Scott Howard
Zero-Shot Denoising for Fluorescence Lifetime Imaging Microscopy with Intensity-Guided Learning
Mar 17, 2025



Abstract:Multimodal and multi-information microscopy techniques such as Fluorescence Lifetime Imaging Microscopy (FLIM) extend the informational channels beyond intensity-based fluorescence microscopy but suffer from reduced image quality due to complex noise patterns. For FLIM, the intrinsic relationship between intensity and lifetime information means noise in each channel is a multivariate function across channels without necessarily sharing structural features. Based on this, we present a novel Zero-Shot Denoising Framework with an Intensity-Guided Learning approach. Our correlation-preserving strategy maintains important biological information that might be lost when channels are processed independently. Our framework implements separate processing paths for each channel and utilizes a pre-trained intensity denoising prior to guide the refinement of lifetime components across multiple channels. Through experiments on real-world FLIM-acquired biological samples, we show that our approach outperforms existing methods in both noise reduction and lifetime preservation, thereby enabling more reliable extraction of physiological and molecular information.
Deconvolution in Fluorescence Lifetime imaging microscopy (FLIM)
Jan 16, 2022


Abstract:Fluorescence lifetime imaging microscopy (FLIM) is an important technique to understand the chemical micro-environment in cells and tissues since it provides additional contrast compared to conventional fluorescence imaging. When two fluorophores within a diffraction limit are excited, the resulting emission leads to non-linear spatial distortion and localization effects in intensity (magnitude) and lifetime (phase) components. To address this issue, in this work, we provide a theoretical model for convolution in FLIM to describe how the resulting behavior differs from conventional fluorescence microscopy. We then present a Richardson-Lucy (RL) based deconvolution including total variation (TV) regularization method to correct for the distortions in FLIM measurements due to optical convolution, and experimentally demonstrate this FLIM deconvolution method on a multi-photon microscopy (MPM)-FLIM images of fluorescent-labeled fixed bovine pulmonary arterial endothelial (BPAE) cells.
Low dosage 3D volume fluorescence microscopy imaging using compressive sensing
Jan 03, 2022
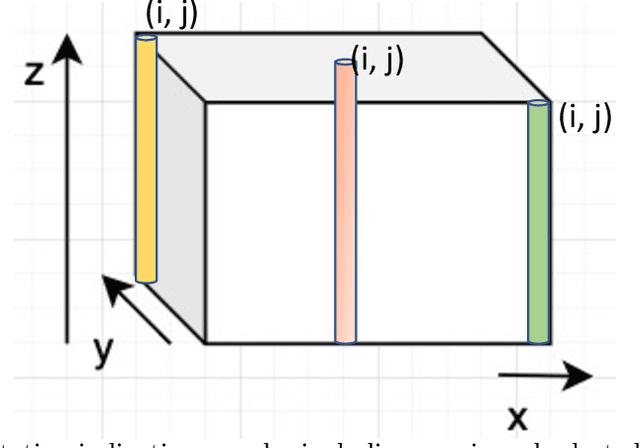
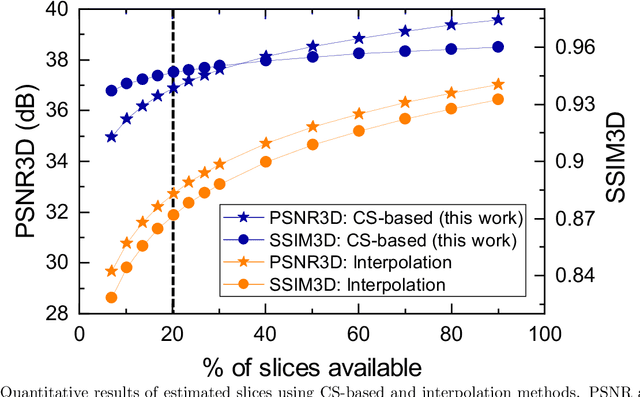

Abstract:Fluorescence microscopy has been a significant tool to observe long-term imaging of embryos (in vivo) growth over time. However, cumulative exposure is phototoxic to such sensitive live samples. While techniques like light-sheet fluorescence microscopy (LSFM) allow for reduced exposure, it is not well suited for deep imaging models. Other computational techniques are computationally expensive and often lack restoration quality. To address this challenge, one can use various low-dosage imaging techniques that are developed to achieve the 3D volume reconstruction using a few slices in the axial direction (z-axis); however, they often lack restoration quality. Also, acquiring dense images (with small steps) in the axial direction is computationally expensive. To address this challenge, we present a compressive sensing (CS) based approach to fully reconstruct 3D volumes with the same signal-to-noise ratio (SNR) with less than half of the excitation dosage. We present the theory and experimentally validate the approach. To demonstrate our technique, we capture a 3D volume of the RFP labeled neurons in the zebrafish embryo spinal cord (30um thickness) with the axial sampling of 0.1um using a confocal microscope. From the results, we observe the CS-based approach achieves accurate 3D volume reconstruction from less than 20% of the entire stack optical sections. The developed CS-based methodology in this work can be easily applied to other deep imaging modalities such as two-photon and light-sheet microscopy, where reducing sample photo-toxicity is a critical challenge.
Convolutional Neural Network Denoising in Fluorescence Lifetime Imaging Microscopy (FLIM)
Mar 07, 2021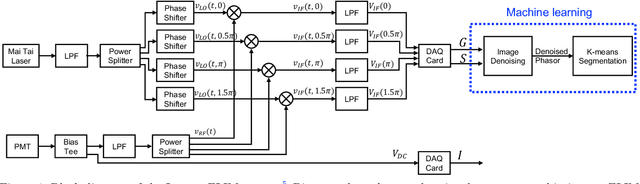



Abstract:Fluorescence lifetime imaging microscopy (FLIM) systems are limited by their slow processing speed, low signal-to-noise ratio (SNR), and expensive and challenging hardware setups. In this work, we demonstrate applying a denoising convolutional network to improve FLIM SNR. The network will be integrated with an instant FLIM system with fast data acquisition based on analog signal processing, high SNR using high-efficiency pulse-modulation, and cost-effective implementation utilizing off-the-shelf radio-frequency components. Our instant FLIM system simultaneously provides the intensity, lifetime, and phasor plots \textit{in vivo} and \textit{ex vivo}. By integrating image denoising using the trained deep learning model on the FLIM data, provide accurate FLIM phasor measurements are obtained. The enhanced phasor is then passed through the K-means clustering segmentation method, an unbiased and unsupervised machine learning technique to separate different fluorophores accurately. Our experimental \textit{in vivo} mouse kidney results indicate that introducing the deep learning image denoising model before the segmentation effectively removes the noise in the phasor compared to existing methods and provides clearer segments. Hence, the proposed deep learning-based workflow provides fast and accurate automatic segmentation of fluorescence images using instant FLIM. The denoising operation is effective for the segmentation if the FLIM measurements are noisy. The clustering can effectively enhance the detection of biological structures of interest in biomedical imaging applications.
Deep learning-based super-resolution fluorescence microscopy on small datasets
Mar 07, 2021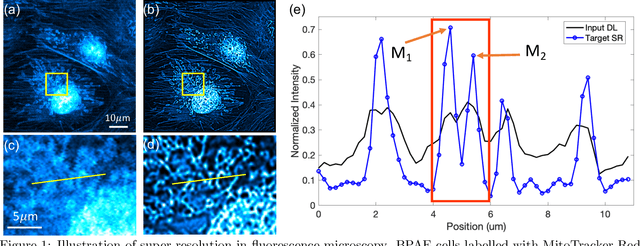


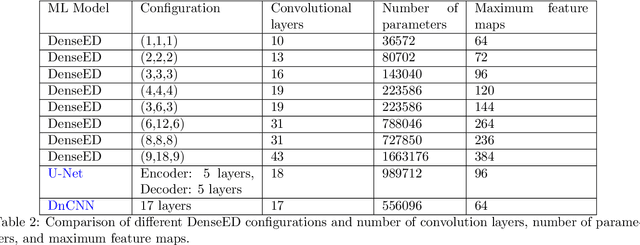
Abstract:Fluorescence microscopy has enabled a dramatic development in modern biology by visualizing biological organisms with micrometer scale resolution. However, due to the diffraction limit, sub-micron/nanometer features are difficult to resolve. While various super-resolution techniques are developed to achieve nanometer-scale resolution, they often either require expensive optical setup or specialized fluorophores. In recent years, deep learning has shown the potentials to reduce the technical barrier and obtain super-resolution from diffraction-limited images. For accurate results, conventional deep learning techniques require thousands of images as a training dataset. Obtaining large datasets from biological samples is not often feasible due to the photobleaching of fluorophores, phototoxicity, and dynamic processes occurring within the organism. Therefore, achieving deep learning-based super-resolution using small datasets is challenging. We address this limitation with a new convolutional neural network-based approach that is successfully trained with small datasets and achieves super-resolution images. We captured 750 images in total from 15 different field-of-views as the training dataset to demonstrate the technique. In each FOV, a single target image is generated using the super-resolution radial fluctuation method. As expected, this small dataset failed to produce a usable model using traditional super-resolution architecture. However, using the new approach, a network can be trained to achieve super-resolution images from this small dataset. This deep learning model can be applied to other biomedical imaging modalities such as MRI and X-ray imaging, where obtaining large training datasets is challenging.
A Poisson-Gaussian Denoising Dataset with Real Fluorescence Microscopy Images
Dec 26, 2018



Abstract:Fluorescence microscopy has enabled a dramatic development in modern biology. Due to its inherently weak signal, fluorescence microscopy is not only much noisier than photography, but also presented with Poisson-Gaussian noise where Poisson noise, or shot noise, is the dominating noise source, instead of Gaussian noise that dominates in photography. To get clean fluorescence microscopy images, it is highly desirable to have effective denoising algorithms and datasets that are specifically designed to denoise fluorescence microscopy images. While such algorithms exist, there are no such datasets available. In this paper, we fill this gap by constructing a dataset - the Fluorescence Microscopy Denoising (FMD) dataset - that is dedicated to Poisson-Gaussian denoising. The dataset consists 12,000 real fluorescence microscopy images obtained with commercial confocal, two-photon, and wide-field microscopes and representative biological samples such as cells, zebrafish, and mouse brain tissues. We use imaging averaging to effectively obtain ground truth images and 60,000 noisy images with different noise levels. We use this dataset to benchmark 10 representative denoising algorithms and find that deep learning methods have the best performance. To our knowledge, this is the first microscopy image dataset for Poisson-Gaussian denoising purposes and it could be an important tool for high-quality, real-time denoising applications in biomedical research.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge