Euan A. Ashley
High-Throughput Precision Phenotyping of Left Ventricular Hypertrophy with Cardiovascular Deep Learning
Jun 23, 2021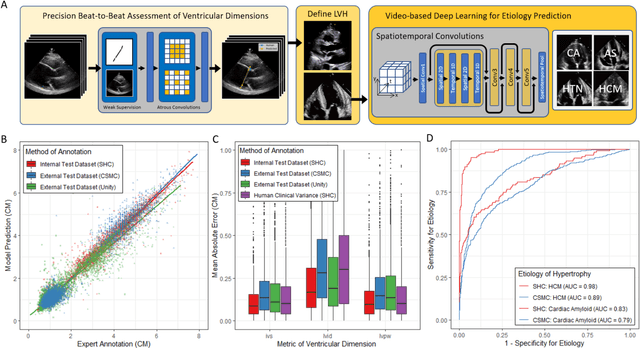
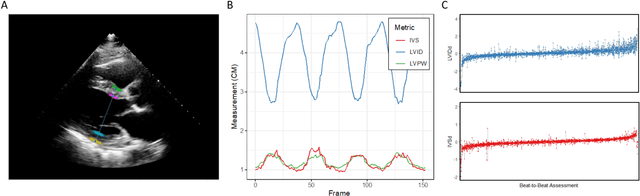
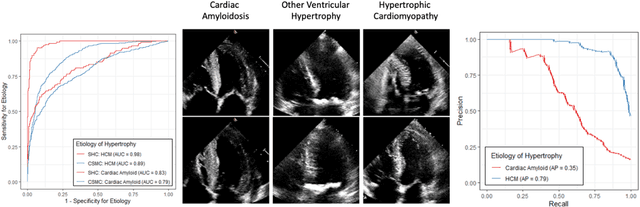
Abstract:Left ventricular hypertrophy (LVH) results from chronic remodeling caused by a broad range of systemic and cardiovascular disease including hypertension, aortic stenosis, hypertrophic cardiomyopathy, and cardiac amyloidosis. Early detection and characterization of LVH can significantly impact patient care but is limited by under-recognition of hypertrophy, measurement error and variability, and difficulty differentiating etiologies of LVH. To overcome this challenge, we present EchoNet-LVH - a deep learning workflow that automatically quantifies ventricular hypertrophy with precision equal to human experts and predicts etiology of LVH. Trained on 28,201 echocardiogram videos, our model accurately measures intraventricular wall thickness (mean absolute error [MAE] 1.4mm, 95% CI 1.2-1.5mm), left ventricular diameter (MAE 2.4mm, 95% CI 2.2-2.6mm), and posterior wall thickness (MAE 1.2mm, 95% CI 1.1-1.3mm) and classifies cardiac amyloidosis (area under the curve of 0.83) and hypertrophic cardiomyopathy (AUC 0.98) from other etiologies of LVH. In external datasets from independent domestic and international healthcare systems, EchoNet-LVH accurately quantified ventricular parameters (R2 of 0.96 and 0.90 respectively) and detected cardiac amyloidosis (AUC 0.79) and hypertrophic cardiomyopathy (AUC 0.89) on the domestic external validation site. Leveraging measurements across multiple heart beats, our model can more accurately identify subtle changes in LV geometry and its causal etiologies. Compared to human experts, EchoNet-LVH is fully automated, allowing for reproducible, precise measurements, and lays the foundation for precision diagnosis of cardiac hypertrophy. As a resource to promote further innovation, we also make publicly available a large dataset of 23,212 annotated echocardiogram videos.
Medical Imaging and Machine Learning
Mar 02, 2021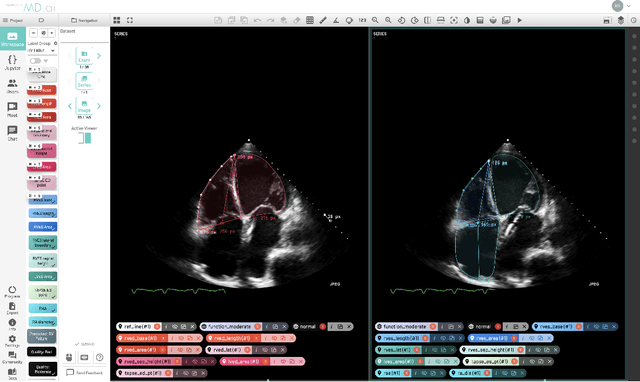

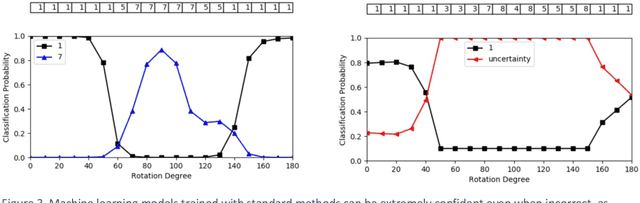
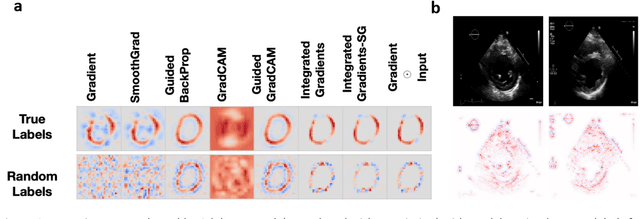
Abstract:Advances in computing power, deep learning architectures, and expert labelled datasets have spurred the development of medical imaging artificial intelligence systems that rival clinical experts in a variety of scenarios. The National Institutes of Health in 2018 identified key focus areas for the future of artificial intelligence in medical imaging, creating a foundational roadmap for research in image acquisition, algorithms, data standardization, and translatable clinical decision support systems. Among the key issues raised in the report: data availability, need for novel computing architectures and explainable AI algorithms, are still relevant despite the tremendous progress made over the past few years alone. Furthermore, translational goals of data sharing, validation of performance for regulatory approval, generalizability and mitigation of unintended bias must be accounted for early in the development process. In this perspective paper we explore challenges unique to high dimensional clinical imaging data, in addition to highlighting some of the technical and ethical considerations in developing high-dimensional, multi-modality, machine learning systems for clinical decision support.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge