Anjula De Silva
Improving Neonatal Care: An Active Dry-Contact Electrode-based Continuous EEG Monitoring System with Seizure Detection
Mar 30, 2025Abstract:Objective: Neonates are highly susceptible to seizures, which can have severe long-term consequences if undetected and left untreated. Early detection is crucial and typically requires continuous electroencephalography (EEG) monitoring in a hospital setting, which is costly, inconvenient, and requires specialized experts for diagnosis. In this work, we propose a new low-cost active dry-contact electrode-based adjustable EEG headset, a new explainable deep learning model to detect neonatal seizures, and an advanced signal processing algorithm to remove artifacts to address the key aspects that lead to the underdiagnosis of neonatal seizures. Methods: EEG signals are acquired through active electrodes and processed using a custom-designed analog front end (AFE) that filters and digitizes the captured EEG signals. The adjustable headset is designed using three-dimensional (3D) printing and laser cutting to fit a wide range of head sizes. A deep learning model is developed to classify seizure and non-seizure epochs in real-time. Furthermore, a separate multimodal deep learning model is designed to remove noise artifacts. The device is tested on a pediatric patient with absence seizures in a hospital setting. Simultaneous recordings are captured using both the custom device and the commercial wet electrode device available in the hospital for comparison. Results: The signals obtained using our custom design and a commercial device show a high correlation (>0.8). Further analysis using signal-to-noise ratio values shows that our device can mitigate noise similar to the commercial device. The proposed deep learning model has improvements in accuracy and recall by 2.76% and 16.33%, respectively, compared to the state-of-the-art. Furthermore, the developed artifact removal algorithm can identify and remove artifacts while keeping seizure patterns intact.
A Novel Transfer Learning-Based Approach for Screening Pre-existing Heart Diseases Using Synchronized ECG Signals and Heart Sounds
Feb 14, 2021


Abstract:Diagnosing pre-existing heart diseases early in life is important as it helps prevent complications such as pulmonary hypertension, heart rhythm problems, blood clots, heart failure and sudden cardiac arrest. To identify such diseases, phonocardiogram (PCG) and electrocardiogram (ECG) waveforms convey important information. Therefore, effectively using these two modalities of data has the potential to improve the disease screening process. We evaluate this hypothesis on a subset of the PhysioNet Challenge 2016 Dataset which contains simultaneously acquired PCG and ECG recordings. Our novel Dual-Convolutional Neural Network based approach uses transfer learning to tackle the problem of having limited amounts of simultaneous PCG and ECG data that is publicly available, while having the potential to adapt to larger datasets. In addition, we introduce two main evaluation frameworks named record-wise and sample-wise evaluation which leads to a rich performance evaluation for the transfer learning approach. Comparisons with methods which used single or dual modality data show that our method can lead to better performance. Furthermore, our results show that individually collected ECG or PCG waveforms are able to provide transferable features which could effectively help to make use of a limited number of synchronized PCG and ECG waveforms and still achieve significant classification performance.
A Thickness Sensitive Vessel Extraction Framework for Retinal and Conjunctival Vascular Tortuosity Analysis
Jan 02, 2021
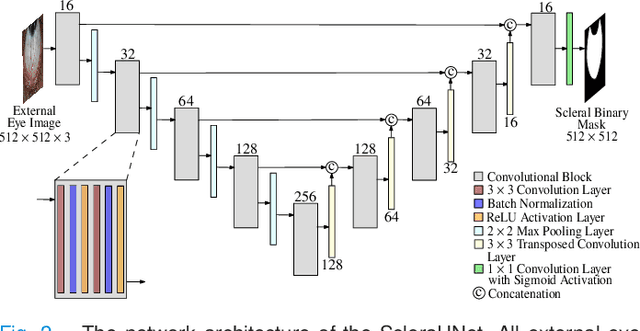
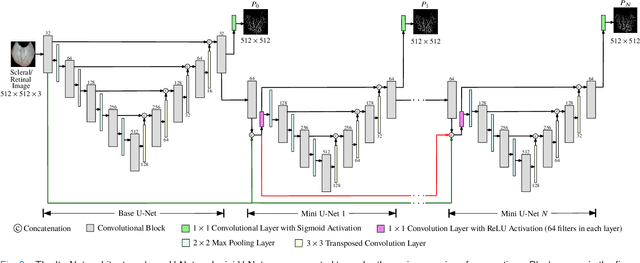

Abstract:Systemic diseases such as diabetes, hypertension, atherosclerosis are among the leading causes of annual human mortality rate. It is suggested that retinal and conjunctival vascular tortuosity is a potential biomarker for such systemic diseases. Most importantly, it is observed that the tortuosity depends on the thickness of these vessels. Therefore, selective calculation of tortuosity within specific vessel thicknesses is required depending on the disease being analysed. In this paper, we propose a thickness sensitive vessel extraction framework that is primarily applicable for studies related to retinal and conjunctival vascular tortuosity. The framework uses a Convolutional Neural Network based on the IterNet architecture to obtain probability maps of the entire vasculature. They are then processed by a multi-scale vessel enhancement technique that exploits both fine and coarse structural vascular details of these probability maps in order to extract vessels of specified thicknesses. We evaluated the proposed framework on four datasets including DRIVE and SBVPI, and obtained Matthew's Correlation Coefficient values greater than 0.71 for all the datasets. In addition, the proposed framework was utilized to determine the association of diabetes with retinal and conjunctival vascular tortuosity. We observed that retinal vascular tortuosity (Eccentricity based Tortuosity Index) of the diabetic group was significantly higher (p < .05) than that of the non-diabetic group and that conjunctival vascular tortuosity (Total Curvature normalized by Arc Length) of diabetic group was significantly lower (p < .05) than that of the non-diabetic group. These observations were in agreement with the literature, strengthening the suitability of the proposed framework.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge