Jens Vogel-Claussen
Requirements for Quality Assurance of AI Models for Early Detection of Lung Cancer
Feb 24, 2025Abstract:Lung cancer is the second most common cancer and the leading cause of cancer-related deaths worldwide. Survival largely depends on tumor stage at diagnosis, and early detection with low-dose CT can significantly reduce mortality in high-risk patients. AI can improve the detection, measurement, and characterization of pulmonary nodules while reducing assessment time. However, the training data, functionality, and performance of available AI systems vary considerably, complicating software selection and regulatory evaluation. Manufacturers must specify intended use and provide test statistics, but they can choose their training and test data, limiting standardization and comparability. Under the EU AI Act, consistent quality assurance is required for AI-based nodule detection, measurement, and characterization. This position paper proposes systematic quality assurance grounded in a validated reference dataset, including real screening cases plus phantom data to verify volume and growth rate measurements. Regular updates shall reflect demographic shifts and technological advances, ensuring ongoing relevance. Consequently, ongoing AI quality assurance is vital. Regulatory challenges are also adressed. While the MDR and the EU AI Act set baseline requirements, they do not adequately address self-learning algorithms or their updates. A standardized, transparent quality assessment - based on sensitivity, specificity, and volumetric accuracy - enables an objective evaluation of each AI solution's strengths and weaknesses. Establishing clear testing criteria and systematically using updated reference data lay the groundwork for comparable performance metrics, informing tenders, guidelines, and recommendations.
$ν$-net: Deep Learning for Generalized Biventricular Cardiac Mass and Function Parameters
Jun 14, 2017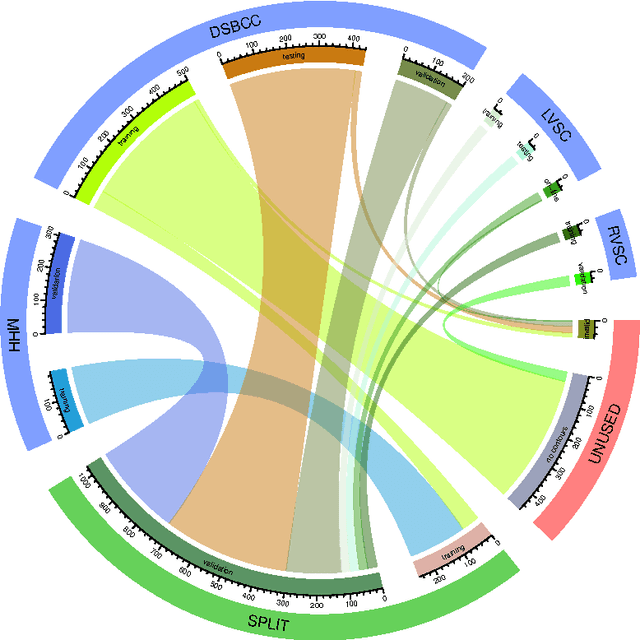

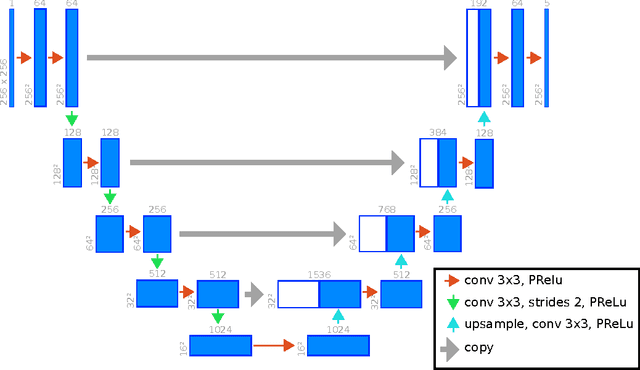
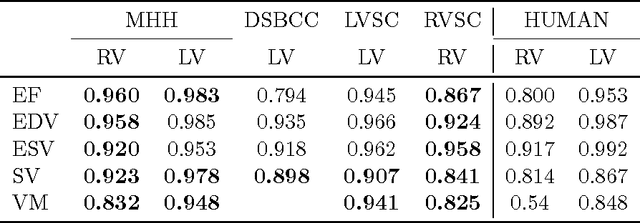
Abstract:Background: Cardiac MRI derived biventricular mass and function parameters, such as end-systolic volume (ESV), end-diastolic volume (EDV), ejection fraction (EF), stroke volume (SV), and ventricular mass (VM) are clinically well established. Image segmentation can be challenging and time-consuming, due to the complex anatomy of the human heart. Objectives: This study introduces $\nu$-net (/nju:n$\varepsilon$t/) -- a deep learning approach allowing for fully-automated high quality segmentation of right (RV) and left ventricular (LV) endocardium and epicardium for extraction of cardiac function parameters. Methods: A set consisting of 253 manually segmented cases has been used to train a deep neural network. Subsequently, the network has been evaluated on 4 different multicenter data sets with a total of over 1000 cases. Results: For LV EF the intraclass correlation coefficient (ICC) is 98, 95, and 80 % (95 %), and for RV EF 96, and 87 % (80 %) on the respective data sets (human expert ICCs reported in parenthesis). The LV VM ICC is 95, and 94 % (84 %), and the RV VM ICC is 83, and 83 % (54 %). This study proposes a simple adjustment procedure, allowing for the adaptation to distinct segmentation philosophies. $\nu$-net exhibits state of-the-art performance in terms of dice coefficient. Conclusions: Biventricular mass and function parameters can be determined reliably in high quality by applying a deep neural network for cardiac MRI segmentation, especially in the anatomically complex right ventricle. Adaption to individual segmentation styles by applying a simple adjustment procedure is viable, allowing for the processing of novel data without time-consuming additional training.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge