Simona Vatrano
Automatic Labels are as Effective as Manual Labels in Biomedical Images Classification with Deep Learning
Jun 20, 2024Abstract:The increasing availability of biomedical data is helping to design more robust deep learning (DL) algorithms to analyze biomedical samples. Currently, one of the main limitations to train DL algorithms to perform a specific task is the need for medical experts to label data. Automatic methods to label data exist, however automatic labels can be noisy and it is not completely clear when automatic labels can be adopted to train DL models. This paper aims to investigate under which circumstances automatic labels can be adopted to train a DL model on the classification of Whole Slide Images (WSI). The analysis involves multiple architectures, such as Convolutional Neural Networks (CNN) and Vision Transformer (ViT), and over 10000 WSIs, collected from three use cases: celiac disease, lung cancer and colon cancer, which one including respectively binary, multiclass and multilabel data. The results allow identifying 10% as the percentage of noisy labels that lead to train competitive models for the classification of WSIs. Therefore, an algorithm generating automatic labels needs to fit this criterion to be adopted. The application of the Semantic Knowledge Extractor Tool (SKET) algorithm to generate automatic labels leads to performance comparable to the one obtained with manual labels, since it generates a percentage of noisy labels between 2-5%. Automatic labels are as effective as manual ones, reaching solid performance comparable to the one obtained training models with manual labels.
Benchmarking Hierarchical Image Pyramid Transformer for the classification of colon biopsies and polyps in histopathology images
May 24, 2024Abstract:Training neural networks with high-quality pixel-level annotation in histopathology whole-slide images (WSI) is an expensive process due to gigapixel resolution of WSIs. However, recent advances in self-supervised learning have shown that highly descriptive image representations can be learned without the need for annotations. We investigate the application of the recent Hierarchical Image Pyramid Transformer (HIPT) model for the specific task of classification of colorectal biopsies and polyps. After evaluating the effectiveness of TCGA-learned features in the original HIPT model, we incorporate colon biopsy image information into HIPT's pretraining using two distinct strategies: (1) fine-tuning HIPT from the existing TCGA weights and (2) pretraining HIPT from random weight initialization. We compare the performance of these pretraining regimes on two colorectal biopsy classification tasks: binary and multiclass classification.
Automated risk classification of colon biopsies based on semantic segmentation of histopathology images
Sep 16, 2021
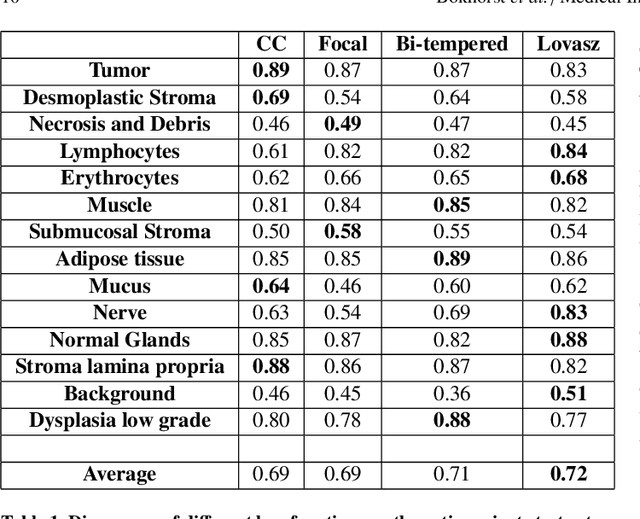

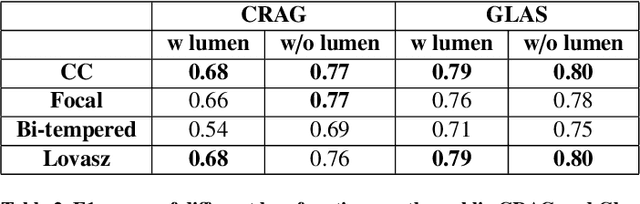
Abstract:Artificial Intelligence (AI) can potentially support histopathologists in the diagnosis of a broad spectrum of cancer types. In colorectal cancer (CRC), AI can alleviate the laborious task of characterization and reporting on resected biopsies, including polyps, the numbers of which are increasing as a result of CRC population screening programs, ongoing in many countries all around the globe. Here, we present an approach to address two major challenges in automated assessment of CRC histopathology whole-slide images. First, we present an AI-based method to segment multiple tissue compartments in the H\&E-stained whole-slide image, which provides a different, more perceptible picture of tissue morphology and composition. We test and compare a panel of state-of-the-art loss functions available for segmentation models, and provide indications about their use in histopathology image segmentation, based on the analysis of a) a multi-centric cohort of CRC cases from five medical centers in the Netherlands and Germany, and b) two publicly available datasets on segmentation in CRC. Second, we use the best performing AI model as the basis for a computer-aided diagnosis system (CAD) that classifies colon biopsies into four main categories that are relevant pathologically. We report the performance of this system on an independent cohort of more than 1,000 patients. The results show the potential of such an AI-based system to assist pathologists in diagnosis of CRC in the context of population screening. We have made the segmentation model available for research use on https://grand-challenge.org/algorithms/colon-tissue-segmentation/.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge