling (ASL) perfusion MRI allows direct quantification of regional cerebral blood flow (CBF) without exogenous contrast, enabling noninvasive measurements that can be repeated without constraints imposed by contrast injection. ASL is increasingly acquired in research studies and clinical MRI protocols. Building on successes in structural imaging, recent efforts have implemented deep learning based methods to improve image quality, enable automated quality control, and derive robust quantitative and predictive biomarkers with ASL derived CBF. However, progress has been limited by variable image quality, substantial inter-site, vendor and protocol differences, and limited availability of labeled datasets needed to train models that generalize across cohorts. To address these challenges, we introduce ICHOR, a self supervised pre-training approach for ASL CBF maps that learns transferable representations using 3D masked autoencoders. ICHOR is pretrained via masked image modeling using a Vision Transformer backbone and can be used as a general-purpose encoder for downstream ASL tasks. For pre-training, we curated one of the largest ASL datasets to date, comprising 11,405 ASL CBF scans from 14 studies spanning multiple sites and acquisition protocols. We evaluated the pre-trained ICHOR encoder on three downstream diagnostic classification tasks and one ASL CBF map quality prediction regression task. Across all evaluations, ICHOR outperformed existing neuroimaging self-supervised pre-training methods adapted to ASL. Pre-trained weights and code will be made publicly available.
Improved Segmentation of Deep Sulci in Cortical Gray Matter Using a Deep Learning Framework Incorporating Laplace's Equation
Mar 03, 2023



Abstract:When developing tools for automated cortical segmentation, the ability to produce topologically correct segmentations is important in order to compute geometrically valid morphometry measures. In practice, accurate cortical segmentation is challenged by image artifacts and the highly convoluted anatomy of the cortex itself. To address this, we propose a novel deep learning-based cortical segmentation method in which prior knowledge about the geometry of the cortex is incorporated into the network during the training process. We design a loss function which uses the theory of Laplace's equation applied to the cortex to locally penalize unresolved boundaries between tightly folded sulci. Using an ex vivo MRI dataset of human medial temporal lobe specimens, we demonstrate that our approach outperforms baseline segmentation networks, both quantitatively and qualitatively.
Machine learning in cardiovascular flows modeling: Predicting pulse wave propagation from non-invasive clinical measurements using physics-informed deep learning
May 13, 2019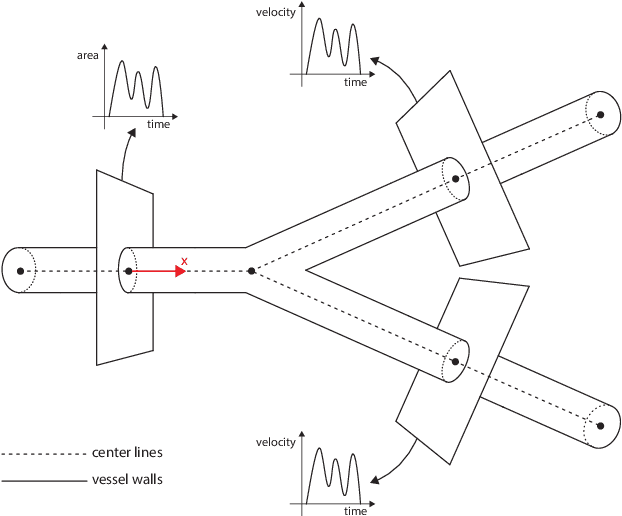

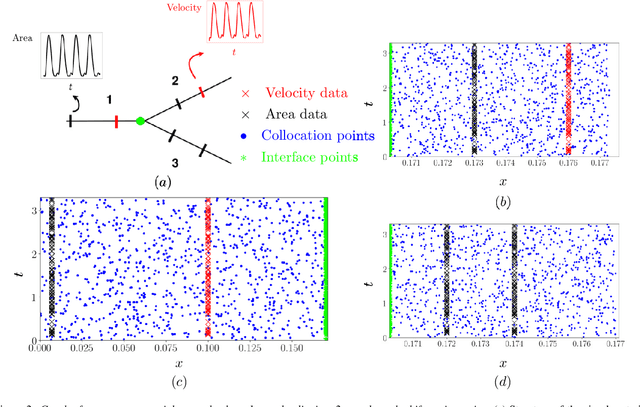
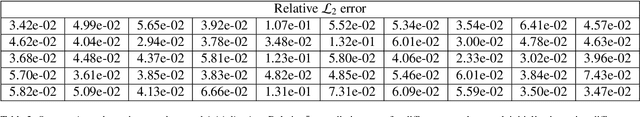
Abstract:Advances in computational science offer a principled pipeline for predictive modeling of cardiovascular flows and aspire to provide a valuable tool for monitoring, diagnostics and surgical planning. Such models can be nowadays deployed on large patient-specific topologies of systemic arterial networks and return detailed predictions on flow patterns, wall shear stresses, and pulse wave propagation. However, their success heavily relies on tedious pre-processing and calibration procedures that typically induce a significant computational cost, thus hampering their clinical applicability. In this work we put forth a machine learning framework that enables the seamless synthesis of non-invasive in-vivo measurement techniques and computational flow dynamics models derived from first physical principles. We illustrate this new paradigm by showing how one-dimensional models of pulsatile flow can be used to constrain the output of deep neural networks such that their predictions satisfy the conservation of mass and momentum principles. Once trained on noisy and scattered clinical data of flow and wall displacement, these networks can return physically consistent predictions for velocity, pressure and wall displacement pulse wave propagation, all without the need to employ conventional simulators. A simple post-processing of these outputs can also provide a cheap and effective way for estimating Windkessel model parameters that are required for the calibration of traditional computational models. The effectiveness of the proposed techniques is demonstrated through a series of prototype benchmarks, as well as a realistic clinical case involving in-vivo measurements near the aorta/carotid bifurcation of a healthy human subject.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge