Anna Oviedova
crossMoDA Challenge: Evolution of Cross-Modality Domain Adaptation Techniques for Vestibular Schwannoma and Cochlea Segmentation from 2021 to 2023
Jun 13, 2025Abstract:The cross-Modality Domain Adaptation (crossMoDA) challenge series, initiated in 2021 in conjunction with the International Conference on Medical Image Computing and Computer Assisted Intervention (MICCAI), focuses on unsupervised cross-modality segmentation, learning from contrast-enhanced T1 (ceT1) and transferring to T2 MRI. The task is an extreme example of domain shift chosen to serve as a meaningful and illustrative benchmark. From a clinical application perspective, it aims to automate Vestibular Schwannoma (VS) and cochlea segmentation on T2 scans for more cost-effective VS management. Over time, the challenge objectives have evolved to enhance its clinical relevance. The challenge evolved from using single-institutional data and basic segmentation in 2021 to incorporating multi-institutional data and Koos grading in 2022, and by 2023, it included heterogeneous routine data and sub-segmentation of intra- and extra-meatal tumour components. In this work, we report the findings of the 2022 and 2023 editions and perform a retrospective analysis of the challenge progression over the years. The observations from the successive challenge contributions indicate that the number of outliers decreases with an expanding dataset. This is notable since the diversity of scanning protocols of the datasets concurrently increased. The winning approach of the 2023 edition reduced the number of outliers on the 2021 and 2022 testing data, demonstrating how increased data heterogeneity can enhance segmentation performance even on homogeneous data. However, the cochlea Dice score declined in 2023, likely due to the added complexity from tumour sub-annotations affecting overall segmentation performance. While progress is still needed for clinically acceptable VS segmentation, the plateauing performance suggests that a more challenging cross-modal task may better serve future benchmarking.
A Clinical Guideline Driven Automated Linear Feature Extraction for Vestibular Schwannoma
Oct 30, 2023Abstract:Vestibular Schwannoma is a benign brain tumour that grows from one of the balance nerves. Patients may be treated by surgery, radiosurgery or with a conservative "wait-and-scan" strategy. Clinicians typically use manually extracted linear measurements to aid clinical decision making. This work aims to automate and improve this process by using deep learning based segmentation to extract relevant clinical features through computational algorithms. To the best of our knowledge, our study is the first to propose an automated approach to replicate local clinical guidelines. Our deep learning based segmentation provided Dice-scores of 0.8124 +- 0.2343 and 0.8969 +- 0.0521 for extrameatal and whole tumour regions respectively for T2 weighted MRI, whereas 0.8222 +- 0.2108 and 0.9049 +- 0.0646 were obtained for T1 weighted MRI. We propose a novel algorithm to choose and extract the most appropriate maximum linear measurement from the segmented regions based on the size of the extrameatal portion of the tumour. Using this tool, clinicians will be provided with a visual guide and related metrics relating to tumour progression that will function as a clinical decision aid. In this study, we utilize 187 scans obtained from 50 patients referred to a tertiary specialist neurosurgical service in the United Kingdom. The measurements extracted manually by an expert neuroradiologist indicated a significant correlation with the automated measurements (p < 0.0001).
Boundary Distance Loss for Intra-/Extra-meatal Segmentation of Vestibular Schwannoma
Aug 09, 2022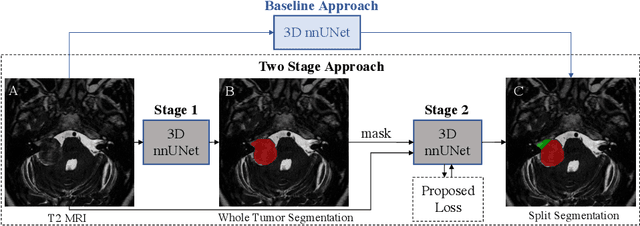
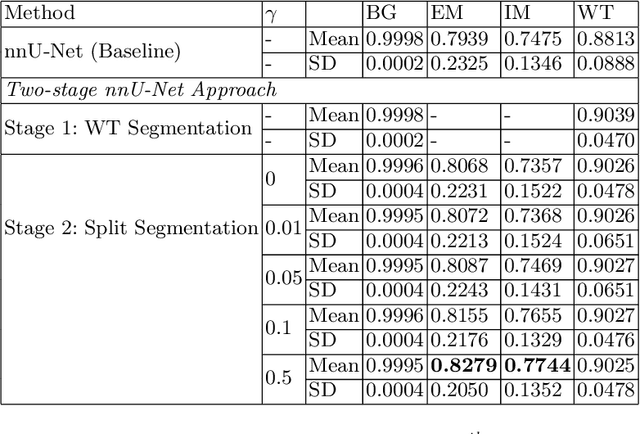
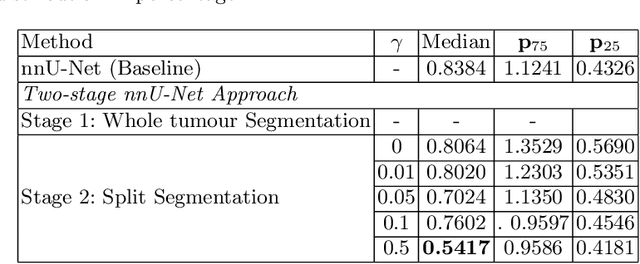
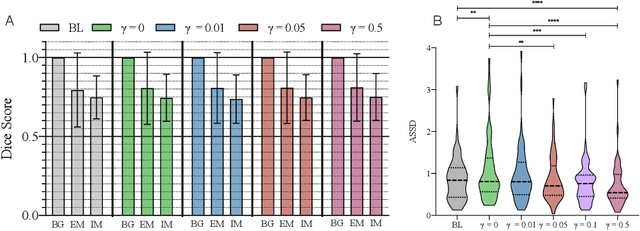
Abstract:Vestibular Schwannoma (VS) typically grows from the inner ear to the brain. It can be separated into two regions, intrameatal and extrameatal respectively corresponding to being inside or outside the inner ear canal. The growth of the extrameatal regions is a key factor that determines the disease management followed by the clinicians. In this work, a VS segmentation approach with subdivision into intra-/extra-meatal parts is presented. We annotated a dataset consisting of 227 T2 MRI instances, acquired longitudinally on 137 patients, excluding post-operative instances. We propose a staged approach, with the first stage performing the whole tumour segmentation and the second stage performing the intra-/extra-meatal segmentation using the T2 MRI along with the mask obtained from the first stage. To improve on the accuracy of the predicted meatal boundary, we introduce a task-specific loss which we call Boundary Distance Loss. The performance is evaluated in contrast to the direct intrameatal extrameatal segmentation task performance, i.e. the Baseline. Our proposed method, with the two-stage approach and the Boundary Distance Loss, achieved a Dice score of 0.8279+-0.2050 and 0.7744+-0.1352 for extrameatal and intrameatal regions respectively, significantly improving over the Baseline, which gave Dice score of 0.7939+-0.2325 and 0.7475+-0.1346 for the extrameatal and intrameatal regions respectively.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge