Kazuki Ishikawa
Bayesian Optimization for Simultaneous Selection of Machine Learning Algorithms and Hyperparameters on Shared Latent Space
Feb 13, 2025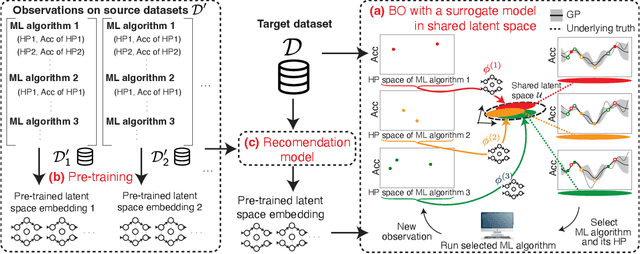
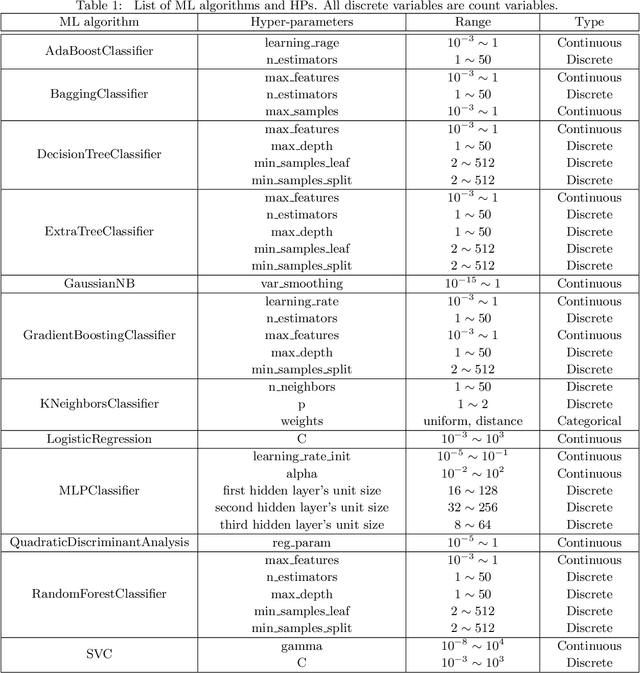
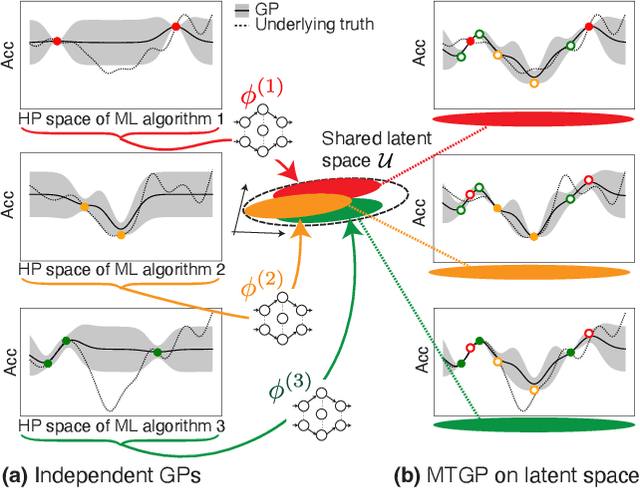
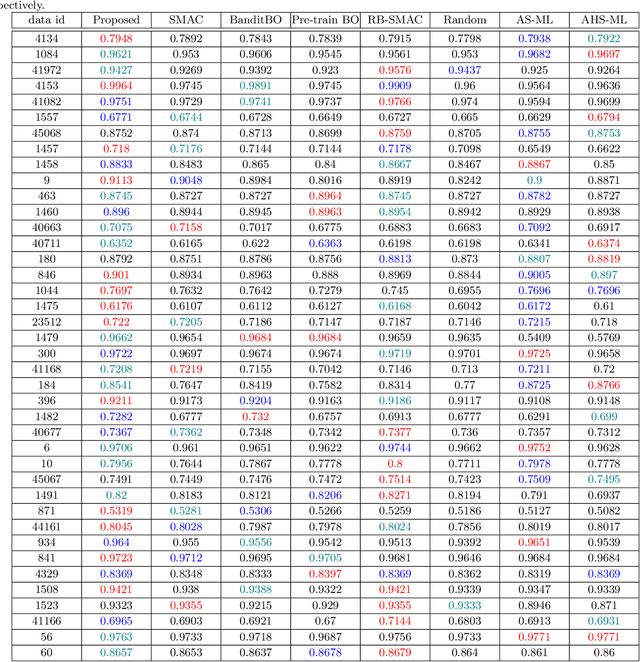
Abstract:Selecting the optimal combination of a machine learning (ML) algorithm and its hyper-parameters is crucial for the development of high-performance ML systems. However, since the combination of ML algorithms and hyper-parameters is enormous, the exhaustive validation requires a significant amount of time. Many existing studies use Bayesian optimization (BO) for accelerating the search. On the other hand, a significant difficulty is that, in general, there exists a different hyper-parameter space for each one of candidate ML algorithms. BO-based approaches typically build a surrogate model independently for each hyper-parameter space, by which sufficient observations are required for all candidate ML algorithms. In this study, our proposed method embeds different hyper-parameter spaces into a shared latent space, in which a surrogate multi-task model for BO is estimated. This approach can share information of observations from different ML algorithms by which efficient optimization is expected with a smaller number of total observations. We further propose the pre-training of the latent space embedding with an adversarial regularization, and a ranking model for selecting an effective pre-trained embedding for a given target dataset. Our empirical study demonstrates effectiveness of the proposed method through datasets from OpenML.
Multi-Objective Bayesian Optimization with Active Preference Learning
Nov 22, 2023Abstract:There are a lot of real-world black-box optimization problems that need to optimize multiple criteria simultaneously. However, in a multi-objective optimization (MOO) problem, identifying the whole Pareto front requires the prohibitive search cost, while in many practical scenarios, the decision maker (DM) only needs a specific solution among the set of the Pareto optimal solutions. We propose a Bayesian optimization (BO) approach to identifying the most preferred solution in the MOO with expensive objective functions, in which a Bayesian preference model of the DM is adaptively estimated by an interactive manner based on the two types of supervisions called the pairwise preference and improvement request. To explore the most preferred solution, we define an acquisition function in which the uncertainty both in the objective functions and the DM preference is incorporated. Further, to minimize the interaction cost with the DM, we also propose an active learning strategy for the preference estimation. We empirically demonstrate the effectiveness of our proposed method through the benchmark function optimization and the hyper-parameter optimization problems for machine learning models.
Development of pericardial fat count images using a combination of three different deep-learning models
Jul 25, 2023



Abstract:Rationale and Objectives: Pericardial fat (PF), the thoracic visceral fat surrounding the heart, promotes the development of coronary artery disease by inducing inflammation of the coronary arteries. For evaluating PF, this study aimed to generate pericardial fat count images (PFCIs) from chest radiographs (CXRs) using a dedicated deep-learning model. Materials and Methods: The data of 269 consecutive patients who underwent coronary computed tomography (CT) were reviewed. Patients with metal implants, pleural effusion, history of thoracic surgery, or that of malignancy were excluded. Thus, the data of 191 patients were used. PFCIs were generated from the projection of three-dimensional CT images, where fat accumulation was represented by a high pixel value. Three different deep-learning models, including CycleGAN, were combined in the proposed method to generate PFCIs from CXRs. A single CycleGAN-based model was used to generate PFCIs from CXRs for comparison with the proposed method. To evaluate the image quality of the generated PFCIs, structural similarity index measure (SSIM), mean squared error (MSE), and mean absolute error (MAE) of (i) the PFCI generated using the proposed method and (ii) the PFCI generated using the single model were compared. Results: The mean SSIM, MSE, and MAE were as follows: 0.856, 0.0128, and 0.0357, respectively, for the proposed model; and 0.762, 0.0198, and 0.0504, respectively, for the single CycleGAN-based model. Conclusion: PFCIs generated from CXRs with the proposed model showed better performance than those with the single model. PFCI evaluation without CT may be possible with the proposed method.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge