Shi Sub Byon
MESAHA-Net: Multi-Encoders based Self-Adaptive Hard Attention Network with Maximum Intensity Projections for Lung Nodule Segmentation in CT Scan
Apr 04, 2023Abstract:Accurate lung nodule segmentation is crucial for early-stage lung cancer diagnosis, as it can substantially enhance patient survival rates. Computed tomography (CT) images are widely employed for early diagnosis in lung nodule analysis. However, the heterogeneity of lung nodules, size diversity, and the complexity of the surrounding environment pose challenges for developing robust nodule segmentation methods. In this study, we propose an efficient end-to-end framework, the multi-encoder-based self-adaptive hard attention network (MESAHA-Net), for precise lung nodule segmentation in CT scans. MESAHA-Net comprises three encoding paths, an attention block, and a decoder block, facilitating the integration of three types of inputs: CT slice patches, forward and backward maximum intensity projection (MIP) images, and region of interest (ROI) masks encompassing the nodule. By employing a novel adaptive hard attention mechanism, MESAHA-Net iteratively performs slice-by-slice 2D segmentation of lung nodules, focusing on the nodule region in each slice to generate 3D volumetric segmentation of lung nodules. The proposed framework has been comprehensively evaluated on the LIDC-IDRI dataset, the largest publicly available dataset for lung nodule segmentation. The results demonstrate that our approach is highly robust for various lung nodule types, outperforming previous state-of-the-art techniques in terms of segmentation accuracy and computational complexity, rendering it suitable for real-time clinical implementation.
MEDS-Net: Self-Distilled Multi-Encoders Network with Bi-Direction Maximum Intensity projections for Lung Nodule Detection
Oct 30, 2022Abstract:In this study, we propose a lung nodule detection scheme which fully incorporates the clinic workflow of radiologists. Particularly, we exploit Bi-Directional Maximum intensity projection (MIP) images of various thicknesses (i.e., 3, 5 and 10mm) along with a 3D patch of CT scan, consisting of 10 adjacent slices to feed into self-distillation-based Multi-Encoders Network (MEDS-Net). The proposed architecture first condenses 3D patch input to three channels by using a dense block which consists of dense units which effectively examine the nodule presence from 2D axial slices. This condensed information, along with the forward and backward MIP images, is fed to three different encoders to learn the most meaningful representation, which is forwarded into the decoded block at various levels. At the decoder block, we employ a self-distillation mechanism by connecting the distillation block, which contains five lung nodule detectors. It helps to expedite the convergence and improves the learning ability of the proposed architecture. Finally, the proposed scheme reduces the false positives by complementing the main detector with auxiliary detectors. The proposed scheme has been rigorously evaluated on 888 scans of LUNA16 dataset and obtained a CPM score of 93.6\%. The results demonstrate that incorporating of bi-direction MIP images enables MEDS-Net to effectively distinguish nodules from surroundings which help to achieve the sensitivity of 91.5% and 92.8% with false positives rate of 0.25 and 0.5 per scan, respectively.
Dual-Stage Deeply Supervised Attention-based Convolutional Neural Networks for Mandibular Canal Segmentation in CBCT Scans
Oct 06, 2022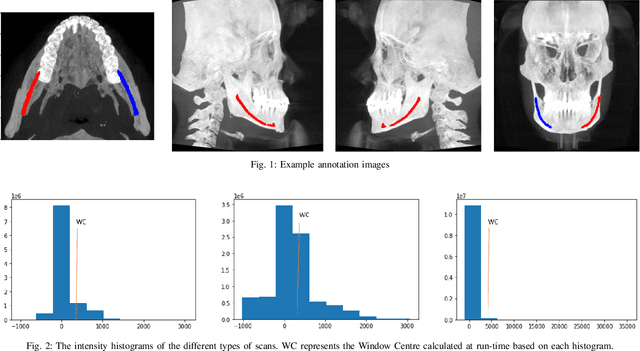
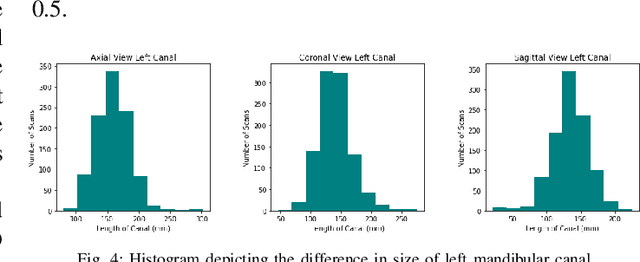
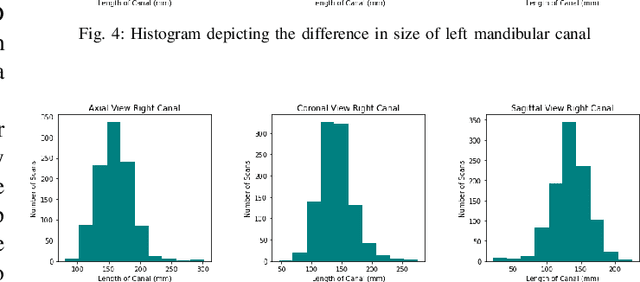
Abstract:Accurate segmentation of mandibular canals in lower jaws is important in dental implantology, in which the implant position and dimensions are currently determined manually from 3D CT images by medical experts to avoid damaging the mandibular nerve inside the canal. In this paper, we propose a novel dual-stage deep learning based scheme for automatic detection of mandibular canal. Particularly, we first we enhance the CBCT scans by employing the novel histogram-based dynamic windowing scheme which improves the visibility of mandibular canals. After enhancement, we design 3D deeply supervised attention U-Net architecture for localize the volume of interest (VOI) which contains the mandibular canals (i.e., left and right canals). Finally, we employed the multi-scale input residual U-Net architecture (MS-R-UNet) to accurately segment the mandibular canals. The proposed method has been rigorously evaluated on 500 scans and results demonstrate that our technique out performs the existing state-of-the-art methods in term of segmentation performance as well as robustness.
Volumetric Lung Nodule Segmentation using Adaptive ROI with Multi-View Residual Learning
Feb 03, 2020
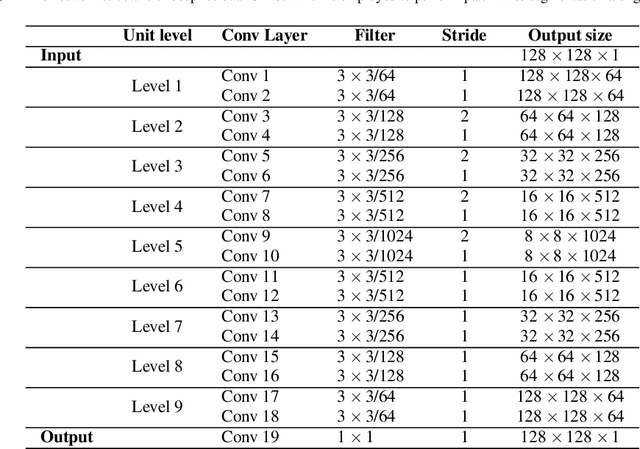


Abstract:Accurate quantification of pulmonary nodules can greatly assist the early diagnosis of lung cancer, which can enhance patient survival possibilities. A number of nodule segmentation techniques have been proposed, however, all of the existing techniques rely on radiologist 3-D volume of interest (VOI) input or use the constant region of interest (ROI) and only investigate the presence of nodule voxels within the given VOI. Such approaches restrain the solutions to investigate the nodule presence outside the given VOI and also include the redundant structures into VOI, which may lead to inaccurate nodule segmentation. In this work, a novel semi-automated approach for 3-D segmentation of nodule in volumetric computerized tomography (CT) lung scans has been proposed. The proposed technique can be segregated into two stages, at the first stage, it takes a 2-D ROI containing the nodule as input and it performs patch-wise investigation along the axial axis with a novel adaptive ROI strategy. The adaptive ROI algorithm enables the solution to dynamically select the ROI for the surrounding slices to investigate the presence of nodule using deep residual U-Net architecture. The first stage provides the initial estimation of nodule which is further utilized to extract the VOI. At the second stage, the extracted VOI is further investigated along the coronal and sagittal axis with two different networks and finally, all the estimated masks are fed into the consensus module to produce the final volumetric segmentation of nodule. The proposed approach has been rigorously evaluated on the LIDC dataset, which is the largest publicly available dataset. The result suggests that the approach is significantly robust and accurate as compared to the previous state of the art techniques.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge