Jeongchul Kim
MCR-VQGAN: A Scalable and Cost-Effective Tau PET Synthesis Approach for Alzheimer's Disease Imaging
Dec 17, 2025
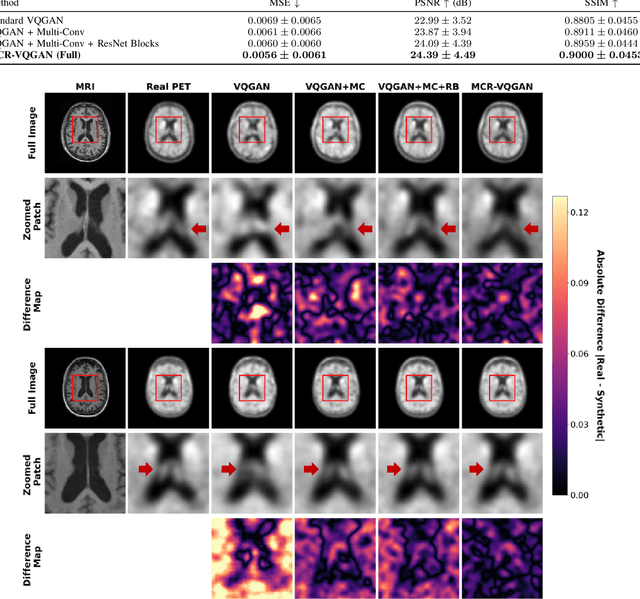


Abstract:Tau positron emission tomography (PET) is a critical diagnostic modality for Alzheimer's disease (AD) because it visualizes and quantifies neurofibrillary tangles, a hallmark of AD pathology. However, its widespread clinical adoption is hindered by significant challenges, such as radiation exposure, limited availability, high clinical workload, and substantial financial costs. To overcome these limitations, we propose Multi-scale CBAM Residual Vector Quantized Generative Adversarial Network (MCR-VQGAN) to synthesize high-fidelity tau PET images from structural T1-weighted MRI scans. MCR-VQGAN improves standard VQGAN by integrating three key architectural enhancements: multi-scale convolutions, ResNet blocks, and Convolutional Block Attention Modules (CBAM). Using 222 paired structural T1-weighted MRI and tau PET scans from Alzheimer's Disease Neuroimaging Initiative (ADNI), we trained and compared MCR-VQGAN with cGAN, WGAN-GP, CycleGAN, and VQGAN. Our proposed model achieved superior image synthesis performance across all metrics: MSE of 0.0056 +/- 0.0061, PSNR of 24.39 +/- 4.49 dB, and SSIM of 0.9000 +/- 0.0453. To assess the clinical utility of the synthetic images, we trained and evaluated a CNN-based AD classifier. The classifier achieved comparable accuracy when tested on real (63.64%) and synthetic (65.91%) images. This result indicates that our synthesis process successfully preserves diagnostically relevant features without significant information loss. Our results demonstrate that MCR-VQGAN can offer a reliable and scalable surrogate for conventional tau PET imaging, potentially improving the accessibility and scalability of tau imaging biomarkers for AD research and clinical workflows.
Nonhuman Primate Brain Tissue Segmentation Using a Transfer Learning Approach
Apr 01, 2025Abstract:Non-human primates (NHPs) serve as critical models for understanding human brain function and neurological disorders due to their close evolutionary relationship with humans. Accurate brain tissue segmentation in NHPs is critical for understanding neurological disorders, but challenging due to the scarcity of annotated NHP brain MRI datasets, the small size of the NHP brain, the limited resolution of available imaging data and the anatomical differences between human and NHP brains. To address these challenges, we propose a novel approach utilizing STU-Net with transfer learning to leverage knowledge transferred from human brain MRI data to enhance segmentation accuracy in the NHP brain MRI, particularly when training data is limited. The combination of STU-Net and transfer learning effectively delineates complex tissue boundaries and captures fine anatomical details specific to NHP brains. Notably, our method demonstrated improvement in segmenting small subcortical structures such as putamen and thalamus that are challenging to resolve with limited spatial resolution and tissue contrast, and achieved DSC of over 0.88, IoU over 0.8 and HD95 under 7. This study introduces a robust method for multi-class brain tissue segmentation in NHPs, potentially accelerating research in evolutionary neuroscience and preclinical studies of neurological disorders relevant to human health.
Synthesizing beta-amyloid PET images from T1-weighted Structural MRI: A Preliminary Study
Sep 26, 2024


Abstract:Beta-amyloid positron emission tomography (A$\beta$-PET) imaging has become a critical tool in Alzheimer's disease (AD) research and diagnosis, providing insights into the pathological accumulation of amyloid plaques, one of the hallmarks of AD. However, the high cost, limited availability, and exposure to radioactivity restrict the widespread use of A$\beta$-PET imaging, leading to a scarcity of comprehensive datasets. Previous studies have suggested that structural magnetic resonance imaging (MRI), which is more readily available, may serve as a viable alternative for synthesizing A$\beta$-PET images. In this study, we propose an approach to utilize 3D diffusion models to synthesize A$\beta$-PET images from T1-weighted MRI scans, aiming to overcome the limitations associated with direct PET imaging. Our method generates high-quality A$\beta$-PET images for cognitive normal cases, although it is less effective for mild cognitive impairment (MCI) patients due to the variability in A$\beta$ deposition patterns among subjects. Our preliminary results suggest that incorporating additional data, such as a larger sample of MCI cases and multi-modality information including clinical and demographic details, cognitive and functional assessments, and longitudinal data, may be necessary to improve A$\beta$-PET image synthesis for MCI patients.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge