Beatriz Costa-Gomes
PERC: a suite of software tools for the curation of cryoEM data with application to simulation, modelling and machine learning
Mar 17, 2025Abstract:Ease of access to data, tools and models expedites scientific research. In structural biology there are now numerous open repositories of experimental and simulated datasets. Being able to easily access and utilise these is crucial for allowing researchers to make optimal use of their research effort. The tools presented here are useful for collating existing public cryoEM datasets and/or creating new synthetic cryoEM datasets to aid the development of novel data processing and interpretation algorithms. In recent years, structural biology has seen the development of a multitude of machine-learning based algorithms for aiding numerous steps in the processing and reconstruction of experimental datasets and the use of these approaches has become widespread. Developing such techniques in structural biology requires access to large datasets which can be cumbersome to curate and unwieldy to make use of. In this paper we present a suite of Python software packages which we collectively refer to as PERC (profet, EMPIARreader and CAKED). These are designed to reduce the burden which data curation places upon structural biology research. The protein structure fetcher (profet) package allows users to conveniently download and cleave sequences or structures from the Protein Data Bank or Alphafold databases. EMPIARreader allows lazy loading of Electron Microscopy Public Image Archive datasets in a machine-learning compatible structure. The Class Aggregator for Key Electron-microscopy Data (CAKED) package is designed to seamlessly facilitate the training of machine learning models on electron microscopy data, including electron-cryo-microscopy-specific data augmentation and labelling. These packages may be utilised independently or as building blocks in workflows. All are available in open source repositories and designed to be easily extensible to facilitate more advanced workflows if required.
Affinity-VAE for disentanglement, clustering and classification of objects in multidimensional image data
Sep 09, 2022

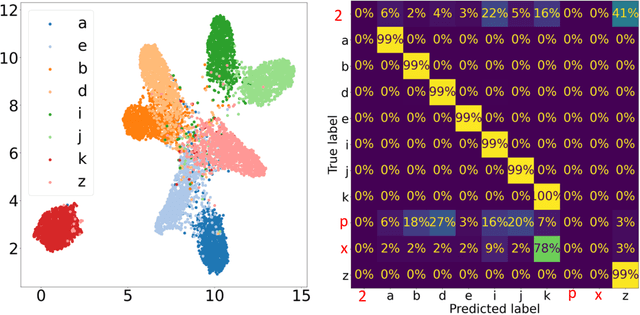

Abstract:In this work we present affinity-VAE: a framework for automatic clustering and classification of objects in multidimensional image data based on their similarity. The method expands on the concept of $\beta$-VAEs with an informed similarity-based loss component driven by an affinity matrix. The affinity-VAE is able to create rotationally-invariant, morphologically homogeneous clusters in the latent representation, with improved cluster separation compared with a standard $\beta$-VAE. We explore the extent of latent disentanglement and continuity of the latent spaces on both 2D and 3D image data, including simulated biological electron cryo-tomography (cryo-ET) volumes as an example of a scientific application.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge