Lemuel Puglisi
for the Alzheimer's Disease Neuroimaging Initiative
GenTract: Generative Global Tractography
Nov 17, 2025Abstract:Tractography is the process of inferring the trajectories of white-matter pathways in the brain from diffusion magnetic resonance imaging (dMRI). Local tractography methods, which construct streamlines by following local fiber orientation estimates stepwise through an image, are prone to error accumulation and high false positive rates, particularly on noisy or low-resolution data. In contrast, global methods, which attempt to optimize a collection of streamlines to maximize compatibility with underlying fiber orientation estimates, are computationally expensive. To address these challenges, we introduce GenTract, the first generative model for global tractography. We frame tractography as a generative task, learning a direct mapping from dMRI to complete, anatomically plausible streamlines. We compare both diffusion-based and flow matching paradigms and evaluate GenTract's performance against state-of-the-art baselines. Notably, GenTract achieves precision 2.1x higher than the next-best method, TractOracle. This advantage becomes even more pronounced in challenging low-resolution and noisy settings, where it outperforms the closest competitor by an order of magnitude. By producing tractograms with high precision on research-grade data while also maintaining reliability on imperfect, lower-resolution data, GenTract represents a promising solution for global tractography.
Brain Latent Progression: Individual-based Spatiotemporal Disease Progression on 3D Brain MRIs via Latent Diffusion
Feb 12, 2025Abstract:The growing availability of longitudinal Magnetic Resonance Imaging (MRI) datasets has facilitated Artificial Intelligence (AI)-driven modeling of disease progression, making it possible to predict future medical scans for individual patients. However, despite significant advancements in AI, current methods continue to face challenges including achieving patient-specific individualization, ensuring spatiotemporal consistency, efficiently utilizing longitudinal data, and managing the substantial memory demands of 3D scans. To address these challenges, we propose Brain Latent Progression (BrLP), a novel spatiotemporal model designed to predict individual-level disease progression in 3D brain MRIs. The key contributions in BrLP are fourfold: (i) it operates in a small latent space, mitigating the computational challenges posed by high-dimensional imaging data; (ii) it explicitly integrates subject metadata to enhance the individualization of predictions; (iii) it incorporates prior knowledge of disease dynamics through an auxiliary model, facilitating the integration of longitudinal data; and (iv) it introduces the Latent Average Stabilization (LAS) algorithm, which (a) enforces spatiotemporal consistency in the predicted progression at inference time and (b) allows us to derive a measure of the uncertainty for the prediction. We train and evaluate BrLP on 11,730 T1-weighted (T1w) brain MRIs from 2,805 subjects and validate its generalizability on an external test set comprising 2,257 MRIs from 962 subjects. Our experiments compare BrLP-generated MRI scans with real follow-up MRIs, demonstrating state-of-the-art accuracy compared to existing methods. The code is publicly available at: https://github.com/LemuelPuglisi/BrLP.
SynthBA: Reliable Brain Age Estimation Across Multiple MRI Sequences and Resolutions
Jun 01, 2024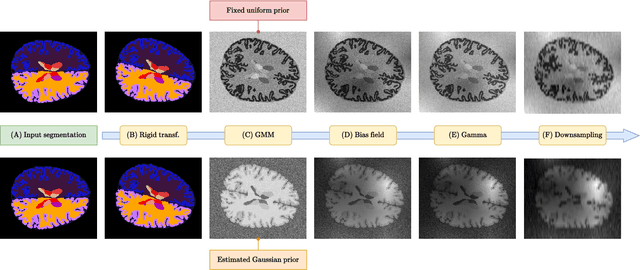
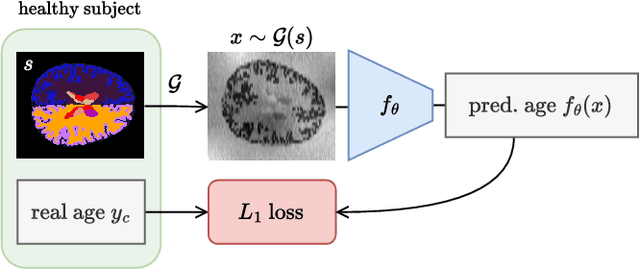

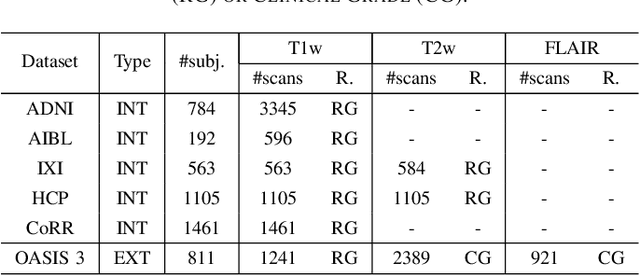
Abstract:Brain age is a critical measure that reflects the biological ageing process of the brain. The gap between brain age and chronological age, referred to as brain PAD (Predicted Age Difference), has been utilized to investigate neurodegenerative conditions. Brain age can be predicted using MRIs and machine learning techniques. However, existing methods are often sensitive to acquisition-related variabilities, such as differences in acquisition protocols, scanners, MRI sequences, and resolutions, significantly limiting their application in highly heterogeneous clinical settings. In this study, we introduce Synthetic Brain Age (SynthBA), a robust deep-learning model designed for predicting brain age. SynthBA utilizes an advanced domain randomization technique, ensuring effective operation across a wide array of acquisition-related variabilities. To assess the effectiveness and robustness of SynthBA, we evaluate its predictive capabilities on internal and external datasets, encompassing various MRI sequences and resolutions, and compare it with state-of-the-art techniques. Additionally, we calculate the brain PAD in a large cohort of subjects with Alzheimer's Disease (AD), demonstrating a significant correlation with AD-related measures of cognitive dysfunction. SynthBA holds the potential to facilitate the broader adoption of brain age prediction in clinical settings, where re-training or fine-tuning is often unfeasible. The SynthBA source code and pre-trained models are publicly available at https://github.com/LemuelPuglisi/SynthBA.
Enhancing Spatiotemporal Disease Progression Models via Latent Diffusion and Prior Knowledge
May 06, 2024


Abstract:In this work, we introduce Brain Latent Progression (BrLP), a novel spatiotemporal disease progression model based on latent diffusion. BrLP is designed to predict the evolution of diseases at the individual level on 3D brain MRIs. Existing deep generative models developed for this task are primarily data-driven and face challenges in learning disease progressions. BrLP addresses these challenges by incorporating prior knowledge from disease models to enhance the accuracy of predictions. To implement this, we propose to integrate an auxiliary model that infers volumetric changes in various brain regions. Additionally, we introduce Latent Average Stabilization (LAS), a novel technique to improve spatiotemporal consistency of the predicted progression. BrLP is trained and evaluated on a large dataset comprising 11,730 T1-weighted brain MRIs from 2,805 subjects, collected from three publicly available, longitudinal Alzheimer's Disease (AD) studies. In our experiments, we compare the MRI scans generated by BrLP with the actual follow-up MRIs available from the subjects, in both cross-sectional and longitudinal settings. BrLP demonstrates significant improvements over existing methods, with an increase of 22% in volumetric accuracy across AD-related brain regions and 43% in image similarity to the ground-truth scans. The ability of BrLP to generate conditioned 3D scans at the subject level, along with the novelty of integrating prior knowledge to enhance accuracy, represents a significant advancement in disease progression modeling, opening new avenues for precision medicine. The code of BrLP is available at the following link: https://github.com/LemuelPuglisi/BrLP.
DeepBrainPrint: A Novel Contrastive Framework for Brain MRI Re-Identification
Feb 25, 2023



Abstract:Recent advances in MRI have led to the creation of large datasets. With the increase in data volume, it has become difficult to locate previous scans of the same patient within these datasets (a process known as re-identification). To address this issue, we propose an AI-powered medical imaging retrieval framework called DeepBrainPrint, which is designed to retrieve brain MRI scans of the same patient. Our framework is a semi-self-supervised contrastive deep learning approach with three main innovations. First, we use a combination of self-supervised and supervised paradigms to create an effective brain fingerprint from MRI scans that can be used for real-time image retrieval. Second, we use a special weighting function to guide the training and improve model convergence. Third, we introduce new imaging transformations to improve retrieval robustness in the presence of intensity variations (i.e. different scan contrasts), and to account for age and disease progression in patients. We tested DeepBrainPrint on a large dataset of T1-weighted brain MRIs from the Alzheimer's Disease Neuroimaging Initiative (ADNI) and on a synthetic dataset designed to evaluate retrieval performance with different image modalities. Our results show that DeepBrainPrint outperforms previous methods, including simple similarity metrics and more advanced contrastive deep learning frameworks.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge