Rod Jackson
A Transformer-based survival model for prediction of all-cause mortality in heart failure patients: a multi-cohort study
Mar 16, 2025



Abstract:We developed and validated TRisk, a Transformer-based AI model predicting 36-month mortality in heart failure patients by analysing temporal patient journeys from UK electronic health records (EHR). Our study included 403,534 heart failure patients (ages 40-90) from 1,418 English general practices, with 1,063 practices for model derivation and 355 for external validation. TRisk was compared against the MAGGIC-EHR model across various patient subgroups. With median follow-up of 9 months, TRisk achieved a concordance index of 0.845 (95% confidence interval: [0.841, 0.849]), significantly outperforming MAGGIC-EHR's 0.728 (0.723, 0.733) for predicting 36-month all-cause mortality. TRisk showed more consistent performance across sex, age, and baseline characteristics, suggesting less bias. We successfully adapted TRisk to US hospital data through transfer learning, achieving a C-index of 0.802 (0.789, 0.816) with 21,767 patients. Explainability analyses revealed TRisk captured established risk factors while identifying underappreciated predictors like cancers and hepatic failure that were important across both cohorts. Notably, cancers maintained strong prognostic value even a decade after diagnosis. TRisk demonstrated well-calibrated mortality prediction across both healthcare systems. Our findings highlight the value of tracking longitudinal health profiles and revealed risk factors not included in previous expert-driven models.
Predicting cardiovascular risk from national administrative databases using a combined survival analysis and deep learning approach
Nov 28, 2020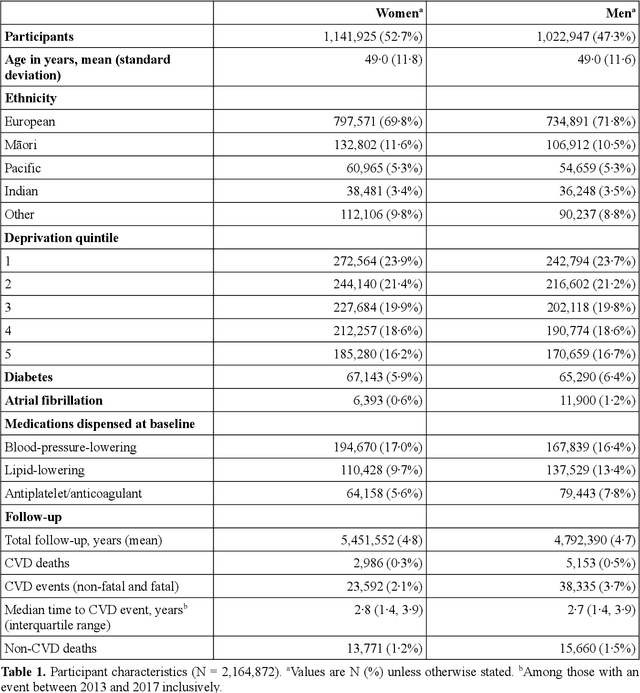
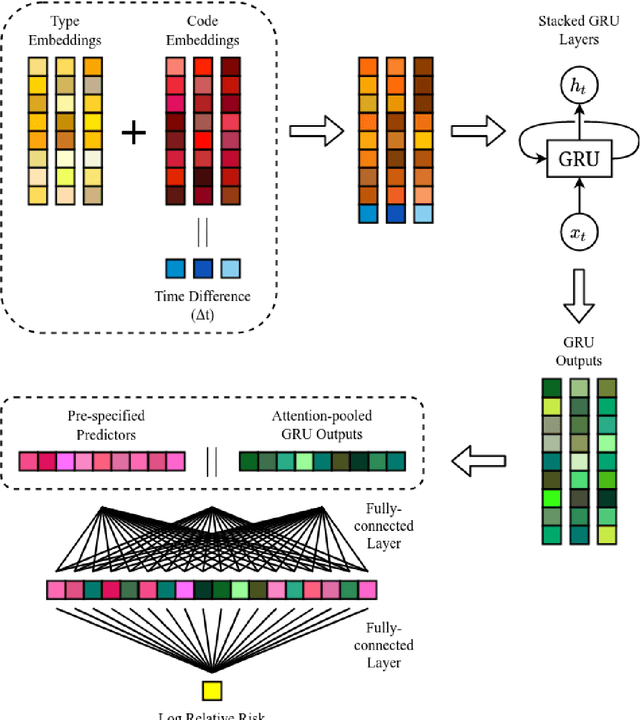
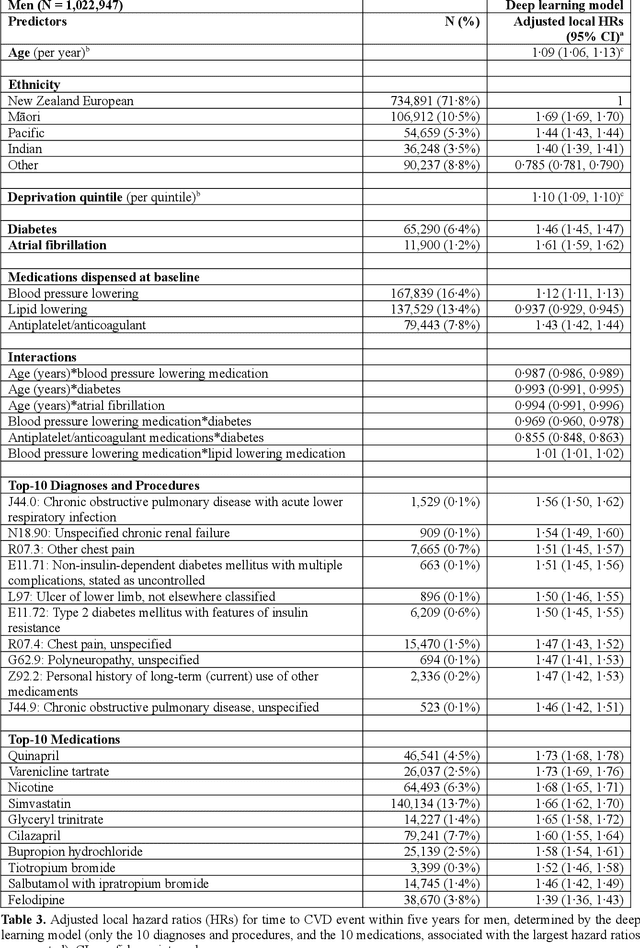
Abstract:AIMS. This study compared the performance of deep learning extensions of survival analysis models with traditional Cox proportional hazards (CPH) models for deriving cardiovascular disease (CVD) risk prediction equations in national health administrative datasets. METHODS. Using individual person linkage of multiple administrative datasets, we constructed a cohort of all New Zealand residents aged 30-74 years who interacted with publicly funded health services during 2012, and identified hospitalisations and deaths from CVD over five years of follow-up. After excluding people with prior CVD or heart failure, sex-specific deep learning and CPH models were developed to estimate the risk of fatal or non-fatal CVD events within five years. The proportion of explained time-to-event occurrence, calibration, and discrimination were compared between models across the whole study population and in specific risk groups. FINDINGS. First CVD events occurred in 61,927 of 2,164,872 people. Among diagnoses and procedures, the largest 'local' hazard ratios were associated by the deep learning models with tobacco use in women (2.04, 95%CI: 1.99-2.10) and with chronic obstructive pulmonary disease with acute lower respiratory infection in men (1.56, 95%CI: 1.50-1.62). Other identified predictors (e.g. hypertension, chest pain, diabetes) aligned with current knowledge about CVD risk predictors. The deep learning models significantly outperformed the CPH models on the basis of proportion of explained time-to-event occurrence (Royston and Sauerbrei's R-squared: 0.468 vs. 0.425 in women and 0.383 vs. 0.348 in men), calibration, and discrimination (all p<0.0001). INTERPRETATION. Deep learning extensions of survival analysis models can be applied to large health administrative databases to derive interpretable CVD risk prediction equations that are more accurate than traditional CPH models.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge