Jon Andre Ottesen
for the Alzheimer's Disease Neuroimaging Initiative
Deep Learning-Based Prediction of PET Amyloid Status Using Multi-Contrast MRI
Nov 18, 2024



Abstract:Identifying amyloid-beta positive patients is crucial for determining eligibility for Alzheimer's disease (AD) clinical trials and new disease-modifying treatments, but currently requires PET or CSF sampling. Previous MRI-based deep learning models for predicting amyloid positivity, using only T1w sequences, have shown moderate performance. We trained deep learning models to predict amyloid PET positivity and evaluated whether multi-contrast inputs improve performance. A total of 4,058 exams with multi-contrast MRI and PET-based quantitative amyloid deposition were obtained from three public datasets: the Alzheimer's Disease Neuroimaging Initiative (ADNI), the Open Access Series of Imaging Studies 3 (OASIS3), and the Anti-Amyloid Treatment in Asymptomatic Alzheimer's Disease (A4). Two separate EfficientNet models were trained for amyloid positivity prediction: one with only T1w images and the other with both T1w and T2-FLAIR images as network inputs. The area under the curve (AUC), accuracy, sensitivity, and specificity were determined using an internal held-out test set. The trained models were further evaluated using an external test set. In the held-out test sets, the T1w and T1w+T2FLAIR models demonstrated AUCs of 0.62 (95% CI: 0.60, 0.64) and 0.67 (95% CI: 0.64, 0.70) (p = 0.006); accuracies were 61% (95% CI: 60%, 63%) and 64% (95% CI: 62%, 66%) (p = 0.008); sensitivities were 0.88 and 0.71; and specificities were 0.23 and 0.53, respectively. The trained models showed similar performance in the external test set. Performance of the current model on both test sets exceeded that of the publicly available model. In conclusion, the use of multi-contrast MRI, specifically incorporating T2-FLAIR in addition to T1w images, significantly improved the predictive accuracy of PET-determined amyloid status from MRI scans using a deep learning approach.
A Densely Interconnected Network for Deep Learning Accelerated MRI
Jul 05, 2022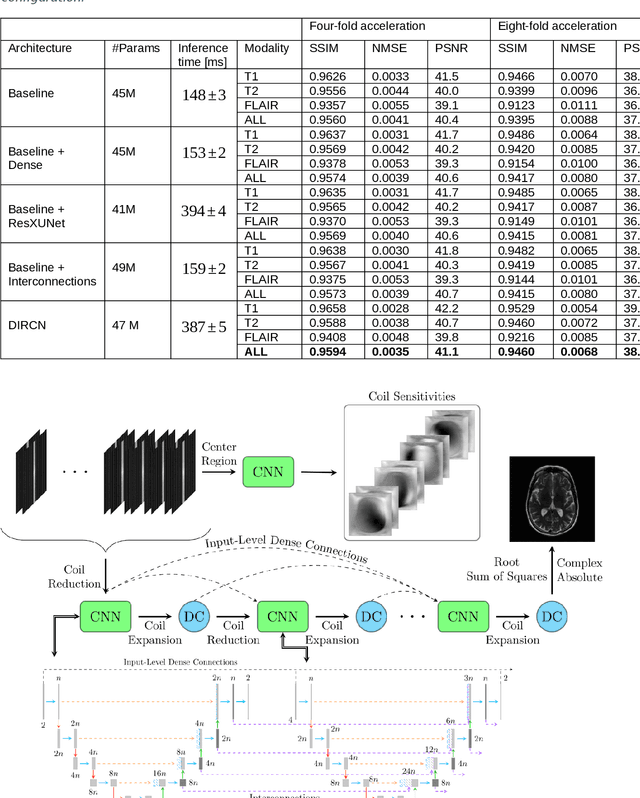
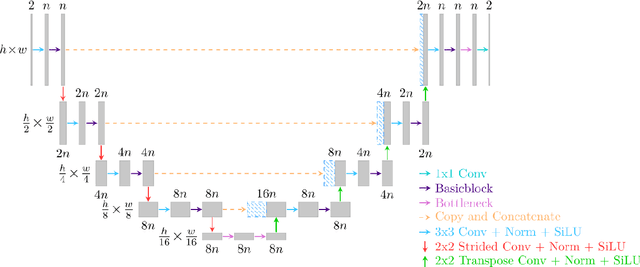
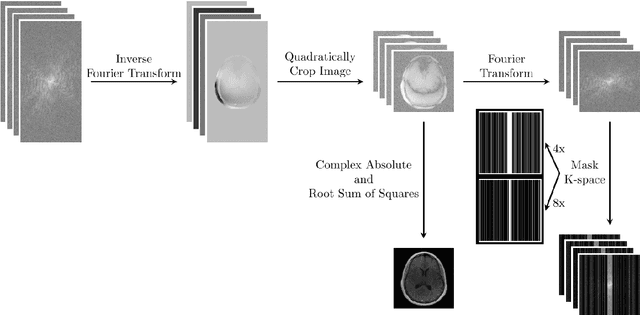
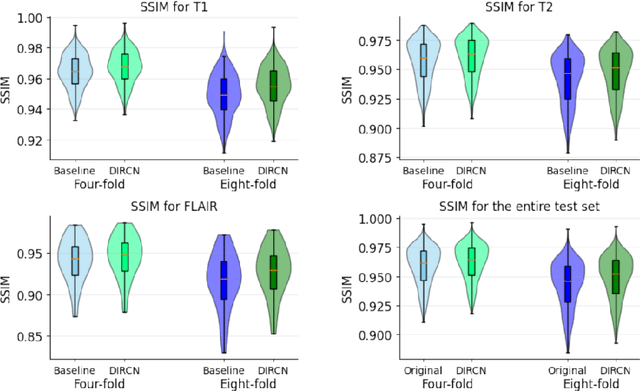
Abstract:Objective: To improve accelerated MRI reconstruction through a densely connected cascading deep learning reconstruction framework. Materials and Methods: A cascading deep learning reconstruction framework (baseline model) was modified by applying three architectural modifications: Input-level dense connections between cascade inputs and outputs, an improved deep learning sub-network, and long-range skip-connections between subsequent deep learning networks. An ablation study was performed, where five model configurations were trained on the NYU fastMRI neuro dataset with an end-to-end scheme conjunct on four- and eight-fold acceleration. The trained models were evaluated by comparing their respective structural similarity index measure (SSIM), normalized mean square error (NMSE) and peak signal to noise ratio (PSNR). Results: The proposed densely interconnected residual cascading network (DIRCN), utilizing all three suggested modifications, achieved a SSIM improvement of 8% and 11% for four- and eight-fold acceleration, respectively. For eight-fold acceleration, the model achieved a 23% decrease in the NMSE when compared to the baseline model. In an ablation study, the individual architectural modifications all contributed to this improvement, by reducing the SSIM and NMSE with approximately 3% and 5% for four-fold acceleration, respectively. Conclusion: The proposed architectural modifications allow for simple adjustments on an already existing cascading framework to further improve the resulting reconstructions.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge