Camel Tanougast
Explainable, Domain-Adaptive, and Federated Artificial Intelligence in Medicine
Nov 17, 2022



Abstract:Artificial intelligence (AI) continues to transform data analysis in many domains. Progress in each domain is driven by a growing body of annotated data, increased computational resources, and technological innovations. In medicine, the sensitivity of the data, the complexity of the tasks, the potentially high stakes, and a requirement of accountability give rise to a particular set of challenges. In this review, we focus on three key methodological approaches that address some of the particular challenges in AI-driven medical decision making. (1) Explainable AI aims to produce a human-interpretable justification for each output. Such models increase confidence if the results appear plausible and match the clinicians expectations. However, the absence of a plausible explanation does not imply an inaccurate model. Especially in highly non-linear, complex models that are tuned to maximize accuracy, such interpretable representations only reflect a small portion of the justification. (2) Domain adaptation and transfer learning enable AI models to be trained and applied across multiple domains. For example, a classification task based on images acquired on different acquisition hardware. (3) Federated learning enables learning large-scale models without exposing sensitive personal health information. Unlike centralized AI learning, where the centralized learning machine has access to the entire training data, the federated learning process iteratively updates models across multiple sites by exchanging only parameter updates, not personal health data. This narrative review covers the basic concepts, highlights relevant corner-stone and state-of-the-art research in the field, and discusses perspectives.
Can autism be diagnosed with AI?
Jun 05, 2022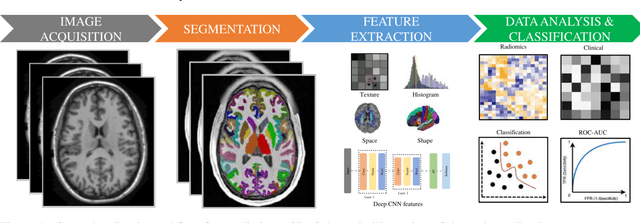
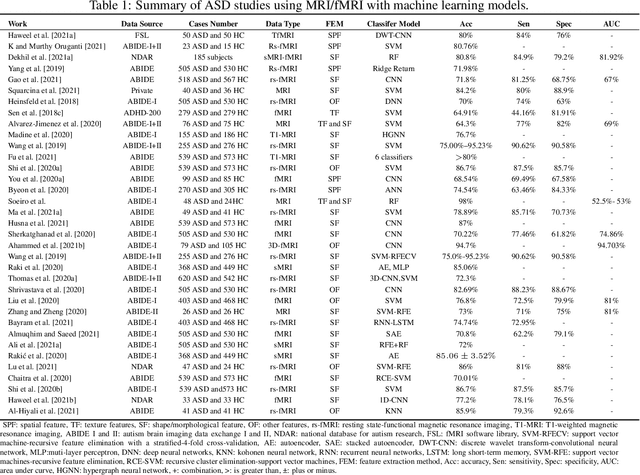

Abstract:Radiomics with deep learning models have become popular in computer-aided diagnosis and have outperformed human experts on many clinical tasks. Specifically, radiomic models based on artificial intelligence (AI) are using medical data (i.e., images, molecular data, clinical variables, etc.) for predicting clinical tasks like Autism Spectrum Disorder (ASD). In this review, we summarized and discussed the radiomic techniques used for ASD analysis. Currently, the limited radiomic work of ASD is related to variation of morphological features of brain thickness that is different from texture analysis. These techniques are based on imaging shape features that can be used with predictive models for predicting ASD. This review explores the progress of ASD-based radiomics with a brief description of ASD and the current non-invasive technique used to classify between ASD and Healthy Control (HC) subjects. With AI, new radiomic models using the deep learning techniques will be also described. To consider the texture analysis with deep CNNs, more investigations are suggested to be integrated with additional validation steps on various MRI sites.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge